Chapter: Biotechnology Applying the Genetic Revolution: RNA-Based Technologies
MicroRNAs Are Antisense RNAs That Modulate Gene Expression
MicroRNAS
ARE ANTISENSE RNAs THAT MODULATE GENE EXPRESSION
The development from embryo to adult of the worm C. elegans requires RNAi to turn off genes at appropriate times. In this case, RNAi is not triggered by intrusion of external sequences such as transgenes or viruses. During development, small noncoding RNA molecules known as microRNAs (miRNA) are transcribed from the worm’s own genome. These miRNAs regulate gene expression by blocking the translation of developmentally appropriate target mRNA. MicroRNAs, first identified in C. elegans, have now been identified in various plants and animals, including humans. RNAi induced by miRNAs is similar
to the mechanism described
above. The mRNA targets are identified by antisense, that is, the miRNA has
sequences that are complementary to the part of the target mRNA. Some miRNAs
bind to the target mRNA and block the initiation of translation. In other
cases, the miRNA binds to the 3′ UTR region of the mRNA.
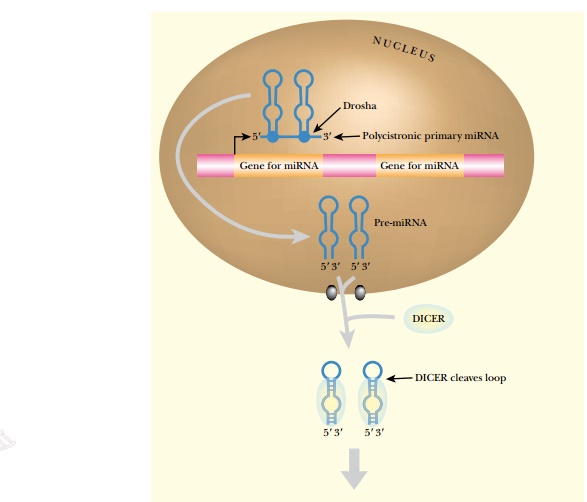
MicroRNAs are transcribed as
longer precursor molecules, pre-microRNAs,
of approximately 70 nucleotides in length. In Drosophila, miRNAs are transcribed as polycistronic messages that
are first cleaved by an endonuclease called Drosha. In plants, the
pre-microRNAs can be even longer, up to 300 nucleotides. Dicer cleaves the
plant pre-microRNA into segments of approximately 20 nucleotides. After
cleavage of the precursor, the released miRNA forms a stem-loop structure by
complementary base pairing. Dicer recognizes the stem-loop and cleaves the loop
structure, thus separating the strands. The RISC complex then unravels the two
strands. The miRNA found in animals such as C.
elegans can tolerate a few mismatched base pairs within the binding domain.
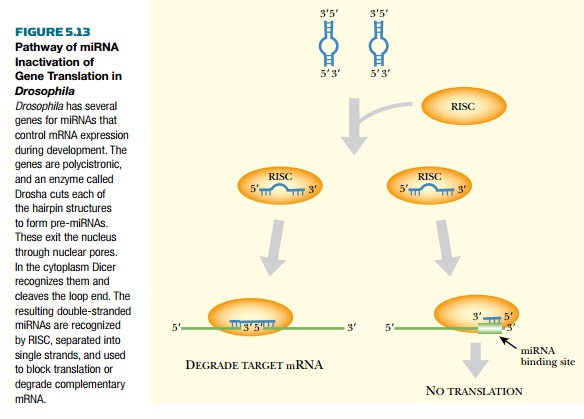
In animals, the antisense miRNA strand blocks translation of the target mRNA
(Fig. 5.13), which is not degraded. In contrast, in plants, microRNA must have
perfect matches and relies on RISC-mediated recognition and cleavage to degrade
the target mRNA.
Related Topics