Chapter: Biology of Disease: Disorders of the Endocrine System
Diabetes Mellitus
DIABETES MELLITUS
Diabetes mellitus is a syndrome characterized by hyperglycemia due to an absolute or relative deficiency of, and/or resistance to, insulin. This is the commonest endocrine disorder, affecting about 2% of the world’s population. Diabetes can be primary when caused directly by malfunction of one or more of the systems regulating blood glucose concentration, or secondary as a result of another disease. Primary diabetes is divided into types 1 and 2. In type 1 diabetes, also known as insulin dependent diabetes mellitus (IDDM), there is a decrease or absence of insulin production. It occurs in 15% of all diabetics and typically presents acutely during childhood or adolescence, although it can occur at any age. Patients have marked weight loss and ketoacidosis (seebelow) can occur readily. Type 1 diabetes mellitus is an autoimmune disease and antibodies that react with β cells of the islets of Langerhans in the pancreas have been demonstrated in over 90% of patients. It also has a strong association with certain histocompatibility antigens such as HLA-DR3, DR4 and certain DQ alleles. Many cases of type 1 diabetes may develop after a viral infection, such as with Coxsackie B, which initiates an autoimmune reaction that destroys the β cells of the pancreas. Type 2 diabetes is also known as noninsulin dependent diabetes mellitus (NIDDM), where insulin secretion tends to be normal or even elevated. It accounts for about 85% of all cases of diabetes, has a gradual onset and tends to occur in middle-aged and elderly individuals. Patients are less likely to develop ketoacidosis. The etiology of type 2 diabetes is still unclear but has a strong association with obesity. The disease may arise because an abnormal insulin, not recognized by its receptor, is produced, lack of insulin receptors, the presence of defective receptors or by a defective secondary messenger system linking the insulin receptor to the glucose transporter in the plasma membrane. Type 2 diabetes has a strong familial incidence. For example, if an identical twin develops type 2 diabetes there is a strong likelihood that the other twin will become diabetic.
Secondary diabetes mellitus is uncommon and is a consequence of other disorders that involve excess secretion of hormones antagonistic to insulin, such as cortisol in Cushing’s syndrome and GH in acromegaly. Damage to the pancreas following, for example, chronic pancreatitis or pancreatic surgery, may result in secondary diabetes.
Patients with diabetes mellitus suffer from a number of symptoms, including polydipsia, the production of large volumes of urine (polyuria), unexplained weight loss, blurred vision, tiredness and an increased susceptibility to infections. Diabetic patients are also susceptible to acute and chronic complications.
Acute complications of diabetes mellitus include diabetic ketoacidosis (DKA), hyperosmolar nonketotic (HONK) coma and hypoglycemia. Diabetic ketoacidosis occurs most commonly in patients with uncontrolled type 1 diabetes mellitus as a result of their failure to comply with insulin therapy or it can also be precipitated by infections, such as the common cold, when the body responds by releasing more glucose into the bloodstream and byreducing the action of insulin. The pathogenesis of DKA is outlined in Figure7.25. In DKA, glucose uptake by cells decreases whereas gluconeogenesisand glycogenolysis are both stimulated, causing severe hyperglycemia.
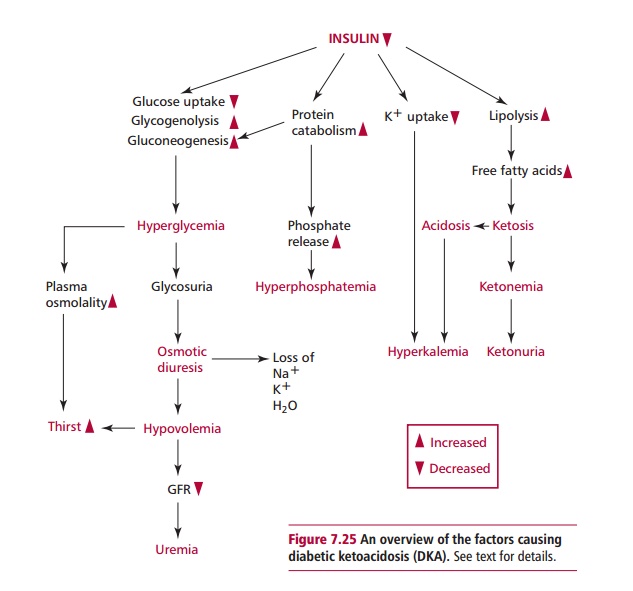
Increased breakdown of proteins occurs and the released amino acids enter gluconeogenesis or are degraded to form urea. As a result of insulin deficiency, there is a shift of both K+ and Pi from the intracellular to the extracellular compartments causing hyperkalemia and hyperphosphatemia respectively . The insulin deficiency also stimulates lipolysis producing free fatty acids (FFAs) and glycerol. In the liver, FFAs are converted to acetyl CoA which is then converted to acetoacetate and finally acetone and β-hydroxybutyrate. Acetoacetate, acetone and β-hydroxybutyrate are called ketone bodies even though β-hydroxybutyrate is not a ketone. The liver cannot utilize ketone bodies and they accumulate in the blood (ketonemia) and may be excreted in the urine (ketonuria). The ketone bodies in the blood are moderately strong acids, H+Ketone–, and react with buffers, such as NaHCO3 ,
H+Ketone– + NaHCO3 ® Na+Ketone– + H2CO3
decreasing the concentration of hydrogen carbonate but increasing that of carbonic acid producing an acidosis. The carbonic acid dissociates to CO2 and H2O and the bloodPCO2 increases. Thus the lung ventilation rate increases as the body attempts to remove the excess CO2. The ketonemia is believed to be responsible for the abdominal pain, vomiting and acidosis associated with DKA. Severe hyperglycemia in DKA exerts a high osmotic pressure causing water to move out of the cells leading to cellular dehydration. Blood volume rises and the kidneys respond with polyuria. If blood glucose levels exceed the renal threshold, glucose is lost in the urine causing glycosuria. Eventually an osmotic diuresis may occur with loss of water and electrolytes. The blood volume therefore declines further (hypovolemia) reducing the glomerular
filtration rate (GFR) and causing uremia. Dehydration and hypovolemia may stimulate the thirst center and the patient may suffer polydipsia.
Diabetic ketoacidosis is a medical emergency and is fatal in about 10% of adults and 5% of children if untreated. Death in untreated cases of DKA is due to poor tissue perfusion, acidosis and cardiovascular failure. The following approaches are taken to correct the metabolic disturbance. Isotonic saline and insulin are administered to correct the dehydration and hyperglycemia respectively. Following administration of insulin, K+ enters the cells and this may cause hypokalemia therefore it may be necessary to administer K+ supplements. In the past, severe cases of acidosis were treated by infusion of hydrogen carbonate although this approach is rarely adopted nowadays. Finally, if the DKA was precipitated by an infection, then it is necessary to identify and treat this infection.
Patients with uncontrolled type 2 diabetes mellitus may enter a HONK coma. This tends to occur in the elderly and develops over a period of days or even weeks. In these patients, the levels of insulin are sufficient to prevent ketosis but they often have severe hyperglycemia, characterized by blood glucose concentration above 35 and often in excess of 50 mmol dm–3, together with severe dehydration and high serum osmolality. A HONK coma is usually precipitated by severe illness, diuretics, dehydration and glucocorticoid therapy. Its treatment is similar to that for DKA, except that the patient is rehydrated at a slower rate and insulin requirements are lower than those for a patient with DKA.
The chronic complications of diabetes mellitus include retinopathy, cataract, atherosclerosis, nephropathy and neuropathy. Diabetics with poor blood glucose control are most susceptible to these chronic complications. It is generally accepted that increased protein glycation and the accumulation of tissue advanced glycation end products are involved in the pathogenesis of chronic complications.
Diagnosis and treatment of diabetes mellitus
Investigation of diabetes is made on the basis of clinical features and laboratory investigations. A preliminary screening test may identify the presence of urinary glucose, although this is not diagnostic of diabetes mellitus. A patient presenting with symptoms of diabetes mellitus must have a venous blood specimen taken and its glucose concentration determined. A patient is diagnosed as diabetic if the fasting plasma glucose concentration is equal to or greater than 7.0 or the random concentration is greater than 11.1 mmol dm–3. Diabetes mellitus is excluded if the fasting or random plasma glucose concentrations are less than 6.1 or 7.8 mmol dm–3 respectively. If the individual under investigation lacks the typical symptoms of diabetes then diagnosis cannot be confirmed by a single glucose determination but reconfirmed by at least one additional positive test on another day or investigated using the oral glucose tolerance test (OGTT). During the OGTT, the patient is kept on a normal diet for three days prior to the test and then fasts overnight prior to the test. A basal (fasting) venous blood sample is taken for glucose determination before the patient drinks 75 g of anhydrous glucose dissolved in a small volume of water. Blood specimens are collected after one and two hours and plasma glucose determined. Plasma glucose values greater than or equal to 7.0 mmol dm–3 for the basal sample or 11.1 mmol dm–3 for the 2 h samples are diagnostic of diabetes mellitus. Individuals with plasma glucose concentrations less than 7.0 for the basal sample or between 7.8 and 11.1 mmol dm–3 for the 2h samples are categorized as having impaired glucose tolerance (IGT). This group has an increased risk of developing cardiovascular disease . Patients with plasma glucose concentrations of 6.1 to 7.0 mmol dm–3 for the basal samples or less than 7.8 mmol dm–3 for the 2 h samples are categorized as having impaired fasting glucose (IFG) and are at risk of developing diabetes. The values given relate to venous plasma samples and are different from those for whole blood samples.
The management of diabetes mellitus aims to provide relief from symptoms and reduce the chances of developing acute and chronic complications. This includes educating the patient that diabetes is a life-long disease and affected individuals must be responsible for their own treatment. Regular clinical and laboratory assessment of the patient is required to ensure that treatment is effective, to detect early signs of treatable complications so as to reduce their progression and ensure compliance with treatment. Management involves the dietary restriction of simple sugars and of saturated fats and cholesterol and the use of complex carbohydrate and fibers. Dietary control is often accompanied by use of injected insulin or oral hypoglycemic drugs, such as sulfonylureas, in patients with type 1 and type 2 diabetes respectively. Occasionally, it may be necessary to use insulin in patients with type 2 diabetes to control blood glucose effectively. Hypoglycemic drugs act by increasing the sensitivity of A cells to glucose therefore stimulating insulin release or by increasing sensitivity of target cells to insulin. Both effects will reduce blood glucose levels. Some hypoglycemic drugs act to reduce the absorption of glucose by the GIT or reduce glycogenolysis in the liver. Diabetic patients on treatment are monitored regularly to ensure that blood glucose is kept in control. Most patients measure their own blood glucose at home regularly using kit methods based on reagent strips and a portable glucose meter (Figure 7.26) and adjust insulin dosage according to needs, perhaps following a change in diet, during illness or after exercise. The amount of glycated hemoglobin (Figure 1.14) in a patient is determined regularly to assess therapy compliance because its presence is an indicator of average glycemia over the previous 6–8 weeks. The amount of glycated hemoglobin tends to be less than 6% in nondiabetics but may exceed 10% in uncontrolled diabetes. Diabetic patients with high values have poor blood glucose control and their treatment or compliance must be reviewed.
Related Topics