Chemistry - p-Block Elements-I | 12th Chemistry : UNIT 2 : p-Block Elements-I
Chapter: 12th Chemistry : UNIT 2 : p-Block Elements-I
p-Block Elements-I
p-BLOCK
ELEMENTS-I
INTRODUCTION
We have already learnt
the classification of elements into four blocks namely s, p, d and f. We have
also learnt the properties of s-block elements and their important compounds in
the XI standard. This year we learn the elements of remaining blocks, starting
with p-block elements.
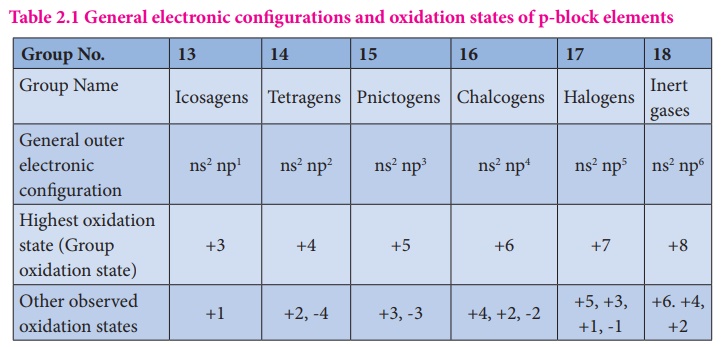
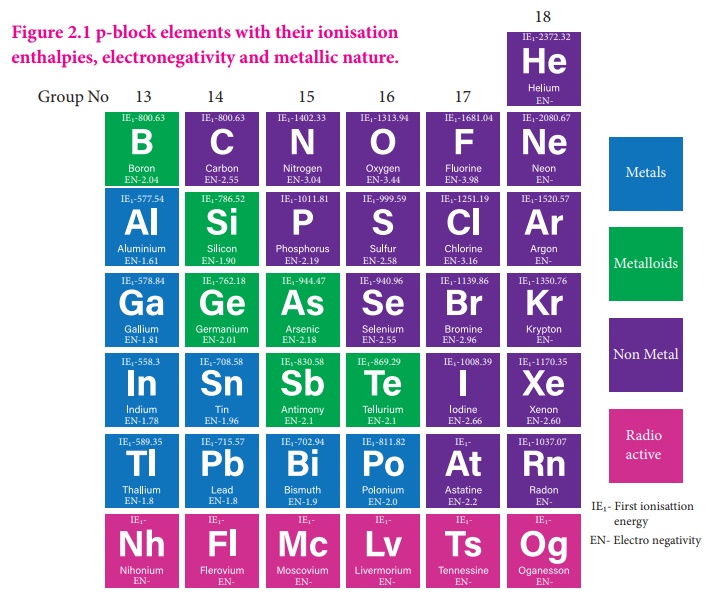
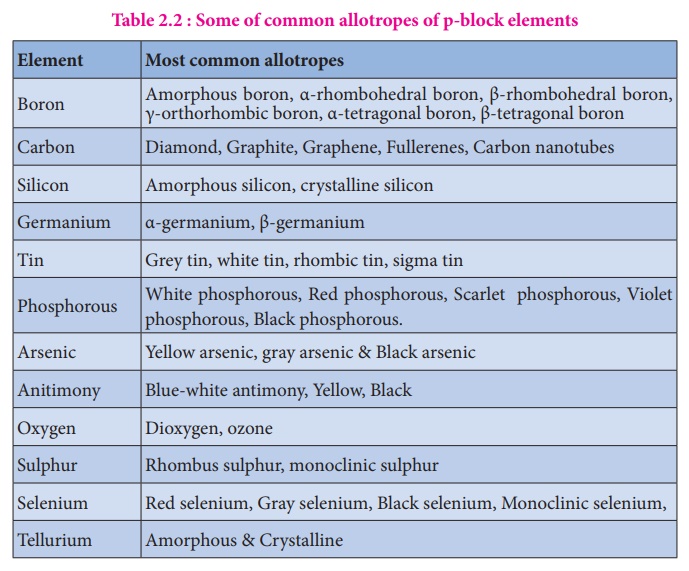
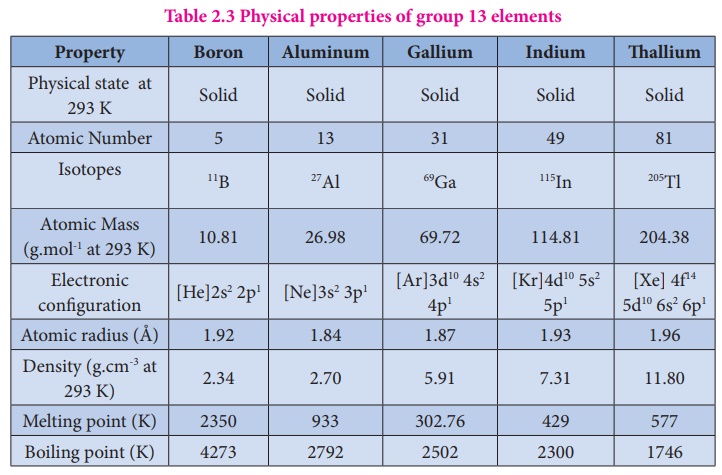
The elements in which their last electron enters the 'p' orbital, constitute the p-block elements. They are placed in 13th to 18th groups of the modern periodic table and the first member of the groups are B, C, N, O, F and He respectively. These elements have quite varied properties and this block contains nonmetals, metals and metalloids. Nonmetallic elements of this group have more varied properties than metals. The elements of this block and their compounds play an important role in our day to day life, for example, without molecular oxygen we cannot imagine the survival of living system. The most abundant metal aluminium and its alloys have plenty of applications ranging from household utensils to parts of aircraft. The semi conducting nature of elements such as silicon and germanium made a revolutionary change in the field of modern electronics. In this unit we discuss the properties of first three groups (Group 13 - 15) of p-block elements namely boron, carbon and nitrogen family elements and their important compounds.
Related Topics