Chapter: Medical Surgical Nursing: Assessment and Management of Patients With Hepatic Disorders
Hepatic Encephalopathy and Coma - Hepatic Dysfunction
HEPATIC
ENCEPHALOPATHY AND COMA
Hepatic encephalopathy, a life-threatening complication ofliver disease, occurs with profound liver failure and may result from the accumulation of ammonia and other toxic metabolites in the blood. Hepatic coma represents the most advanced stage of hepatic encephalopathy. Some researchers describe a false or weak neurotransmitter as a cause, but the exact mechanism is not fully understood. These false neurotransmitters may be generated from an intestinal source and result in the precipitation of en-cephalopathy. Many other theories exist about the causes of en-cephalopathy, including excess tryptophan and its metabolites, and endogenous benzodiazepines or opiates. Benzodiazepine-like chemicals (compounds) have been detected in the plasma and cerebrospinal fluid of patients with hepatic encephalopathy due to cirrhosis (Bacon & Di Bisceglie, 2000).
Portal-systemic
encephalopathy, the most common type of he-patic encephalopathy, occurs
primarily in patients with cirrhosis with portal hypertension and
portal-systemic shunting.
Pathophysiology
Ammonia
accumulates because damaged liver cells fail to detox-ify and convert to urea
the ammonia that is constantly entering the bloodstream. Ammonia enters the
bloodstream as a result of its absorption from the GI tract and its liberation
from kidney and muscle cells. The increased ammonia concentration in the blood
causes brain dysfunction and damage, resulting in hepatic encephalopathy.
Circumstances
that increase serum ammonia levels tend to aggravate or precipitate hepatic
encephalopathy. The largest source of ammonia is the enzymatic and bacterial
digestion of dietary and blood proteins in the GI tract. Ammonia from these
sources is increased as a result of GI bleeding (ie, bleeding esophageal
varices or chronic GI bleeding), a high-protein diet, bacterial infections, and
uremia. The ingestion of ammonium salts also increases the blood ammonia level.
In the presence of alkalosis or hypokalemia, increased amounts of ammonia are
absorbed from the GI tract and from the renal tubular fluid. Conversely, serum
ammonia is decreased by elimination of pro-tein from the diet and by the
administration of antibiotic agents, such as neomycin sulfate, that reduce the
number of intestinal bacteria capable of converting urea to ammonia (Dudek,
2001).
Other
factors unrelated to increased serum ammonia levels that may cause hepatic
encephalopathy in susceptible patients include excessive diuresis, dehydration,
infections, surgery, fever, and some medications (sedative agents,
tranquilizers, analgesic agents, and diuretic medications that cause potassium
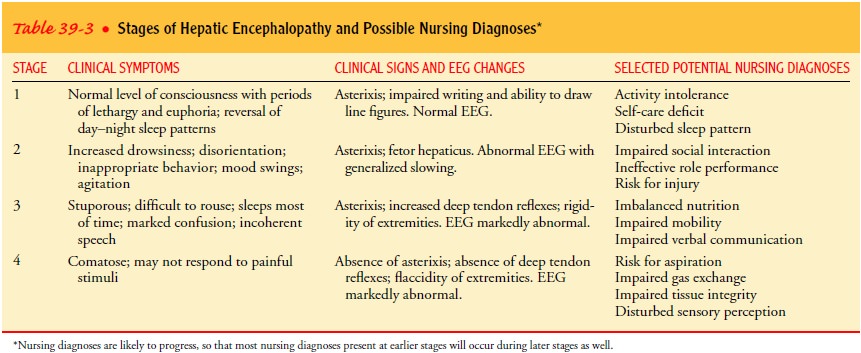
loss). Table 39-3 presents the stages of hepatic encephalopathy, common signs
and symptoms, and potential nursing diagnoses for each stage.
Clinical Manifestations
The
earliest symptoms of hepatic encephalopathy include minor mental changes and motor
disturbances. The patient appears slightly confused, has alterations in mood,
becomes unkempt, and has altered sleep patterns. The patient tends to sleep
during the day and have restlessness and insomnia at night. As hepatic
en-cephalopathy progresses, the patient may be difficult to awaken.
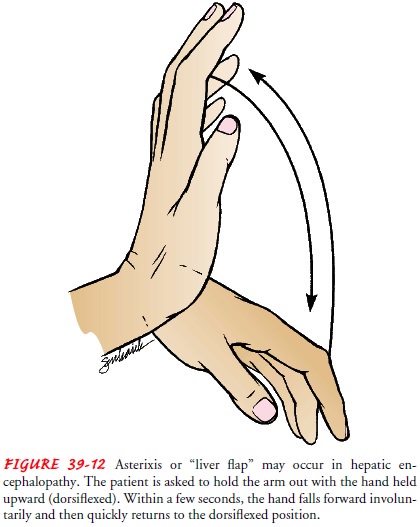
Asterixis (flapping tremor of the hands) may occur
(Fig. 39-12).Simple tasks, such as handwriting, become difficult. A handwriting
or drawing sample (eg, star figure), taken daily, may provide graphic evidence
of progression or reversal of hepatic encephalopathy. In-ability to reproduce a
simple figure (Fig. 39-13) is referred to as con-structional apraxia. In the early stages of hepatic
encephalopathy,the deep tendon reflexes are hyperactive; with worsening of he-patic
encephalopathy, these reflexes disappear and the extremities may become
flaccid.
Assessment and Diagnostic Findings
The
electroencephalogram (EEG) shows generalized slowing, an increase in the
amplitude of brain waves, and characteristic tripha-sic waves. Occasionally, fetor hepaticus, a sweet, slightly
fecal odor to the breath presumed to be of intestinal origin may be noticed.
The odor has also been described as similar to that of freshly mowed grass,
acetone, or old wine. Fetor hepaticus is prevalent with extensive collateral
portal circulation in chronic liver disease. In a more advanced stage, there
are gross disturbances of con-sciousness and the patient is completely
disoriented with respect to time and place. With further progression of the disorder,
the pa-tient lapses into frank coma and may have seizures. Approximately 35% of
all patients with cirrhosis of the liver die in hepatic coma.
Medical Management
Lactulose (Cephulac) is administered to reduce serum ammo-nia levels. It acts by several mechanisms that promote the ex-cretion of ammonia in the stool: (1) ammonia is kept in the ionized state, resulting in a fall in colon pH, reversing the nor-mal passage of ammonia from the colon to the blood; (2) evac-uation of the bowel takes place, which decreases the ammonia absorbed from the colon; and (3) the fecal flora are changed to organisms that do not produce ammonia from urea. Two or three soft stools per day are desirable; this indicates that lactu-lose is performing as intended.
Possible
side effects include intestinal bloating and cramps, which usually disappear
within a week. To mask the sweet taste,to which some patients object, lactulose
can be diluted with fruit juice. The patient is closely monitored for
hypokalemia and de-hydration. Other laxatives are not prescribed during
lactulose ad-ministration because their effects would disturb dosage
regulation. Lactulose can be administered by nasogastric tube or enema for
patients who are comatose or in whom oral administration is con-traindicated or
impossible.
Other
aspects of management include intravenous admin-istration of glucose to
minimize protein breakdown, adminis-tration of vitamins to correct deficiencies,
and correction of electrolyte imbalances (especially potassium). Additional
prin-ciples of management of hepatic encephalopathy include the following:
•
Therapy is directed toward treating or removing the
cause.
•
Neurologic status is assessed frequently. A daily
record is kept of handwriting and performance in arithmetic to mon-itor mental
status.
•
Fluid intake and output and body weight are
recorded each day.
•
Vital signs are measured and recorded every 4
hours.
•
Potential sites of infection (peritoneum, lungs)
are assessed frequently, and abnormal findings are reported promptly.
•
Serum ammonia level is monitored daily.
•
Protein intake is restricted in patients who are
comatose or who have encephalopathy that is refractory to lactulose and
antibiotic therapy (Chart 39-4).
•
Reduction in the absorption of ammonia from the GI
tract is accomplished by the use of gastric suction, enemas, or oral
antibiotics.
•
Electrolyte status is monitored and corrected if
abnormal.
•
Sedatives, tranquilizers, and analgesic medications
are dis-continued.
•
Benzodiazepine antagonists (flumazenil [Romazicon])
may be administered to improve encephalopathy whether or not the patient has
previously taken benzodiazepines.

Nursing Management
The nurse is responsible for maintaining a safe environment to prevent injury, bleeding, and infection. The nurse administers the prescribed treatments and monitors the patient for the many po-tential complications.
The nurse also communicates with the patient’s
family to keep them informed about the patient’s status, and supports them by
explaining the procedures and treatments that are part of the patient’s care.
If the patient recovers from he-patic encephalopathy and coma, rehabilitation
is likely to be pro-longed. Thus, the patient and family will require
assistance to understand the causes of this severe complication and to
recognize that it may recur.
PROMOTING HOME AND COMMUNITY-BASED CARE
Teaching Patients Self-Care.
If the
patient has recovered fromhepatic encephalopathy and is to be discharged home,
the nurse instructs the family to watch for subtle signs of recurrent
en-cephalopathy. In the acute phase of hepatic encephalopathy, di-etary protein
may be reduced to 0.8 to 1.0 g/kg per day. During recovery, and in the home
situation, it is important to instruct the patient in maintenance of a
low-protein, high-calorie diet. Protein may then be added in 10-g increments
every 3 to 5 days. Any relapse is treated by a return to the previous level.
The lim-its of tolerance are usually 40 to 60 g/day (1.0 to 1.5 g/kg per day).
Continued use of lactulose in the home environment is not uncommon, and the
patient and family should monitor its efficacy and side effects closely. Use of
vegetable rather than an-imal protein may be indicated in patients whose total
daily pro-tein tolerance is less than 1 g/kg. Vegetable protein intake may
result in improved nitrogen balance without precipitating or ad-vancing hepatic
encephalopathy (Menon & Kamath, 2000; Seymour & Whelan, 1999).
Continuing Care.
Referral for
home care is warranted for the pa-tient who returns home after recovery from
hepatic encephalopa-thy. The home care nurse assesses the patient’s physical
and mental status and collaborates closely with the physician. The home visit
also provides an opportunity for the nurse to assess the home environment and
the ability of the patient and fam-ily to monitor signs and symptoms and to
follow the treatment regimen. Home care visits are particularly important if
the pa-tient lives alone, because encephalopathy may affect the pa-tient’s
ability to remember or follow the treatment regimen. The nurse reinforces
previous teaching and reminds the patient and family about the importance of
dietary restrictions, close monitoring, and follow-up.
Related Topics