Chapter: Biochemistry: The Three-Dimensional Structure of Proteins
Protein Folding Dynamics: The Importance of Correct Folding
The Importance of Correct Folding
The primary structure conveys all the information necessary to produce the correct tertiary structure, but the folding process in vivo can be a bit trickier. In the protein-dense environment of the cell, proteins may begin to fold incorrectly as they are produced, or they may begin to associate with other proteins before completing their folding process. In eukaryotes, proteins may need to remain unfolded long enough to be transported across the membrane of a subcellular organelle.
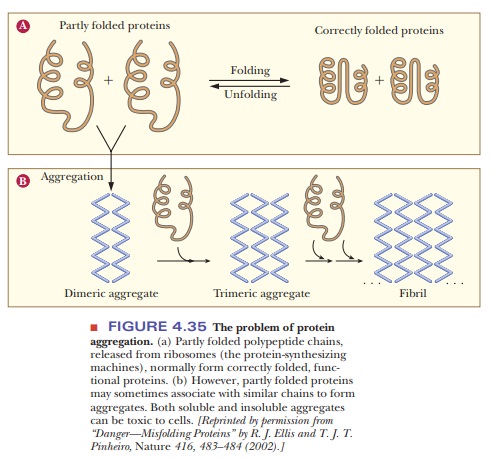
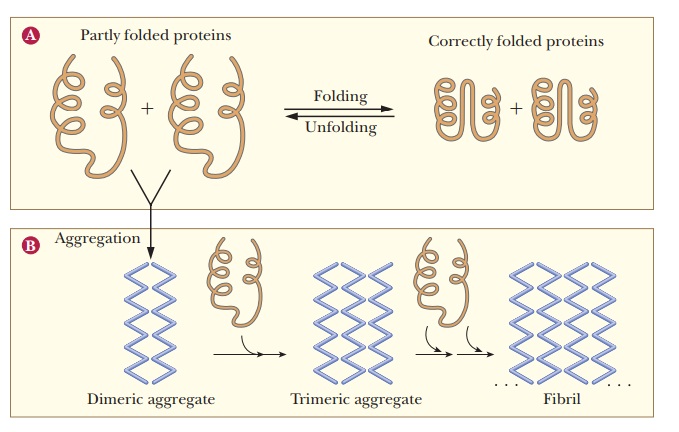
Correctly folded proteins are usually soluble in the aqueous cell environ-ment, or they are correctly attached to membranes. However, when proteins do not fold correctly, they may interact with other proteins and form aggre-gates as shown in Figure 4.35. This occurs because hydrophobic regions that should be buried inside the protein remain exposed and interact with other hydrophobic regions on other molecules. Several neurodegenerative disor-ders, such as Alzheimer’s, Parkinson’s, and Huntington’s diseases are caused by accumulation of protein deposits from such aggregates. See the following Biochemical Connections box for a description of a deadly disease caused by protein misfolding.
Related Topics