Chapter: Essential Microbiology: Microbiology: What, Why and How?
Electron microscopy
Electron
microscopy
From the equation shown , you can see that if it
were possible to use a shorter wave-length of light, we could improve the resolving
power of a microscope. However, because we are limited by the wavelength of
light visible to the human eye, we are not able to do this with the light
microscope. The electron microscope is able to achieve greater magnification
and resolution because it uses a high voltage beam of electrons, whose
wavelength is very much shorter than that of visible light. Consequently we are
able to resolve points that are much closer together than is possible even with
the very best light microscope. The resolving power of an electron microscope
may be as low as 1ŌĆō2 nm, enabling us to see viruses, for example, and the
internal structure of cells. The greatly im-proved resolution means that
specimens can be meaningfully magnified over 100 000├Ś.
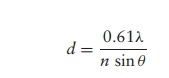
where ╬╗ is the wavelength of the light source, n is the refractive index of the air or liquid between the objec-tive lens and the specimen and ╬Ė is the aperture angle (a measure of the light-gathering ability of the lens).
The expression n sin╬Ė is called the numerical aperture and for high quality lenses has a value of around 1.4. The lowest wavelength of light visible to the human eye is approximately 400 nm, so the maximum resolving power for a light microscope is approximately
Electron microscopes, which were first developed in
the 1930s and 1940s, use ring-shaped electromagnets as ŌĆślensesŌĆÖ to focus the
beam of electrons onto the specimen. Because the electrons would collide with,
and be deflected by, molecules in the air, electron microscopes require a pump
to maintain a vacuum in the column of the instru-ment. There are two principal
types of electron microscope, the transmission electron microscope (TEM) and
the scanning electron microscope (SEM).
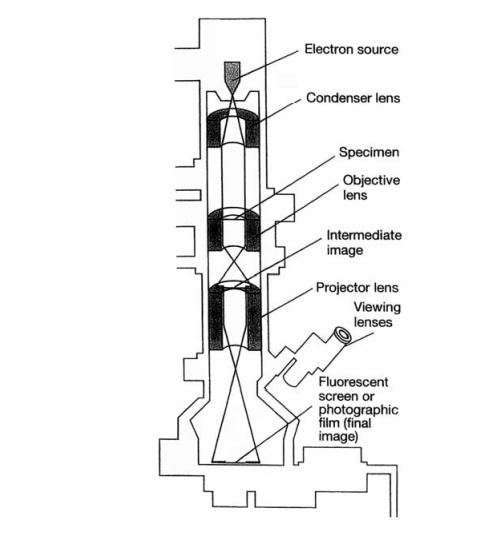
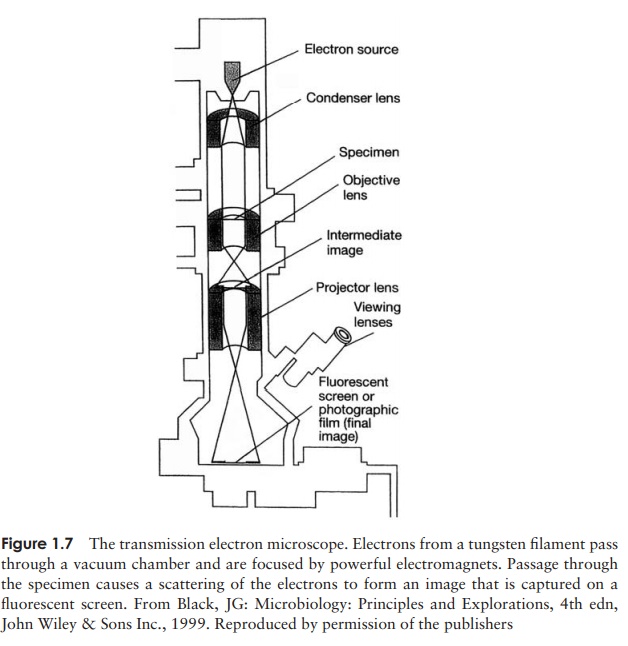
Figure 1.7 shows the main features of a TEM. As the
name suggests, the electron beam passes through
the specimen and is scattered according to the density of the different parts.
Due to the limited penetrating power of the electrons, extremely thin sections
(<100 nm, or less
than one-tenth of the diameter of a bacterial cell) must be cut, using a
diamond knife. To allow this, the specimen must be fixed and dehydrated, a
process that can introduce shrinkage and distortion to its structure if not
correctly performed.
After being magnified by an objective ŌĆślensŌĆÖ, an
image of the specimen is projected onto a fluorescent screen or photographic
plate. More dense areas, which scatter the beam, appear dark, and those where
it has passed through are light. It is often necessary to enhance contrast
artificially, by means of ŌĆśstainingŌĆÖ techniques that involve coating the
specimen with a thin layer of a compound containing a heavy metal, such as
osmium or palladium. It will be evident from the foregoing description of
sample preparation and use of a vacuum that electron microscopy cannot be used
to study living specimens.
The TEM has been invaluable in advancing our
knowledge of the fine structure of cells, microbial or otherwise. The resulting
image is, however, a flat, two-dimensional one, and of limited use if we wish
to learn about the surface of a cell or a virus. For this, we turn to SEM. The
scanning electron microscope was developed in the 1960s and provides vivid,
sometimes startling, three-dimensional images of surface structure. Samples are
dehydrated and coated with gold to give a layer a few nanometres thick. A fine
beam of electrons probes back and forth across the surface of the specimen and
causes secondary electrons to be given off. The number of these, and the angle
at which they are emitted, depends on the topography of the specimenŌĆÖs surface.
SEM does not have quite the resolving power of the TEM, and therefore does not
operate at such high magnifications. Between them, SEM and TEM have opened up a
whole new world to microbiologists, allowing us to put advances in our
knowledge of microbial biochemistry and genetics into a structural context.
Related Topics