Chapter: 11th Chemistry : UNIT 4 : Hydrogen
Hard and Soft Water
Hard and Soft Water:
Hard water contains high amounts of mineral ions. The most common ions found in hard water are the soluble metal cations such as magnesium & calcium, though iron, aluminium, and manganese may also be found in certain areas. Presence of these metal salts in the form of bicarbonate, chloride and sulphate in water makes water ‘hard’. When hard water is boiled carbonates of magnesium and calcium present in it gets precipitated. On the other hand, water free from soluble salts of calcium and magnesium is called soft water. The hardness of water is of two types, viz., temporary hardness and permanent hardness.
Temporary Hardness and its removal:
Temporary hardness is primarily due to the presence of soluble bicarbonates of magnesium and calcium. This can be removed by boiling the hard water followed by filtration. Upon boiling, these salts decompose into insoluble carbonate which leads to their precipitation. The magnesium carbonate thus formed further hydrolysed to give insoluble magnesium hydroxide.
Ca(HCO3)2 → CaCO3 + H2O + CO2
Mg(HCO3)2 → MgCO3 + H2O + CO2
MgCO3 + H2O → Mg(OH)2 + CO2
The resulting precipitates can be removed by filtration.
Alternatively, we can use Clark’s method in which, calculated amount of lime is added to hard water containing the magnesium and calcium, and the resulting carbonates and hydroxides can be filtered-off.
Ca(HCO3)2 + Ca(OH )2 → 2CaCO3 + 2H2O
Mg (HCO3)2 + 2 Ca(OH )2 → 2CaCO3 + Mg(OH)2 +2 H2O
Permanent Hardness:
Permanent hardness of water is due to the presence of soluble salts of magnesium and calcium in the form of chlorides and sulphates in it. It can be removed by adding washing soda, which reacts with these metal (M = Ca or Mg) chlorides and sulphates in hard water to form insoluble carbonates.
MCl2 + Na2CO3 → MCO3 + 2 NaCl
MSO4 + Na2CO3 → MCO3 + Na2SO4
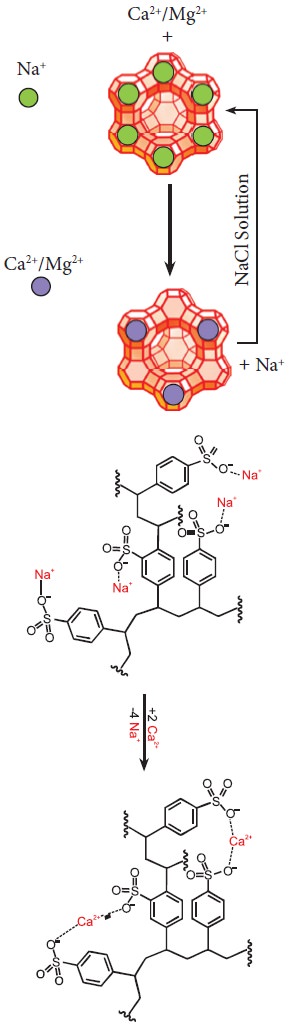
In another way to soften the hard water is by using a process called ion-exchange. That is, hardness can be removed by passing through an ion-exchange bed like zeolites or column containing ion-exchange resin. Zeolites are hydrated sodium alumino-silicates with a general formula, NaO∙Al2O3∙xSiO2∙yH2O (x = 2 to 10, y = 2 to 6). Zeolites have porous structure in which the monovalent sodium ions are loosely held and can be exchanged with hardness producing metal ions (M = Ca or Mg) in water. The complex structure can conveniently be represented as Na2-Z with sodium as exchangeable cations.
Na2-Z + M2+ → M-Z+ 2 Na+
When exhausted, the materials can be regenerated by treating with aqueous sodium chloride. The metal ions (Ca2+ and Mg2+) caught in the zeolite (or resin) are released and they get replenished with sodium ions.
M-Z + 2NaCl → Na2-Z + MCl2
Related Topics