Chapter: Clinical Anesthesiology: Regional Anesthesia & Pain Management: Chronic Pain Management
Neuromodulation
Neuromodulation
Electrical stimulation of the nervous system can produce analgesia in
patients with acute and chronic pain. Current may be applied transcutaneously,
epi-durally, or by electrodes implanted into the central nervous system.
Transcutaneous Electrical Nerve Stimulation
Transcutaneous electrical nerve stimulation (TENS)
is thought to produce analgesia by stimulating large afferent fibers. It may
have a role for patients with mild to moderate acute pain and those with
chronic low back pain, arthritis, and neuropathic pain. The gate theory of pain
processing suggests that the afferent input from large epicritic fibers
competes with that from the smaller pain fibers. An alternative theory proposes
that at high rates of stimulation, TENS causes conduction block in small
afferent pain fibers. With conventional TENS, electrodes are applied to the
same dermatome as the pain and are stimulated periodically by direct current
from a generator (usually for 30 min sev-eral times a day). A current of 10–30
mA with a pulse width of 50–80 µs is applied at a frequency of 80–100 Hz. Some patients whose pain is
refractory to conventional TENS respond to low-frequency TENS (acupuncture-like
TENS), which employs stimuli with a pulse width greater than 200 µs at frequencies less than 10 Hz (for 5–15
min). Unlike conventional TENS, low-frequency stimulation is at least partly
reversed by naloxone, suggesting a role for endogenous opioids. This technique
is also called dorsal column stimulation
because it was thought to produce analgesia by directly stimulat-ing large Aβ fibers in the dorsal columns of the spi-nal
cord. Proposed mechanisms include activation of descending modulating systems
and inhibition of sympathetic outflow.
Spinal Cord Stimulation
Spinal cord stimulation (SCS) may be
effec-tive for neuropathic pain; accepted indications include sympathetically
mediated pain, spinal cord lesions with localized segmental pain, phantom limb
pain, ischemic lower extremity pain due to peripheral vascular disease,
adhesive arach-noiditis, peripheral neuropathies, post-thoracotomy pain, intercostal
neuralgia, postherpetic neuralgia, angina, visceral abdominal pain, and
visceral pelvic pain. Patients with persisting pain after back surgery, which
is typically a mixed nociceptive–neuropathic disorder, also appear to benefit
from SCS.
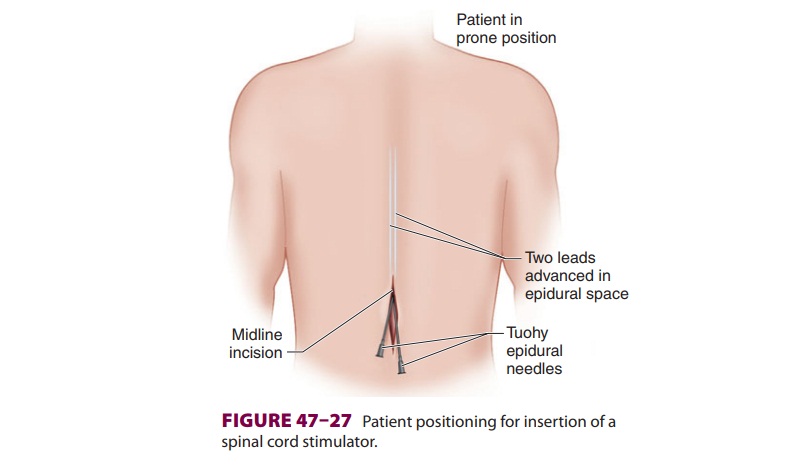
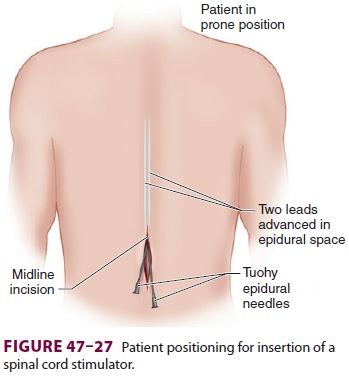
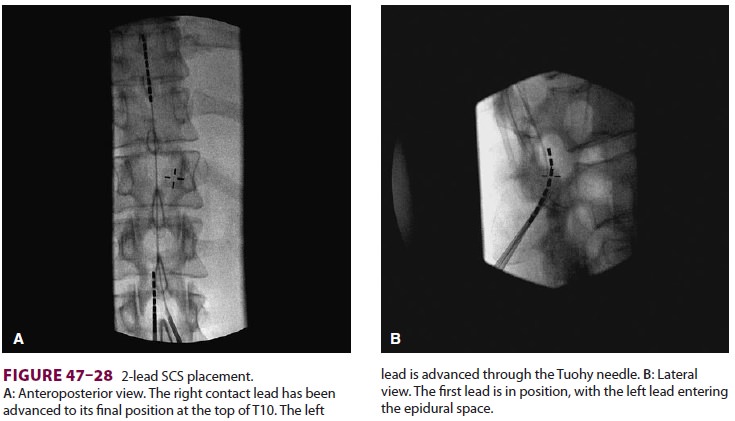
Temporary electrodes are initially placed in
the posterior epidural space and connected to an exter-nal generator to
evaluate efficacy in a 5- to 7-day trial ( Figures 47–27 and 47–28). The trial may be extended, particularly if
it allows a patient, such as one with CRPS, to tolerate more aggressive
physical therapy. If a favorable response is obtained, a fully
implantable system is inserted. Unfortunately, the effectiveness of the
technique decreases with time in some patients. Complications include
infection, lead migration, and lead breakage.
Peripheral Nerve Stimulation
Peripheral nerve stimulation (PNS) differs
from SCS in that leads are placed in close anatomic prox-imity to an injured
peripheral nerve. The leads may be placed percutaneously, with or without
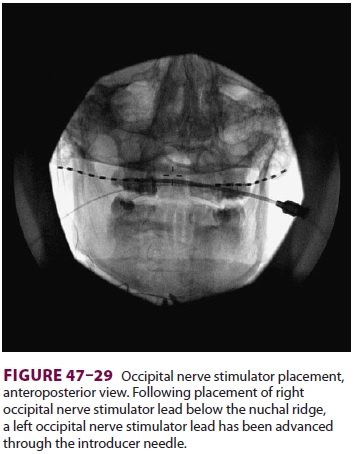
ultra-sound guidance, or surgically under direct vision of the nerve. Occipital
nerve stimulators are one form of peripheral nerve stimulator that may be
helpful in treating occipital neuralgia and migraine head-ache (Figure
47–29).
Deep Brain Stimulation
Deep brain stimulation (DBS) is used for intrac-table cancer pain and for intractable nonmalig-nant neuropathic pain. Electrodes are implanted stereotactically into the periaqueductal and peri-ventricular gray areas for nociceptive pain, usually in patients with cancer or chronic low back pain.
For neuropathic pain, the electrodes are frequently implanted into the
ventral posterolateral and ven-tral posteromedial thalamic nuclei. DBS may also
be helpful for patients with movement disorders, headache, and neuropsychiatric
disorders. The most serious complications are intracranial hemorrhage and
infection.
Related Topics