Chapter: 11th 12th std standard Class Organic Inorganic Physical Chemistry Higher secondary school College Notes
Electro Chemistry - Cell Terminology
Electro Chemistry - Cell
Terminology
Before taking up the study of
the electrochemical cells, we should be familiar with a few common terms.
Current is the flow of electrons through a wire or any
conductor.
Electrode
is the material : a metallic rod/bar/strip
which conducts electrons into and
out of a solution.
Anode is the
electrode at which oxidation occurs. It sends electrons into the outer circuit. It has negative charge and is shown as (-)
in cell diagrams.
Cathode is the electrode at which electrons are
received from the outer circuit. It
has a positive charge and is known as (+) in the cell diagrams.
Electrolyte
is the salt solution in a cell.
Anode
compartment is the compartment of the
cell in which oxidation half-reaction
occurs. It contains the anode.
Cathode
compartment is the compartment of the
cell in which reduction half-reaction
occurs. It contains the cathode.
Half-cell.
Each half of an electrochemical cell, where
oxidation occurs and the half where
reduction occurs, is called the half cell.
Cell diagram or
Representation of a Cell
A cell diagram is an abbreviated symbolic
depiction of an electrochemical cell. For this purpose, we will consider that a
cell consists of two half-cells. Each half-cell is again made of a metal
electrode in contact with metal ion in solution.
IUPAC
Conventions. In 1953 IUPAC recommended the
following conventions for writing
cell diagrams. We will illustrate these with reference to Zinc-Copper cell.
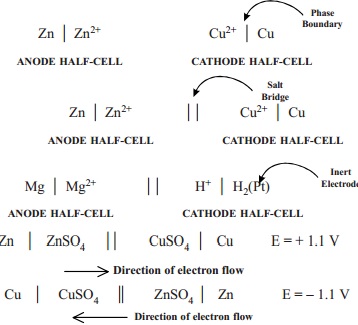
(1) a single vertical line (|) represents a
phase boundary between metal electrode and ion solution (electrolyte). Thus the
two half-cells in a voltaic cell are indicated as
Zn | Zn2+ Cu2+ | Cu
ANODE HALF-CELL CATHODE HALF-CELL
It may be noted that the metal electrode in
anode half-cell is on the left, while in cathode half-cell it is on the right
of the metal ion.
A double vertical line
represents the salt bridge, porous partition or any other means of permitting
ion flow while preventing the electrolyte from mixing.
Anode half-cell is written on
the left and cathode half-cell on the
right.
In the complete cell diagram,
the two half-cells are separated by a double vertical line (salt bridge) in
between. The zinc-copper cell can now be written as
Zn | Zn2+ | | Cu2+ | Cu
ANODE HALF-CELL CATHODE HALF-CELL
(5) The symbol for an inert
electrode, like the platinum electrode is often enclosed in a bracket. For example,
Mg | Mg2+
| | H+ | H2
(Pt)
ANODE HALF-CELL CATHODE HALF-CELL
(6) The value of emf of a
cell is written on the right of the cell diagram. Thus a zinc-copper cell has
emf 1.1 V and is represented as
Zn | ZnSO4 | | CuSO4 | Cu E = + 1.1 V
--- -- > Direction of electron flow
If the emf acts in the
opposite direction through the cell circuit it is denoted as a negative value.
Cu | CuSO4 || ZnSO4 | Zn E = - 1.1 V
< -- -- -- Direction of electron flow
The negative sign also indicates that the cell
is not feasible in the given direction and the reaction will take place in the
reverse direction only. The overall cell reaction for E = -1.1 V of the daniel
cell is
Cu(s) + Zn(aq)2+ -- -- -- - > Cu(aq)2+
+ Zn(s)
The reversal of the cell current is accompanied
by the reversal of direction of the cell reaction. Thus a reversible cell is
defined as that operates by reversal of cell reactions when the direction of
flow of current is reversed. The reversible behaviour operates only for
infinitesimal changes in e.m.f. of the cell on the positive and negative sides
from the balance point which corresponds to the actual emf of the cell.
Related Topics