Chapter: Basic & Clinical Pharmacology : Antiprotozoal Drugs
Malaria: Chloroquine
CHLOROQUINE
Chloroquine has been
the drug of choice for both treatment and chemoprophylaxis of malaria since the
1940s, but its usefulness against P
falciparum has been seriously compromised by drug resistance. It remains
the drug of choice in the treatment of sensi-tive P falciparum and other species of human malaria parasites.
Chemistry & Pharmacokinetics
Chloroquine is a synthetic 4-aminoquinoline (Figure 52–2) for-mulated as the phosphate salt for oral use. It is rapidly and almost completely absorbed from the gastrointestinal tract, reaches maxi-mum plasma concentrations in about 3 hours, and is rapidly dis-tributed to the tissues. It has a very large apparent volume of distribution of 100–1000 L/kg and is slowly released from tissues and metabolized. Chloroquine is principally excreted in the urine with an initial half-life of 3–5 days but a much longer terminal elimination half-life of 1–2 months.
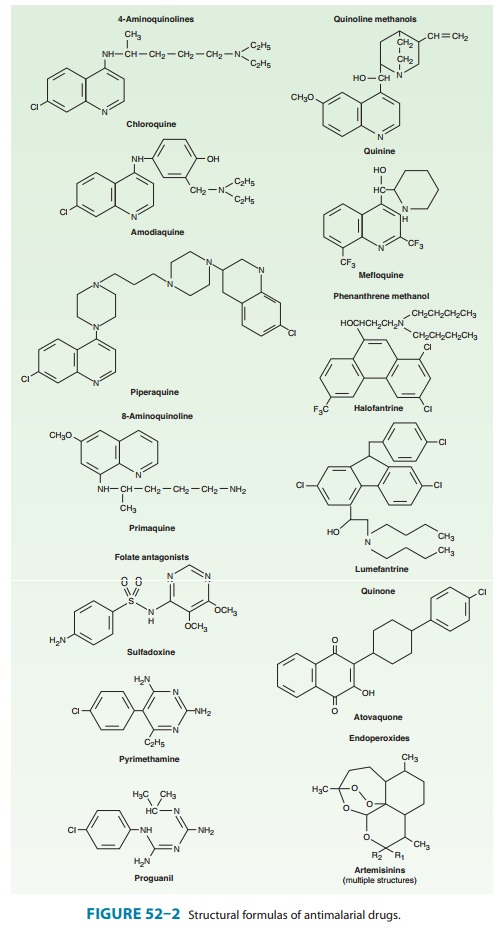
Antimalarial Action & Resistance
When not limited by resistance, chloroquine is a highly effective blood schizonticide. It is also moderately effective against gameto-cytes of P vivax, P ovale, and P malariae but not against those of P falciparum. Chloroquine is not active against liver stage para-sites. Chloroquine probably acts by concentrating in parasite food vacuoles, preventing the biocrystallization of the hemoglobin breakdown product, heme, into hemozoin, and thus eliciting parasite toxicity due to the buildup of free heme.
Resistance to
chloroquine is now very common among strains of P falciparum and uncommon but increasing for P vivax. In Pfalciparum, mutations
in a putative transporter, PfCRT, have beencorrelated with resistance.
Chloroquine resistance can be reversed by certain agents, including verapamil,
desipramine, and chlor-pheniramine, but the clinical value of
resistance-reversing drugs is not established.
Clinical Uses
A. Treatment
Chloroquine is the
drug of choice in the treatment of nonfalci-parum and sensitive falciparum
malaria. It rapidly terminates fever (in 24–48 hours) and clears parasitemia
(in 48–72 hours) caused by sensitive parasites. It is still used to treat
falciparum malaria in some areas with widespread resistance, in particular much
of Africa, owing to its safety, low cost, antipyretic proper-ties, and partial
activity, but continued use of chloroquine for this purpose is discouraged,
especially in nonimmune individuals. Chloroquine has been replaced by other
drugs, principally artemisinin-based combination therapies, as the standard
therapy to treat falciparum malaria in most endemic countries.Chloroquine does
not eliminate dormant liver forms of P
vivax and P ovale, and for that
reason primaquine must be added for the radical cure of these species.
B. Chemoprophylaxis
Chloroquine is the
preferred chemoprophylactic agent in malari-ous regions without resistant
falciparum malaria. Eradication of P
vivax and P ovale requires a
course of primaquine to clearhepatic stages.
C. Amebic Liver Abscess
Chloroquine reaches
high liver concentrations and may be used for amebic abscesses that fail
initial therapy with metronidazole .
Adverse Effects
Chloroquine is usually
very well tolerated, even with prolonged use. Pruritus is common, primarily in
Africans. Nausea, vomit-ing, abdominal pain, headache, anorexia, malaise,
blurring of vision, and urticaria are uncommon. Dosing after meals may reduce
some adverse effects. Rare reactions include hemolysis in glucose-6-phosphate
dehydrogenase (G6PD)-deficient persons, impaired hearing, confusion, psychosis,
seizures, agranulocytosis, exfoliative dermatitis, alopecia, bleaching of hair,
hypotension,
The
long-term administration of high doses of chloro-quine for rheumatologic
diseases can result in irreversible
ototoxicity, retinopathy, myopathy, and peripheral neuropathy. These
abnormalities are rarely if ever seen with stan-dard-dose weekly
chemoprophylaxis, even when given for pro-longed periods. Large intramuscular
injections or rapid intravenous infusions of chloroquine hydrochloride can
result in severe hypotension and respiratory and cardiac arrest. Parenteral
admin-istration of chloroquine is best avoided, but if other drugs are not
available for parenteral use, it should be infused slowly.
Contraindications & Cautions
Chloroquine
is contraindicated in patients with psoriasis or por-phyria, in whom it may
precipitate acute attacks of these diseases. It should generally not be used in
those with retinal or visual field abnormalities or myopathy. Chloroquine
should be used with cau-tion in patients with a history of liver disease or
neurologic or hematologic disorders. The antidiarrheal agent kaolin and
cal-cium- and magnesium-containing antacids interfere with the absorption of
chloroquine and should not be co-administered with the drug. Chloroquine is
considered safe in pregnancy and for young children.
Related Topics