Chapter: Microbiology
Innate and Adaptive Immunity
INNATE AND ADAPTIVE IMMUNITY
Introduction
Immunity is a state of protection from infectious diseases. It has two components namely nonspecific and specific immunities. The non-specific component is called innate immunity.. Innate immunity is defined as disease resistance mechanisms that are not specific to a particular pathogen. The specific component is called acquired im-munity or adaptive immunity.
Adaptive immunity shows a high degree of specificity andmemory and requires more time to develop. Innate immunity provides thefirst line of defense during the critical period just after the host’s exposure to a pathogen. A healthy individual when exposed to a vari-ety of microorganisms the innate immunity clears most of them within a few days. During this time the adaptive immune response is not trig-gered. However if the innate immunity fails and the microorganisms try to invade the tissues the specific immune response of adaptive immu-nity is triggered.
Innate and adaptive immunity do not operate independently. They function as a highly interactive and cooperative systems.
Innate immunity consists of four types of defensive barriers namely anatomic, physiologic, phagocytic and inflammatory barriers.
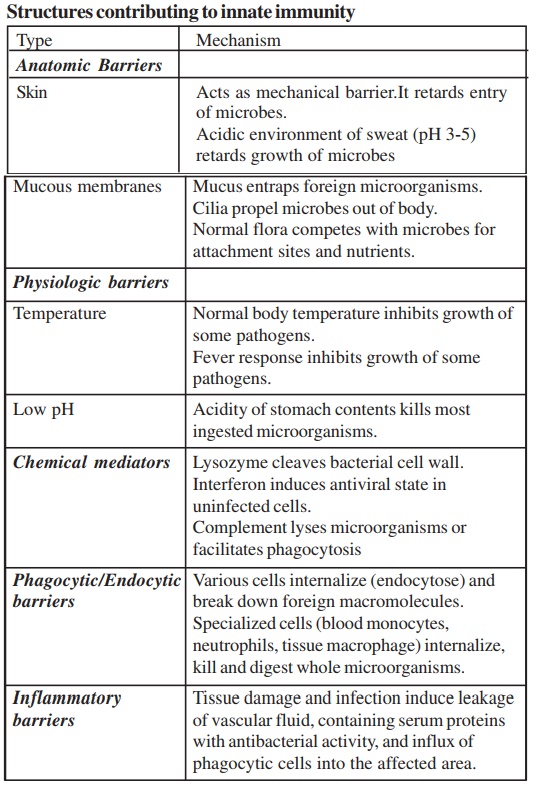
FACTORS CONTRIBUTING TO INNATE IMMUNITY:
STRUCTURE AND ROLE PLAYED
Anatomic Barriers
In the first line of defense against infection, physical and anatomic barriers try to prevent the entry of pathogens. The skin and the surface of mucous membranes effectively prevent the entry.
Skin
The skin consists of two distinct layers. A thinner outer layer is called the epidermis and a thicker inner layer is called the dermis.The epidermis contains many layers of tightly packed epithelial cells. The outermost epidermal layer consists of dead cells and is filled with a hydrophobic protein called keratin. The dermis, which is composed of connective tissue contains blood vessels, hair follicles, sebaceous glands, and sweat glands. The sebaceous glands are associated with the hair follicles and produce an oily secretion called sebum. Sebum consists of lactic acid and fatty acids. These acids maintain the pH of the skin between 3 and 5. This low pH inhibits the growth of most microorgan-isms. Intact skin prevents the entry of pathogens and also its low pH inhibits most bacterial growth. Breaks in the skin, even small ones, result in the entry of pathogens to cause infections. The skin is also penetrated by biting insects (mosquitoes, mites, ticks, fleas, flies); if these insects harbor pathogenic organisms, they can introduce the patho-gens into the body as they feed.
Mucous membranes
The conjunctiva and the alimentary, respiratory, urogenital tracts are lined by mucous membranes. These membranes consist of an outer epithelial layer and an underlying layer of connective tissue. A number of nonspecific defense mechanisms exist to prevent the entry of patho-gens. For example, saliva, tears, and mucous secretions act to wash away potential invaders. They contain antibacterial and antiviral sub-stances. The viscous fluid called mucus, which is secreted by epithelial cells entraps foreign microorganisms. In the respiratory and gastrointes-tinal tract, the mucus membrane is covered by cilia. The synchronous movement of cilia propels mucus-entrapped microorganisms towards the exterior from these tissues. In addition, nonpathogenic organisms tend to colonize epithelial cells of mucosal surfaces. These normal flora generally compete with pathogens for attachment sites on the epithelial cell surface and for necessary nutrients. Mucus also contains the en-zyme lysozyme which lyses the bacterial cell. Mucoproteins present in the mucus also inhibit the haemagglutinins of influenza virus
Many pathogens enter the body by binding to and penetrating mucous membranes, overcoming the protective effects.
Physiologic Barriers
The following act as physiologic barriers of innate immune response:
1. Antibodies against blood group antigens
2. Alternate pathway of complement system
3. Macrophages
4. Interferons
5. γδ Cells
6. CD5-B Cells
Also, the physiologic barriers that contribute to innate immunity include temperature, pH and various soluble factors. Many animal spe-cies are not susceptible to certain diseases simply because their normal body temperature inhibits the growth of the pathogens. Chicken for example have innate immunity to anthrax because their high body tem-perature inhibits the growth of the bacteria. Gastric acidity acts as physi-ologic barrier to infection because very few ingested microorganisms can survive the low pH of the stomach contents. In the mouth, strepto-cocci produce peroxides that compete with bacteria for iron and en-hance respiratory activity in neutrophils. Many soluble factors contrib-ute to nonspecific immunity such as the enzyme lysozyme, interferon, and complement.
The flushing action of urine combined with the low pH of the urinogenital tract prevents the pathogens from establishing in the uro-genital tract. Desquamation of epithelial cells from the vaginal wall in adult women provides a substrate for lactobacilli growth. These bacte-ria produce lactic acid, and also compete with pathogenic bacteria for nutrients and space.
The flushing action of milk in the mammary gland that contains lactenins, bacterial inhibitors, iron binding protein lactoferrin lactoper-oxidase, and IgA enhancers also contributes to innate immunity. Ph-agocytic cells released into the mammary gland caused in response toirritation due to sucking contributes to phagocytic action, lactoferrins and hydrogen peroxide.
Antimicrobial peptides: Cells of many animals produce anti-microbial substances that act as endogenous natural antibiotics or dis-infectants. These micropeptides take many forms.
α-Defensins:There are six known human alpha defensins. Fourbelong to neutrophils and the other two are present in vagina and cer-vix
β-Defensins:Large amounts of β-Defensins appear in Henley’sloop, distal and collecting tubules of kidney and also in the vagina, cervix, uterus and fallopian tubes. These peptides have broad spec-trum, salt-sensitive antibacterial activity and show synergy with lysozyme and lactoferrin.
Cathelicidins: Humans express only one cathelicidin, aprepropeptide that is released after neutrophil elastase action.
Protegrins: They are broad-spectrum antimicrobial peptidesfound in porcine neutrophils, where they are stored as cathelin contain-ing precursors.
Granulysin: They are found in granules of human cytolytic Tlymphocytes and natural killer cells. They act in combination with perforins, gain access to intracellular compartment of microbes and kill them.
Histatins: These are small histidine-rich human salivary proteinsthat display moderate activity against Candida albicans at acidic pH and also have antifungal actions.
Lysozyme: It is a hydrolytic enzyme found in mucus secretionsand tears, and is able to cleave the peptidoglycan layer of the bacterial cell wall.
Interferon: comprises a group of proteins produced by virus-infected cells. One of the many functions of the interferons is the ability to bind to nearby cells. It also induces a generalized antiviral state. There are three different types of interferons IFN-α, IFN-β and IFN-γ. They are synthesized by leucocytes on exposure to viruses, fibro-blasts and effector T cells on induction respectively. The second effect of interferons in host defense is to increase expression of the MHC class I complex and TAP transporter proteins, enhancing the ability of virus-infected cells to present viral peptides to CD-8 cells. The third property is the activation of natural killer cells.
Complement is a group of serum proteins that circulate in aninactive state. A variety of specific and nonspecific immunologic mecha-nisms can convert the inactive forms of complement proteins into ac-tive form. When activated complement can cause damage to the mem-branes of pathogenic organisms, so that they are either destroyed or phagocytosed and cleared
Phagocytic Barriers
Another important innate defense mechanism is the ingestion of extracellular particulate material by phagocytosis. Phagocytosis is a phenomenon in which there is uptake of material by a cell from its environment. In phagocytosis, a cell’s plasma membrane expands around the particulate material to form large vesicles called phagosomes. Most phagocytosis is conducted by specialized cells, such as blood monocytes, neutrophils, and tissue macrophages. Phagocytosis may be enhanced by a variety of factors collectively termed asopsonins which consist of antibodies and various serum components of comple-ment.
Polymorphonuclear (PMN) leucocytes also referred to asgranulocytes include basophils, mast cells, eosinophils and neutrophils. These short-lived phagocytic cells contain lysosomes filled with hydro-lytic enzymes. They play a major role in protection against infection.
Macrophages: These cells enter the blood as monocytes andmigrate to different tissues In these tissues they undergo different changes. The monocyte is a small, spherical cell with few projections, abundant cytoplasm, little endoplasmic reticulum, and many granules. Macroph-ages of different tissues have different names. In liver they are called Kupffer cells, in lungs - alveolar macrophages, in spleen - splenic mac-rophages, in brain – microglial cells etc. Macrophages have the follow-ing functions: 1.They phagocytose particles from the environment,(2) process antigens and present to T cells,thus function as antigen-pre-senting cells.
Certain cytotoxic or killer cells destroy the target cell not by phagocytosis but by releasing biologically potent molecules. Such killer cells include the cytotoxic T lymphocytes and Natural killer cells (NK Cells).
Inflammatory Barriers
Tissue damage caused by a wound or by an invading pathogenic microorganism induces a complex sequence of events collectively called as the inflammatory response. As early as the first century AD, the Roman physician Celsus described the “five cardinal signs of infection” as rubor (redness), tumor (swelling), calor (heat), dolor (pain) and functio laesa (loss of function). The cardinal signs of inflammation reflect the major events of an inflammatory response.
1. Vasodilation: An increase in the diameter of blood vessels of nearby capillaries occurs. The vessels that carry blood away from the af-fected area constrict. This results in engorgement of the capillary network. The engorged capillaries are responsible for tissue red-ness (erythema) and in increase in tissue temperature.
2. An increase in capillary permeability facilitates an influx of fluid and cells from the engorged capillaries into the tissue. The fluid that accumulates (exudates) has a much higher protein content. Accu-mulation of fluid contributes to tissue swelling (edema).
3. Influx of phagocytes from the capillaries into the tissue is facilitated by the increased capillary permeability. As phagocytic cells accu-mulate at the site and begin to phagocytose bacteria, they releaselytic enzymes, which can damage nearby healthy cells. The accu-mulation of dead cells, digested material, and fluid forms a sub-stance called pus.
The events in the inflammatory response are initiated by a com-plex series of events. During the inflammatory response, many chemi-cal mediators are released, in response to tissue damage. They are called acute-phase proteins. The concentrations of these proteins increase dramatically in tissue-damaging infections. C-reactive protein is a major acute-phase protein produced by the liver. Another media-tor of the inflammatory response is histamine, a chemical released by a variety of cells in response to tissue damage. Histamine causes va-sodilation and increased permeability. Another important group of in- flammatory mediators are called They are normally present in blood plasma in an inactive form. Tissue injury activates these pep-tides, which then cause vasodilation and increased permeability. A par-ticular kinin, called bradykinin, also stimulates pain receptors in the skin. Vasodilation and the increase in capillary permeability in an in-jured tissue also enable enzymes of the blood-clotting system to enter the tissue. These enzymes activate an enzyme cascade that results in the deposition of insoluble strands of fibrin.. The fibrin strands wall off the injured area from the rest of the body and serve to prevent the spread of infection.
Once the inflammatory response has subsided and most of the debris has been cleared away by phagocytic cells, tissue repair and regeneration of new tissue begins. Capillaries grow into the fibrin of a blood clot. New connective tissue cells, called fibroblasts and capillar-ies accumulate, and scar tissue forms.
Collaboration between Innate and Adaptive Immunity
Innate and Adaptive immunity do not operate in total indepen-dence of each other. They cooperate in important ways to produce more effective immunity. For example, the encounter between mac-rophages and microbes can generate antigen presenting cells that stimu-late and direct adaptive immune responses. This facilitates the partici-pation of the adaptive immune system in the elimination of the patho-gen. Macrophages also secrete immunoregulatory hormone-like mol-ecules, called cytokines. The cytokines and other signals generated by innate immunity play important roles in triggering lymphocyte responses.
The adaptive immune system produces signals and components that stimulate and increase the effectiveness of innate responses. There is increase in the ability of macrophages to kill the microbes they have ingested. The production of antibodies against an invading pathogen also has important effects on the recruitment of the complement system to the defense of the host. By binding to the pathogen, antibodies mark it as a target for attack by complement, and the complex of antibody and pathogen is also a potent activator of this attack. Thus, these two systems, nonspecific and specific immunity, form an interactive and mutually supportive network that erects an effective and formidable barrier to infection.
Related Topics