Chapter: Psychology: Consciousness
Neural Correlates of Consciousness
Neural
Correlates of Consciousness
Further insights into the
biological basis for consciousness come from studies of the so- called neural correlates of consciousness—specific
states of the brain that correspond to the exact content of someone’s conscious
experience. In one study, for example, the researchers exploited
a phenomenon known
as binocular rivalry (Tong,
Nakayama, Vaughan, and Kanwisher, 1998). In the study, one picture is
placed in front of one of aperson’s eyes and another, entirely different
picture is placed in front of her other eye. Inthis setup, the visual system is
unable to handle both stimuli at once,or to fuse the stimuli into a single
complex perception. Instead, thevisual
system seems to
flip-flop between the
stimuli so that, for
awhile, the person is aware of only one picture, then for a while
awareof only the other, and so on. Notice therefore that this is a setting in
which the physical
situation doesn’t change—the
two pictures arealways
present. What is
changing is the
person’s
consciousexperience—that is, which picture she’s aware of. This allows
us to askwhat changes take place in the brain when the experience changes.
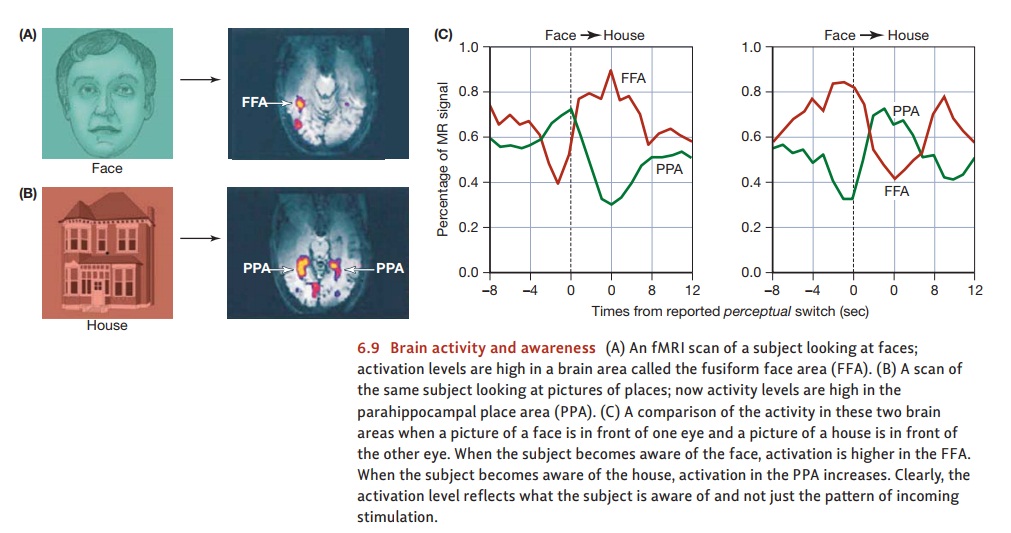
In this study, the researchers placed a picture of a face in front oneof the participant’s eyes and a picture of a house in front of the other eye. We know from many other studies that when people are looking at faces, neuroimaging reveals high levels of activity in a brain region called the fusiform face area (FFA). We also know that when people are looking at houses, there’s a lot of activity in a brain region known as the parahippocampal place area (PPA).
But what exactly does this brain
activity—in the FFA or PPA—indicate? If these brain areas respond simply to the
available stimuli, then the pattern of activity should be constant in the Tong
et al. procedure. The stimuli, after all, were present all the time. But if
these brain areas reflect the participants’ conscious perception, then activity
should fluctuate—with a change in brain activity each time the binocular
rivalry produces a new perception.
In the Tong et al. study,
participants pressed buttons to indicate at each moment which picture they were
aware of seeing—the house or the face. At the same time, the researchers used
fMRI to keep track of the activity levels in the FFA (again, normally
responsive to faces) and the PPA (normally responsive to places).
The results are summarized in
Figure 6.9. Immediately before the moments in which the participant reported a
conscious switch from seeing the face to seeing the house, activity in the FFA
went down and activity in the PPA went up. At moments in which the participant
reported the reverse switch, the activity levels in these two brain areas
showed the opposite pattern.
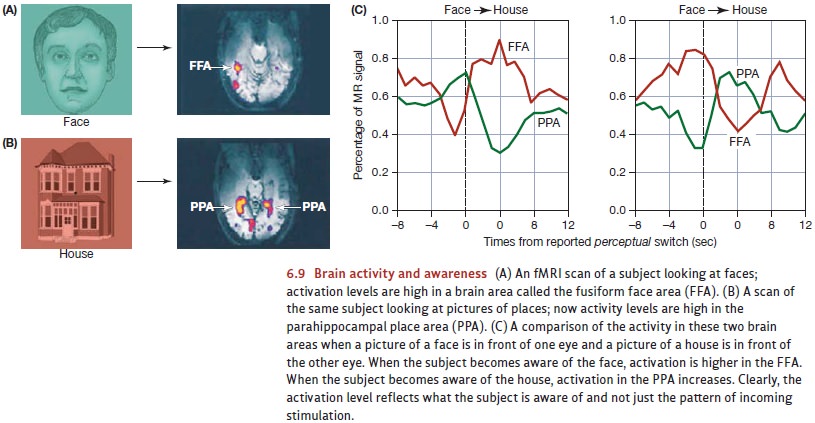
Apparently, then, activity levels
in the FFA or PPA change whenever the participant’s conscious experience
changes. Put differently, activity in the FFA doesn’t indicate “a face is in
view.” Instead, activity here seems to indicate “the participant is aware of
seeing a face.” Likewise for the PPA; activity here seems to indicate “the
participant is aware of seeing a house.” In this fashion, it does seem that
that we can use brain scans to identify some of the biological correlates of
specific conscious states. (For related results, including some with other
species, see Kim & Blake, 2005; Haynes, 2009; Koch, 2008; Logothetis, 1998;
Rees & Frith, 2007.)
A different example
concerns—remarkably—the conscious sensation of “free will.” In a classic study,
participants watched a dot moving in a circular pattern on a computer screen,
and they were asked to move their hands occasionally (Libet, 1983; also see
Haggard & Eimer, 1999; Wegner, 2002). It was up to the participants to
decide when they would move their
hands; but they were asked to note the dot’s position at the exact moment when
they
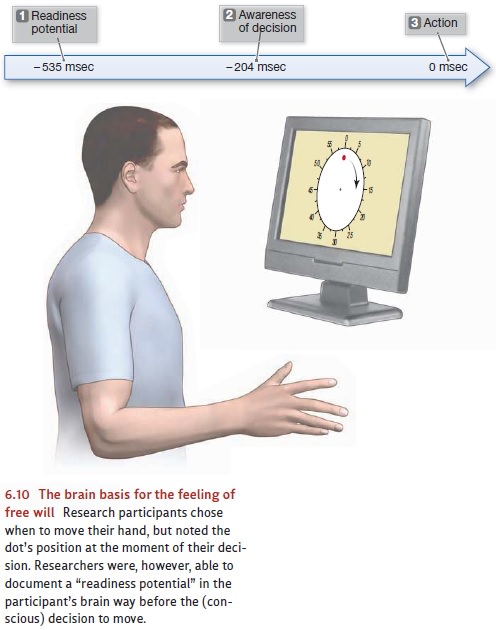
chose to
make this movement, and
later they were asked to report this position. This
response tells us in essence when the conscious decision to move actu-ally took
place, and we can compare that moment to when the movement itself occurred.
Not surprisingly, there was
a brief gap—about
200 millisec-onds—between the
moment of decision
and the actual
move- ment. It took
a fraction of
a second, it
seems, to translate
the decision into an action (Figure 6.10). The real surprise was that
recordings of brain activity showed a marked change—a so-called readiness potential—almost a half-second before participantsreported any
awareness of a decision to move. In other words, the participants’ brains had
launched the action well before
the participants themselves felt
they had initiated the action. This result seems to imply that the feeling of
“I will move my hand now” is not the cause
of brain activity, as common sense might suggest. Instead, it’s the result of brain activity— in particular,
brain activity in the pre-motor and anterior cin-gulate cortices (Lau et al.,
2004). In other words, the “decision to move” and the initiation of action
happen out-side of awareness, and the person (consciously) learns only a moment
later what they’ve just decided. (For some complica-tions and possible
challenges to this result, see Banks & Isham, 2009; Desmurget et al., 2009;
Haggard, 2009; Obhi, Planetta, & Scantlebury, 2009.)
Related Topics