Chapter: Biology of Disease: Disorders of the Endocrine System
Pituitary Gland
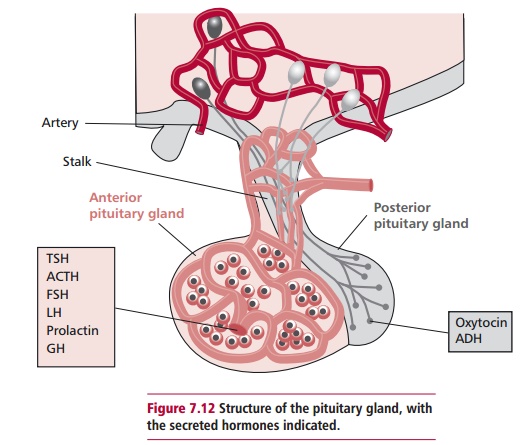
THE PITUITARY GLAND
The pituitary gland is often referred to as ‘the master gland’ given that its secretions regulate the activities of many of the other hormone-producing glands. Despite its crucial role, it weighs only about 0.5 g. The pituitary is found in a bony cavity at the base of the skull and is connected to the hypothalamus by a pituitary stalk composed of blood vessels and nerve fibers. It is composed of two lobes, an anterior pituitary or adenohypophysis and a posterior pituitary or neurohypophysis (Figure 7.12).
The anterior pituitary secretes a number of hormones (Figure 7.13) that are regulated by the release of peptides from the hypothalamus with stimulatory or inhibitory effects on the anterior pituitary. The principal peptides released by the hypothalamus reach the anterior pituitary by a portal blood circulation. These peptides include thyrotrophin releasing hormone (TRH), growth hormone releasing hormone (GHRH), gonadotrophin releasing hormone (GnRH), corticotrophin releasing hormone (CRH) and dopamine. With the exception of dopamine, all are stimulatory, controlling the release of thyroid stimulating hormone (TSH), growth hormone (GH), follicle stimulating hormone (FSH) together with luteinizing hormone (LH) and adrenocorticotrophic hormone (ACTH) respectively. Thyroid stimulating hormone targets the thyroid gland causing it to release thyroid hormones.
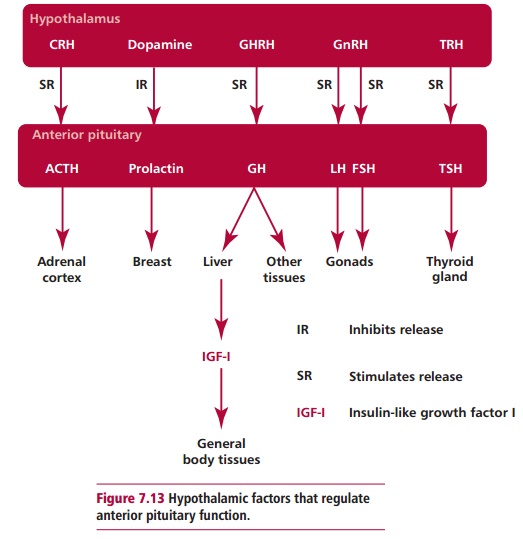
Growth hormone acts on general body tissues to promote growth and development. Follicle stimulating hormone and LH, collectively referred to as gonadotrophins, act on the testes and ovaries and are essential for reproduction. Adrenocorticotrophic hormone acts on the adrenal cortex and stimulates release of cortisol. Prolactin stimulates the female mammary glands to control lactation. The release of prolactin is inhibited by dopamine.
The posterior pituitary is composed of a collection of nerve fibers originating in the hypothalamus. It secretes two main hormones, antidiuretic hormone (ADH) or vasopressin, and oxytocin although both are made in the hypothalamus and reach the posterior pituitary in the nerve fibers of the stalk. The former stimulates the kidneys to conserve water , whereas the latter promotes uterine contractions during childbirth and stimulates the release of milk in breast feeding.
DISORDERS OF PITUITARY FUNCTION
The majority of disorders in pituitary function are caused by tumors of the gland although some pituitary diseases can lead to underproduction of its hormones. Approximately 80% of pituitary tumors are functional, that is they cause an excessive secretion of hormones. The most common secrete prolactin, and account for 50% of functional tumors. The incidence of tumors affecting other hormones is 15% GH, 10% ACTH, 4% FSH/LH with less than 1% promoting TSH secretion. Large pituitary tumors may also exert pressure on nerves, causing headaches and visual disturbances. Radiological investigations such as X-rays, computer-aided tomography (CAT) scans and magnetic resonance imaging (MRI), outlined, are important in locating tumors and estimating their sizes.
Prolactin secreting tumors, prolactinomas, cause hyperprolactinemia that, in turn, can lead to infertility in both males and females. Nonprolactin secreting tumors or pituitary stalk section by surgery, which block the dopaminergic inhibition of prolactin secretion, also result in hyperprolactinemia. Hyperprolactinemia abolishes menstruation (amenorrhea) and causes an inappropriate release of breast milk (galactorrhea) and impotence and breast development (gynecomastia) in males. Other causes of hyperprolactinemia include drugs, for example phenothiazines, that block dopamine receptors, or methyldopa that reduces the level of dopamine in the brain.
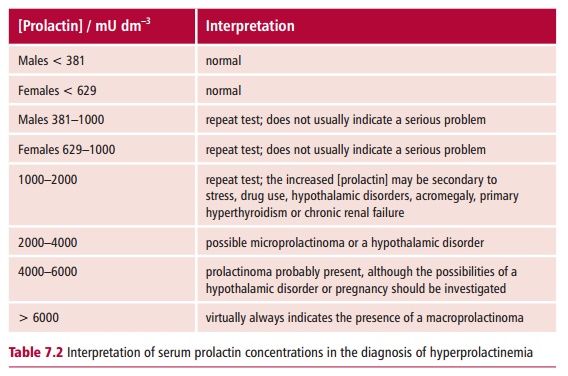
Investigating a patient with a possible prolactinoma includes assessing the concentrations of prolactin in the plasma (Table 7.2) following stimulation with TRH, although this test is not commonly used in most hospitals. In addition to TRH, prolactin is also secreted in response to stress and estrogens. Patients with a prolactinoma have plasma prolactin concentrations in excess of 2000 mU dm–3. These high values are generally not affected by TRH stimulation in individuals with prolactinomas. The first line of treatment is with a dopamine antagonist, such as bromocriptine, although surgical removal of the tumor may be necessary in cases which do not respond to drug therapy.
Disorders of oxytocin are uncommon and have little clinical significance. However, ADH release is essential for life and disorders of its release are well recognized. The release of ADH is stimulated by increased osmolality of the plasma and a decrease in blood volume detected by hypothalamic osmoreceptors and cardiac baroreceptors respectively. The role of ADH in fluid regulation is outlined. A decreased output of ADH gives rise to diabetes insipidus, characterized by excessive production of dilute urine (polyuria). Patients are constantly thirsty (polydipsia), have hypernatremia and a plasma osmolality in excess of 295 mmol kg–1.
Cranial diabetes insipidus may be caused by brain tumors, meningitis, trauma and following surgery, but can be idiopathic. Nephrogenic diabetes insipidus occurs when the kidneys fail to respond to ADH. The lack of response may be caused by drugs, such as lithium, chronic renal disease, hypercalcemia or it may be congenital. Patients with suspected diabetes insipidus are investigated by performing a fluid deprivation test in which the patient is deprived of fluid intake for a period of 8 h. In normal individuals, this results in concentrated urine with a plasma osmolality below 295 mmol kg–1. However, in patients with diabetes insipidus, the urine does not become concentrated and the plasma osmolality increases. At the end of the 8 h period, the patient is allowed to drink water and given desmopressin, a synthetic analog of ADH, after which the urine is collected hourly for a further 4 h. In cranial diabetes insipidus the urine becomes concentrated, but with nephrogenic diabetes insipidus this does not occur as the kidneys are insensitive to ADH (or desmopressin in this case). Hence the test discriminates between cranial and nephrogenic diabetes insipidus.
Patients suffering from diabetes insipidus require access to rehydrating fluids but, in each case, the underlying cause must be treated. Patients with cranial diabetes insipidus are often given desmopressin in a nasal spray or chloropropamide which increases renal sensitivity to ADH. The use of the latter drug requires careful monitoring since it can lead to hypoglycemia. Individuals with nephrogenic diabetes insipidus do not respond to analogs of ADH and often there is no suitable treatment. Especial attention to adequate water intake is essential.
A number of patients present with hypopituitarism, which is a failure to secrete one or more pituitary hormones, although this is a relatively uncommon complaint. Hypopituitarism may result from a tumor, infarction, infections, trauma affecting the pituitary or may be econdary to disorders of the hypothalamus. The clinical presentation of hypopituitarism often depends on the age of the affected individual. A decreased release of GH is often an early feature, leading to dwarfism in children . Inadequate secretion of gonadotrophins may cause amenorrhea (see above) and infertility in adult females, and loss of secondary sexual characteristics in males. lderly patients with hypopituitarism may complain of symptoms, such as hypoglycemia and hypothermia, relating to ACTH and TSH deficiencies respectively. In most cases, GH and gonadotrophin deficiencies tend to present before that of ACTH. Hyposecretion is assessed by stimulatory tests where the ability of the anterior pituitary to secrete the hormone in question is assessed following stimulation of the patient with the hypothalamic peptide or its analog. A failure to respond would suggest hypopituitarism.
Related Topics