Chapter: Medical Surgical Nursing: Assessment and Management of Patients With Diabetes Mellitus
Diabetes Management: Nutritional Management
NUTRITIONAL
MANAGEMENT
Nutrition,
diet, and weight control are the foundation of diabetes management. The most
important objective in the dietary and nutritional management of diabetes is
control of total caloric in-take to attain or maintain a reasonable body weight
and control of blood glucose levels. Success of this alone is often associated
with reversal of hyperglycemia in type 2 diabetes. However, achieving this goal
is not always easy. Because nutritional management of diabetes is so complex, a
registered dietitian who un-derstands diabetes management has the major
responsibility for this aspect of the therapeutic plan. However, the nurse and
all other members of the health care team need to be knowledgeable about
nutritional therapy and supportive of the patient who needs to implement
dietary and lifestyle changes (ADA, Expert Com-mittee on the Diagnosis and
Classification of Diabetes Mellitus, 2003). Nutritional management of the
diabetic patient includes the following goals (ADA, Evidence-Based Nutrition
Principles and Recommendations for the Treatment and Prevention of Diabetes and
Related Complications, 2003):
•
Providing all the essential food constituents (eg,
vitamins, minerals) necessary for optimal nutrition
•
Meeting energy needs
•
Achieving and maintaining a reasonable weight
•
Preventing wide daily fluctuations in blood glucose
levels, with blood glucose levels as close to normal as is safe and practical
to prevent or reduce the risk for complications
•
Decreasing serum lipid levels, if elevated, to
reduce the risk for macrovascular disease
For
patients who require insulin to help control blood glucose levels, maintaining
as much consistency as possible in the amount of calories and carbohydrates
ingested at different meal times is essential. In addition, consistency in the
approximate time inter-vals between meals, with the addition of snacks if
necessary, helps in preventing hypoglycemic reactions and in maintaining
overall blood glucose control.
For
obese diabetic patients (especially those with type 2 dia-betes), weight loss
is the key to treatment. (It is also a major factor in preventing diabetes.) In
general, overweight is considered to be a body mass index (BMI) of 25 to 29;
obesity is defined as 20% above ideal body weight or a BMI equal to or greater
than 30 (National Institutes of Health, 2000). BMI is a weight-to-height ratio
calculated by dividing body weight (in kilograms) by the square of the height
(in meters).. Obesity is associated with an increased resistance to in-sulin;
it is also a main factor in type 2 diabetes. Some obese patients who have type
2 diabetes and who require insulin or oral agents to control blood glucose
levels may be able to reduce or eliminate the need for medication through
weight loss. A weight loss as small as 10% of total weight may significantly
improve blood glucose lev-els. For obese diabetic patients who do not take insulin,
consistent meal content or timing is not as critical. Rather, decreasing the
overall caloric intake assumes more importance. However, meals should not be
skipped. Pacing food intake throughout the day places more manageable demands
on the pancreas.
Long-term
adherence to the meal plan is one of the most chal-lenging aspects of diabetes
management. For obese patients, it may be more realistic to restrict calories
only moderately. For those who have lost weight, maintaining the weight loss
may be difficult. To help these patients incorporate new dietary habits into
their lifestyles, diet education, behavioral therapy, group support, and
ongoing nutrition counseling are encouraged.
Meal Planning and Related Teaching
For all patients with diabetes, the meal plan must consider the pa-tient’s food preferences, lifestyle, usual eating times, and ethnic and cultural background. For patients using intensive insulin therapy, there may be greater flexibility in the timing and content of meals by allowing adjustments in insulin dosage for changes in eating and exercise habits.
Advances in insulin management (new insulin
analogs, insulin algorithms, insulin pumps) permit greater flexibility of
schedules than previously possible. This is in con-trast to the older concept
of maintaining a constant dose of in-sulin and requiring the patient to adjust
his or her schedule to the actions and duration of the insulin.
The
first step in preparing a meal plan is a thorough review of the patient’s diet
history to identify his or her eating habits and lifestyle. A thorough
assessment of the patient’s need for weight loss, gain, or maintenance is also
undertaken. In most instances, the person with type 2 diabetes requires weight
reduction.
In
teaching about meal planning, the clinical dietitian uses various educational
tools, materials, and approaches. Initial edu-cation addresses the importance
of consistent eating habits, the relationship of food and insulin, and the
provision of an individ-ualized meal plan. In-depth follow-up education then
focuses on management skills, such as eating at restaurants, reading food
labels, and adjusting the meal plan for exercise, illness, and special
occasions. The nurse plays an important role in communicating pertinent
information to the dietitian and reinforcing the patient’s understanding.
For
some patients, certain aspects of meal planning, such as the food exchange
system, may be difficult to learn. This may be related to limitations in the
patient’s intellectual level or to emotional issues, such as difficulty
accepting the diagnosis of di-abetes or feelings of deprivation and undue
restriction in eating. In any case, it helps to emphasize that using the
exchange sys-tem (or any food classification system) provides a new way of
thinking about food rather than a new way of eating. It is also important to
simplify information as much as possible and to provide opportunities for the
patient to practice and repeat ac-tivities and information.
CALORIC REQUIREMENTS
Calorie-controlled
diets are planned by first calculating the indi-vidual’s energy needs and
caloric requirements based on the pa-tient’s age, gender, height, and weight.
An activity element is then factored in to provide the actual number of
calories required for weight maintenance. To promote a 1- to 2-pound weight
loss per week, 500 to 1,000 calories are subtracted from the daily total. The
calories are distributed into carbohydrates, proteins, and fats, and a meal
plan is then developed.
The
1995 Exchange Lists for Meal Planning (ADA, 1995) are presented to the patient
using the appropriate amount of calories, with strict diet adherence as the
goal. Unfortunately, calorie-controlled diets are often confusing and difficult
to comply with. They require patients to measure precise portions and to eat
spe-cific foods and amounts at each meal and snack. In this instance,
developing a meal plan based on the individual’s usual eating habits and
lifestyle may be a more realistic approach to glucose control and weight loss
or weight maintenance. In both instances, the patient needs to work closely
with a registered dietitian to as-sess current eating habits and to achieve
realistic, individualized goals. The priority for a young patient with type 1
diabetes, for example, should be a diet with enough calories to maintain normal
growth and development. Some patients may be underweight at the onset of type 1
diabetes because of rapid weight loss from severe hyperglycemia. The goal with
these patients initially may be to provide a higher-calorie diet to regain lost
weight.
CALORIC DISTRIBUTION
A
diabetic meal plan also focuses on the percentage of calories to come from
carbohydrates, proteins, and fats. In general, carbohy-drate foods have the
greatest effect on blood glucose levels because they are more quickly digested
than other foods and are converted into glucose rapidly. Several decades ago it
was recommended that diabetic diets contain more calories from protein and fat
foods than from carbohydrates to reduce postprandial increases in blood glucose
levels. However, this resulted in a dietary intake inconsis-tent with the goal
of reducing the cardiovascular disease com-monly associated with diabetes (ADA,
Evidence-Based Nutrition Principles and Recommendations for the Treatment and
Preven-tion of Diabetes and Related Complications, 2003).
Carbohydrates.
The caloric
distribution currently recommendedis higher in carbohydrates than in fat and
protein. However, re-search into the appropriateness of a higher-carbohydrate
diet in patients with decreased glucose tolerance is ongoing, and
recom-mendations may change accordingly. Currently, the ADA and the American
Dietetic Association recommend that for all levels of caloric intake, 50% to
60% of calories should be derived from carbohydrates, 20% to 30% from fat, and
the remaining 10% to 20% from protein. These recommendations are also
consistent with those of the American Heart Association, American Cancer
Society, and the U.S. Department of Agriculture (2000).
Carbohydrates
consist of sugars and starches. Little scientific evidence supports the belief
that sugars, such as sucrose, promote a greater blood glucose level compared to
starches (eg, rice, pasta, or bread). Although low glycemic index diets
(described below) may reduce postprandial glucose levels, there seem to be no
clear effects on outcomes (ADA, Expert Committee on the Diagnosis and
Classification of Diabetes Mellitus, 2003). Thus, the latest nutrition
guidelines recommend that all carbohydrates be eaten in moderation to avoid
high postprandial blood glucose levels (ADA, Exchange Lists for Meal Planning,
1995). Foods high in carbohydrates, such as sucrose, are not eliminated from
the diet but should be eaten in moderation (up to 10% of total calories) because
these foods are typically high in fat and lack vitamins, minerals, and fiber.
Carbohydrate
counting is another nutritional tool used for blood glucose management because
carbohydrates are the main nutrients in food that influence blood glucose
levels. This method provides flexibility in food choices, can be less
complicated to un-derstand than the diabetic food exchange list, and allows
more ac-curate management with multiple daily injections (insulin before each
meal). However, if carbohydrate counting is not used with other meal-planning
techniques, weight gain can result. A variety of methods are used to count
carbohydrates. When developing a diabetic meal plan using carbohydrate
counting, all food sources should be considered. Once digested, 100% of carbohydrates
are converted to glucose. However, approximately 50% of protein foods (meat,
fish, and poultry) are also converted to glucose.
One
method of carbohydrate counting includes counting grams of carbohydrates. If
target goals are not reached by count-ing carbohydrates alone, protein will be
factored into the calcu-lations. This is especially true if the meal consists
of only meat, fish, and non-starchy vegetables.
An
alternative to counting grams of carbohydrate is measur-ing servings or
choices. This method is used more often by peo-ple with type 2 diabetes. It is
similar to the food exchange list and emphasizes portion control of total
servings of carbohydrate at meals and snacks. One carbohydrate serving is
equivalent to 15 g of carbohydrate. Examples of one serving are an apple 2
inches in diameter and one slice of bread. Vegetables and meat are counted as
one third of a carbohydrate serving.
Although
carbohydrate counting is now commonly used for blood glucose management with
type 1 and type 2 diabetes, it is not a perfect system. All carbohydrates, to
some extent, affect the blood glucose to different degrees, regardless of
equivalent serving size.
Fats.
The recommendations regarding fat
content of the diabeticdiet include both reducing the total percentage of
calories from fat sources to less than 30% of the total calories and limiting
the amount of saturated fats to 10% of total calories. Additional
rec-ommendations include limiting the total intake of dietary choles-terol to
less than 300 mg/day. This approach may help to reduce risk factors such as
elevated serum cholesterol levels, which are as-sociated with the development
of coronary artery disease, the lead-ing cause of death and disability among
people with diabetes.
The
meal plan may include the use of some nonanimal sources of protein (eg, legumes
and whole grains) to help reduce saturated fat and cholesterol intake. In
addition, the amount of protein in-take may be reduced in patients with early
signs of renal disease.
Fiber.
The use of fiber in diabetic
diets has received increasedattention as researchers study the effects on
diabetes of a high-carbohydrate, high-fiber diet. This type of diet plays a
role in low-ering total cholesterol and low-density lipoprotein cholesterol in
the blood. Increasing fiber in the diet may also improve blood glucose levels
and decrease the need for exogenous insulin.
There
are two types of dietary fibers: soluble and insoluble. Soluble fiber—in foods
such as legumes, oats, and some fruits— plays more of a role in lowering blood
glucose and lipid levels than does insoluble fiber, although the clinical
significance of this effect is probably small (ADA, Expert Committee on the
Diag-nosis and Classification of Diabetes Mellitus, 2003). Soluble fiber is thought
to be related to the formation of a gel in the GI tract. This gel slows stomach
emptying and the movement of food through the upper digestive tract. The
potential glucose-lowering effect of fiber may be caused by the slower rate of
glucose ab-sorption from foods that contain soluble fiber. Insoluble fiber is
found in whole-grain breads and cereals and in some vegetables. This type of
fiber plays more of a role in increasing stool bulk and preventing
constipation. Both insoluble and soluble fibers in-crease satiety, which is
helpful for weight loss.
One
risk involved in suddenly increasing fiber intake is that it may require
adjusting the dosage of insulin or oral agents to pre-vent hypoglycemia. Other
problems may include abdominal fullness, nausea, diarrhea, increased
flatulence, and constipation if fluid intake is inadequate. If fiber is added
to or increased in the meal plan, it should be done gradually and in
consultation with a dietitian. The 1995 Exchange Lists for Meal Planning (ADA,
1995) is an excellent guide for increasing fiber intake. Fiber-rich food
choices within the vegetable, fruit, and starch/bread exchanges are highlighted
in the lists.
FOOD CLASSIFICATION SYSTEMS
To
teach diet principles and to help patients in meal planning, several systems
have been developed in which foods are orga-nized into groups with common
characteristics, such as number of calories, composition of foods (ie, amount
of protein, fat, or carbohydrate in the food), or effect on blood glucose
levels.
Exchange Lists.
A commonly
used tool for nutritional manage-ment is the Exchange Lists for Meal Planning
(ADA, 1995).There
are six main exchange lists: bread/starch, vegetable, milk, meat, fruit, and
fat. Foods included on one list (in the amounts specified) contain equal
numbers of calories and are approxi-mately equal in grams of protein, fat, and
carbohydrate. Meal plans (tailored to the patient’s needs and preferences) are
based on a recommended number of choices from each exchange list. Foods on one
list may be interchanged with one another, allow-ing the patient to choose a
variety while maintaining as much consistency as possible in the nutrient
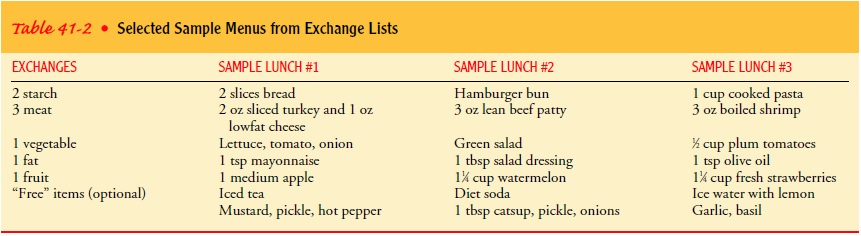
content of foods eaten. Table 41-2 presents three sample lunch menus that are
inter-changeable in terms of carbohydrate, protein, and fat content.
Exchange
list information on combination foods, such as pizza, chili, and casseroles,
and convenience foods, desserts, snack foods, and fast foods is available from
the ADA. Some food man-ufacturers and restaurants publish exchange lists that
describe their products as well. For more nutrition information, contact the
ADA.
The Food Guide Pyramid.
The Food
Guide Pyramid is anothertool used to develop meal plans. It is commonly used
for patients with type 2 diabetes who have a difficult time complying with a
calorie-controlled diet. The food pyramid consists of six food groups: (1)
bread, cereal, rice, and pasta; (2) fruits; (3) vegetables; (4) meat, poultry, fish, dry
beans, eggs, and nuts; (5) milk, yogurt, and cheese; and (6) fats, oils, and
sweets. The pyra-mid shape was chosen to emphasize that the foods in the
largest area, the base of the pyramid (starches, fruits, and vegetables), are
lowest in calories and fat and highest in fiber and should make up the basis of
the diet. For those with diabetes, as well as for the general population, 50%
to 60% of the daily caloric intake should be from these three groups. As one
moves up the pyramid, foods higher in fat (particularly saturated fat) are
illustrated; these foods should account for a smaller percentage of the daily
caloric intake. The very top of the pyramid comprises fats, oils, and sweets,
foods that should be used sparingly by people with diabetes to ob-tain weight
and blood glucose control and to reduce the risk for cardiovascular disease.
Reliance on the Food Guide Pyramid, however, may result in fluctuations in
blood glucose levels because high-carbohydrate foods may be grouped with
low-carbohydrate foods. The pyramid is appropriately used only as a first-step
teaching tool (Dixon, Cronin, & Krebs-Smith, 2001) for pa-tients learning
how to control food portions and how to identify which foods contain
carbohydrate, protein, and fat.
Glycemic Index.
One of the
main goals of diet therapy in dia-betes is to avoid sharp, rapid increases in
blood glucose levels after food is eaten. The term “glycemic index” is used to
describe how much a
given food raises the blood glucose level compared with an equivalent amount of
glucose; however, the effects on blood glucose levels and on long-term patient
outcomes have been ques-tioned (ADA, Expert Committee on the Diagnosis and
Classifi-cation of Diabetes Mellitus, 2003). Although more research is
necessary, the following guidelines can be helpful when making dietary
recommendations:
•
Combining starchy foods with protein- and
fat-containing foods tends to slow their absorption and lower the glycemic
response.
•
In general, eating foods that are raw and whole
results in a lower glycemic response than eating chopped, puréed, or cooked
foods.
•
Eating whole fruit instead of drinking juice
decreases the glycemic response because fiber in the fruit slows absorption.
•
Adding foods with sugars to the diet may produce a
lower glycemic response if these foods are eaten with foods that are more
slowly absorbed.
Patients
can create their own glycemic index by monitoring their blood glucose level
after ingesting a particular food. This can help patients improve blood glucose
levels through individualized manipulation of the diet. Many patients who use
frequent mon-itoring of blood glucose levels can use this information to adjust
their insulin doses for variations in food intake.
Other Dietary Concerns
ALCOHOL CONSUMPTION
Patients with diabetes do not need to give up alcoholic beverages entirely, but patients and health care professionals need to be aware of the potential adverse effects of alcohol specific to diabetes. In general, the same precautions regarding the use of alcohol by peo-ple without diabetes should be applied to patients with diabetes. Moderation is recommended. The main danger of alcohol con-sumption by a diabetic patient is hypoglycemia, especially for pa-tients who take insulin. Alcohol may decrease the normal physiologic reactions in the body that produce glucose (gluconeo-genesis). Thus, if a diabetic patient takes alcohol on an empty stom-ach, there is an increased likelihood that hypoglycemia will develop. In addition, excessive alcohol intake may impair the pa-tient’s ability to recognize and treat hypoglycemia and to follow a prescribed meal plan to prevent hypoglycemia. To reduce the risk of hypoglycemia, the patient should be cautioned to eat while drinking alcohol (ADA, Expert Committee on the Diagnosis and Classification of Diabetes Mellitus, 2003).
For
the person with type 2 diabetes treated with the sulfonylurea agent
chlorpropamide (Diabinese), a potential side effect of alcohol consumption is a
disulfiram (Antabuse) type of reaction, which in-volves facial flushing,
warmth, headache, nausea, vomiting, sweat-ing, or thirst within minutes of
consuming alcohol. The intensity of the reaction depends on the amount of
alcohol consumed; the reaction seems to be less common with other
sulfonylureas.
Alcohol
consumption may lead to excessive weight gain (from the high caloric content of
alcohol), hyperlipidemia, and elevated glucose levels (especially with mixed
drinks and liqueurs).
Patient
teaching regarding alcohol intake must emphasize moderation in the amount of
alcohol consumed. Lower-calorie or less sweet drinks, such as light beer or dry
wine, and food intake along with alcohol consumption are advised. For patients
with type 2 diabetes especially, incorporating the calories from alcohol into
the overall meal plan is important for weight control.
SWEETENERS
Using
sweeteners is acceptable for patients with diabetes, espe-cially if it assists
in overall dietary adherence. Moderation in the amount of sweetener used is
encouraged to avoid potential ad-verse effects. There are two main types of
sweeteners: nutritive and non-nutritive. The nutritive sweeteners contain
calories, and the non-nutritive sweeteners have few or no calories in the
amounts normally used.
Nutritive
sweeteners include fructose (fruit sugar), sorbitol, and xylitol. They are not calorie-free;
they provide calories in amounts similar to those in sucrose (table sugar).
They cause less elevation in blood sugar levels than sucrose and are often used
in “sugar-free” foods. Sweeteners containing sorbitol may have a lax-ative
effect.
Non-nutritive
sweeteners have minimal or no calories. They are used in food products and are
also available for table use. They produce minimal or no elevation in blood
glucose levels and have been approved by the Food and Drug Administration as
safe for people with diabetes. Saccharin contains no calories. Aspartame
(NutraSweet) is packaged with dextrose; it contains 4 calories per packet and
loses sweetness with heat. Acesulfame-K (Sunnette) is also packaged with
dextrose; it contains 1 calorie per packet. Sucralose (Splenda) is a newer
non-nutritive, high-intensity sweet-ener that is about 600 times sweeter than
sugar. The Food and Drug Administration has approved it for use in baked goods,
nonalcoholic beverages, chewing gum, coffee, confections, frost-ings, and
frozen dairy products.
MISLEADING FOOD LABELS
Foods
labeled “sugarless” or “sugar-free” may still provide calo-ries equal to those
of the equivalent sugar-containing products if they are made with nutritive
sweeteners. Thus, for weight loss, these products may not always be useful. In
addition, patients must not consider them “free” foods to be eaten in unlimited
quantity, because they may elevate blood glucose levels.
Foods
labeled “dietetic” are not necessarily reduced-calorie foods. They may be lower
in sodium or have other special dietary uses. Patients are advised that foods
labeled “dietetic” may still contain significant amounts of sugar or fat.
Patients
must also be taught to read the labels of “health foods”—especially
snacks—because they often contain carbohy-drates such as honey, brown sugar,
and corn syrup. In addition, these supposedly healthy snacks frequently contain
saturated veg-etable fats (eg, coconut or palm oil), hydrogenated vegetable
fats, or animal fats, which may be contraindicated in patients with elevated
blood lipid levels.
Related Topics