Chapter: 11th Chemistry : UNIT 6 : Gaseous State
Solved Example Problems: DaltonŌĆÖs law of partial pressures
Chemistry : Gaseous State : Mixture of gases - DaltonŌĆÖs law of partial pressures : Solved Example Problems:
Let us understand Dalton's law by solving these problems.
1. A mixture of gases contains 4.76 mole of Ne, 0.74 mole of Ar and 2.5 mole of Xe. Calculate the partial pressure of gases, if the total pressure is 2 atm. at a fixed temperature.
Solution:
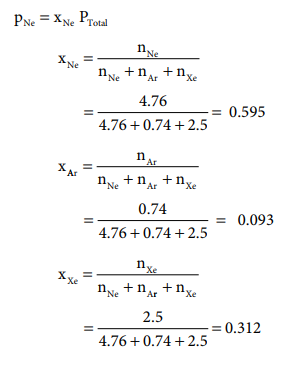
PNe = xNe PTotal = 0.595 ├Ś 2
= 1.19 atm.
PAr = xAr PTotal = 0.093 ├Ś 2
= 0.186 atm.
PXe = xXe PTotal = 0.312 ├Ś 2
= 0.624 atm.
2. An unknown gas diffuses at a rate of 0.5 time that of nitrogen at the same temperature and pressure. Calculate the molar mass of the unknown gas
Solution:

Study Material, Lecturing Notes, Assignment, Reference, Wiki description explanation, brief detail
11th Chemistry : UNIT 6 : Gaseous State : Solved Example Problems: DaltonŌĆÖs law of partial pressures |
Related Topics
11th Chemistry : UNIT 6 : Gaseous State