Gaseous State | Chemistry - Compressibility factor Z | 11th Chemistry : UNIT 6 : Gaseous State
Chapter: 11th Chemistry : UNIT 6 : Gaseous State
Compressibility factor Z
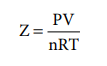
Compressibility factor Z
The deviation of real gases from ideal behaviour is measured in terms of a ratio of PV to nRT. This is termed as compressibility factor. Mathematically,
For ideal gases PV = nRT, hence the compressibility factor, Z = 1 at all temperatures and pressures. For these gases the plot of Z vs P should be a straight line parallel to the pressure axis. When a gas deviates from ideal behaviour, its Z value deviates from unity. For all gases, at very low pressures and very high temperature the compressibility factor approaches unity and they tend to behave ideally. The plot of the compressibility factor vs pressure for some common gases are shown in Figure 6.8.

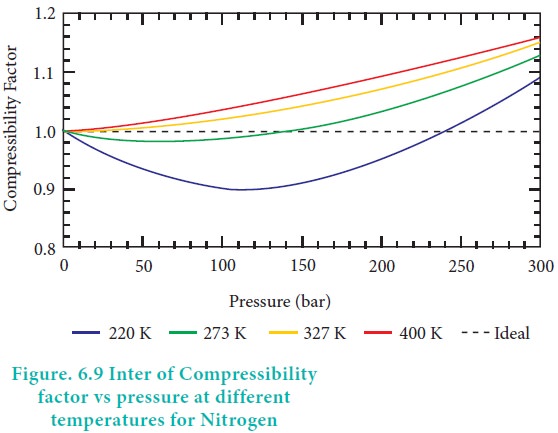
When the pressure is low, the volume of the container is very large compared to the volume of the gas molecules so that individual volume of the gas molecules can be neglected. In addition, the molecule in a gas are far apart and attractive forces are negligible. As the pressure increases, the density of gas also increases and the molecules are much closer to one another. Hence, the intermolecular force becomes significant enough to affect the motion of the molecules and the gas will not behave ideally.
At high temperatures the average kinetic energy of the molecules is very high and hence inter moleclular attractions will become insignificant. As the temperature decreases, the average kinetic energy of molecules also decreases, hence the molecular attraction is enhanced. The temperature at which a real gas obeys ideal gas law over an appreciable range of pressure is called Boyle temperature or Boyle point. The Boyle point varies with the nature of the gas. Above the Boyle point, for real gases, Z > 1, ie., the real gases show positive deviation. Below the Boyle point, the real gases first show a decrease for Z, reaches a minimum and then increases with the increase in pressure. So, it is clear that at low pressure and at high temperature, the real gases behave as ideal gases.
Related Topics