Chapter: 11th Chemistry : UNIT 6 : Gaseous State
The Gas Laws
The
Gas Laws
The gas laws have played a major role in the development
of chemistry. The physical properties of all gases are governed by the gas laws
that were formulated based on the studies of the properties like pressure,
volume, etc., as a function of temperature. Before studying the gas laws in
detail, let us understand an important parameter, namely, the pressure.
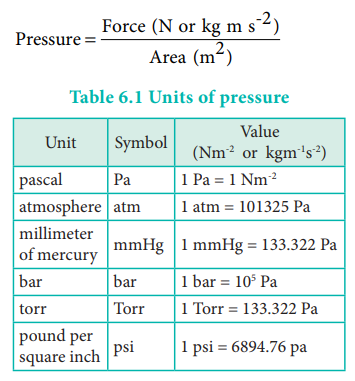
Pressure is defined as force divided by the area to which
the force is applied. The SI unit of pressure is pascal which is defined as 1
Newton per square meter (Nm-2). There are other units that are
commonly used and their relation with the SI unit is as follows.
BoyleŌĆÖs Law: Pressure-Volume Relationship
Robert Boyle performed a series of experiments to study
the relation between the pressure and volume of gases. The schematic of the
apparatus used by him is shown in figure 6.1.
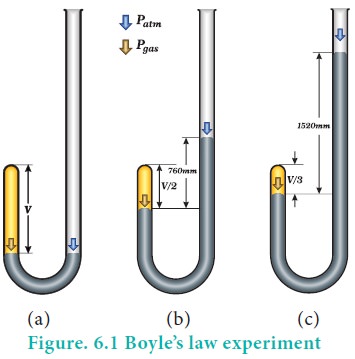
Mercury was added through the open end of the apparatus
such that the mercury level on both ends are equal as shown in the figure
6.1(a). Add more amount of mercury until the volume of the trapped air is
reduced to half of its original volume as shown in figure 6.1(b). The pressure
exerted on the gas by the addition of excess mercury is given by the difference
in mercury levels of the tube. Initially the pressure exerted by the gas is
equal to 1 atm as the difference in height of the mercury levels is zero. When
the volume is reduced to half, the difference in mercury levels increases to
760 mm. Now the pressure exerted by the gas is equal to 2 atm. It led him to
conclude that at a given temperature the volume occupied by a fixed mass of a
gas is inversely proportional to its pressure.
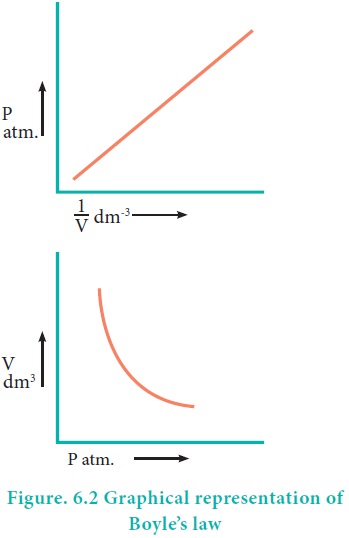
Mathematically, the BoyleŌĆÖs law can be written as

(T and n are
fixed, T-temperature, n- number
of moles)

k ŌĆō proportionality constant
When we rearrange equation 6.2.
PV = k at constant temperature and mass
BoyleŌĆÖs law is applicable to all gases regardless of their
chemical identity (provided the pressure is low). Therefore, for a given mass
of a gas under two different sets of conditions at constant temperature we can
write
P1V1 = P2V2 = k
------(6.3)
The PV relationship can be understood as follows. The
pressure is due to the force of the gas particles on the walls of the
container. If a given amount of gas is compressed to half of its volume, the
density is doubled and the number of particles hitting the unit area of the
container will be doubled. Hence, the pressure would increase twofold.
Consequence of BoyleŌĆÖs law
The pressure-density relationship can be derived from the
BoyleŌĆÖs law as shown below.
P1V1 = P2V2 (BoyleŌĆÖs law)
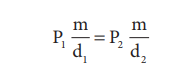
where ŌĆ£mŌĆØ is the mass, d1 and d2 are
the densities of gases at pressure P1 and P2.
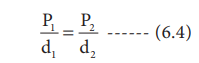
In other words, the density of a gas is directly
proportional to pressure.
All the passenger aeroplane cabins
have to be artificially pressurised. do you know why?
Most commercial aeroplanes fly at about 30,000 feet
altitude. The pressure decreases with the increase in altitude as there are
fewer molecules per unit volume of air. Hence, while at air, the pressure
around the aeroplane will be so low that one could pass out for lack of oxygen.
For this reason aeroplanes cabins are artificially pressurized.

Similarly, the effect of drop in pressure is felt as a little pain in the ears by a person while ascending a mountain in a plain. Though the external pressure drops, the internal pressure within the ear cavities remains the same. This creates an imbalance. The greater internal pressure forces the eardrum to bulge outward causing pain. The excess air within the ear cavities escapes after some time and with the help of yawning and thereby equalizing the internal and external pressure to relieve the pain.
Underwater divers are advised not to hold the
breath unnecessarily while diving. Do you know why?
In figure (6.3) let us find the missing parameters (volume
in 6.3 (b) and pressure in 6.3(c))

Solution:
According to BoyleŌĆÖs law, at constant temperature for a
given mass of gas at constant temperature,
P1V1 = P2V2 = P3V3
1 atm ├Ś 1 dm3 = 2 atm ├Ś V2 = P3 ├Ś 0.25 dm3
Ōł┤ 2 atm ├Ś V2 = 1 atm ├Ś 1 dm3
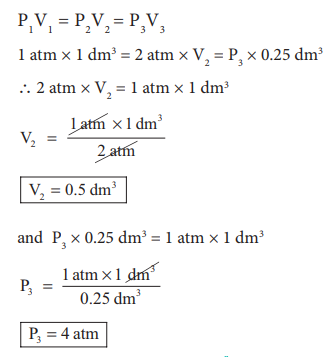
P3 = 4 atm
Charles Law (Volume-temperature relationship)
The relationship between volume of a gas and its
temperature was examined by J. A. C. Charles. He observed that for a fixed mass
of a gas at constant pressure, the volume is directly proportional to its
temperature (K). Mathematically it can be represented as (at constant P and n)
or V = kT ------- (6.5)
or V/T = Constant
If the temperature of the gas increases, the volume also
increases in direct proportion, so that V/T is a constant.
For the same system at constant pressure, one can write
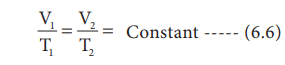
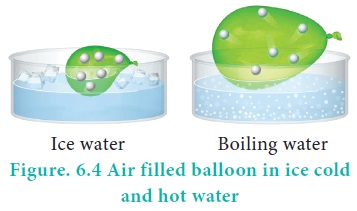
For example, if a balloon is moved from an ice cold water
bath to a boiling water bath, the temperature of the gas increases. As a
result, the gas molecules inside the balloon move faster and gas expands.
Hence, the volume increases.
Variation of volume with temperature at constant pressure
The plot of the volume of the gas against its temperature
at a given pressure is shown in the
figure 6.5. From the graph it is clear that the volume of the gas linearly
increases with temperature at a given pressure. Such lines are called isobars.
It can be expressed by the following straight line equation.
V = mT + C where T is the temperature in degree Celsius
and m & C are constants.
When T= 0 ┬║C the volume becomes V0. Hence, V0
= C and slope of the straight line m is equal to ΔV / ΔT. Therefore the above
equation can be written in the following form.
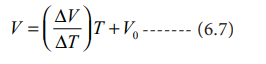
(n, P are constant)
Divide the equation 6.7 by Vo
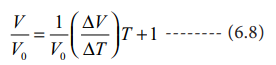
Charles and Gay Lussac found that under constant pressure,
the relative increase in volume per degree increase in temperature is same for
all gases. The relative increase in volume per ┬║ C (╬▒) is equal to 1/V0(ŌłåV/ŌłåT)
Therefore
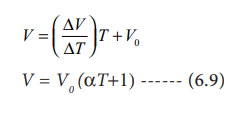
Charles found that the coefficient of expansion is
approximately equal to 1/273. It means that at constant temperature for a given
mass, for each degree rise in temperature, all gases expand by 1/273 of their
volume at 0 0C
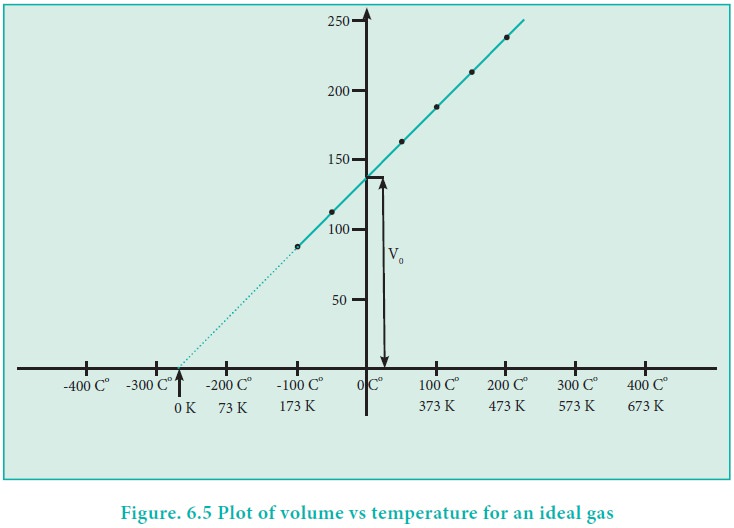
If we extrapolate the straight line in the figure 6.5
beyond the experimental measurements, the straight line intersects the
temperature axis (x-axis) at -2730 C. This shows that the volume of the gas
becomes zero at -2730 C, more precisely this temperature is -273.150 C. Beyond this
temperature the gas would have a negative volume which is physically
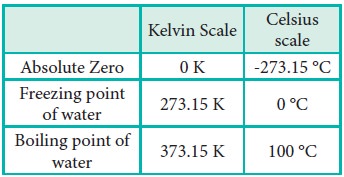
impossible. For this reason, this temperature was defined as absolute zero by
Kelvin and he proposed a new temperature scale with absolute zero as starting
point which is now called Kelvin scale. The only difference between the Kelvin
scale of temperature and Celsius scale of temperature is that the zero position
is shifted. The boiling and freezing point of water in both scales are given
below.
Example:
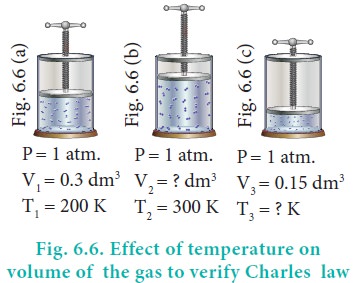
In figure 6.6 let us find the missing parameters (volume
in 6.6 (b) and temperature in 6.6(c))

Gay-LussacŌĆÖs Law (Pressure-temperature relationship)
Joseph Gay-Lussac stated that, at constant volume the
pressure of a fixed mass of a gas is directly proportional to temperature.
P ╬▒ T
or P/T = Constant k
If P1 and P2 are the pressures at
temperatures T1 and T2, respectively, then from Gay
LussacŌĆÖs law
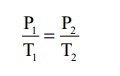
AvogadroŌĆÖs Hypothesis
Avogadro hypothesised that equal volumes of all gases
under the same conditions of temperature and pressure contain equal number of
molecules. The mathematical form of AvogadroŌĆÖs hypothesis may be expressed as
V ╬▒ n

where V1 & n1 are the volume and number of moles of a gas
and V2 & n2 are a different set of values of volume
and number of moles of the same gas at same temperature and pressure.
Related Topics