Chapter: 11th 12th std standard Class Organic Inorganic Physical Chemistry Higher secondary school College Notes
Metallurgical processes
Metallurgical processes
Metallurgy is a branch of chemistry which deals with,
1.
Extraction of metals from ores
2. Refining of crude metal
3. Producing alloys and the study of their constitution, structure and properties.
4.
The relationship of physical and mechanical treatment
of metals to alloys.
The extraction of metals cannot be carried out by any universal method
because extraction of each metal requires different methods of extraction. This
depends upon the nature and preparation of metals. In general, noble metals such
as Au, Ag, etc are usually extracted by electrolysis of their chlorides, oxides
or hydroxides. Heavy metals, e.g. Cu, Zn, Fe, Pb, Sn, etc., are extracted by
making use of roasting and smelting methods.
1.Roasting- oxidation
Roasting is one of the oxidation method where ore is converted into
metal oxide. In the process of roasting, the ore either alone or with the
addition of suitable material, is subjected to the action of heat in excess of
air at temperature below its melting point. Roasting is usually carried out in
a reverberatory furnace or in a blast furnace. During roasting
(a)
Volatile impurities like S, As, Sb etc. get oxidized and escape out as volatile
gases SO2, As2O3 and Sb2O3
(b) The sulphide ores decompose to their oxides evolving SO2 (c) The
moisture is removed. Mass becomes porous and thus it can easily be reduced.
Roasting may be of many types.
Oxidising Roasting - In this type of roasting S, As, and Sb impurities are removed in the form of their volatile oxides as SO2,
As2O3 and Sb2O3 etc. due to
combined action of heat and air. The ore is simultaneously converted into its
oxides. This type of roasting is carried out for ores like copper pyrites, zinc
blende and lead ores (PbS) etc.
2ZnS + 3O2 � 2ZnO + 2SO2
2PbS + 3O2 � 2PbO + 2SO2
Calcination
Another method of conversion of ore into metal oxide (oxidation) is
called calcination. It is the process in which the ore is subjected to the
action of heat at high temperature in the absence of air below its melting
point. The process of calcination is carried out in the case of carbonate and
hydrated ore. As a result of calcination (a) The moisture is removed.
(b) Gases
may be expelled. (c) Volatile impurities are removed. (d) The mass becomes
porous. (e) Thermal decomposition of the ore takes place. For example,
CaCO3 (limestone) � CaO + CO2 - MgCO3 (Magnesite) � MgO + CO2 -
MgCO3.CaCO3 (Dolomite) � MgO + CaO + 2CO2 - CuCO3.Cu(OH)2 (Malachite)
� 2CuO + H2O
+ CO2 - ZnCO3
(Calamine) � ZnO + CO2-
2Fe2O3.3H2O (Limonite) � 2Fe2O3 + 3H2O -
The name calcination originated from the ore calcite which on thermal
decomposition gives quick lime. Calcination is usually carried out in
reverberatory furnace.
2.Smelting - Reduction
Smelting
is one of reduction method where the metal oxide is converted into metal. It is
the process used for all operations where the metal is separated by fusion from
the ore. The process of smelting is that in which ore is melted with a flux and
often with a reducing agent, and it involves, calcination, roasting and
reduction. In general, the process of separation of a metal or its sulphide
mixture from its ore in fused state is called smelting. Smelting is generally
carried out in a blast furnace and high temperature is produced by burning coal
or by using electric energy.
In smelting, the roasted or calcined ore is mixed with coke and then
heated in a furnace. As a result, (carbon and CO produced by the incomplete
combustion) carbon reduces the oxide to the metal. For example, in the
extraction of iron, haematite ore(Fe2O3) is smelted with
coke and limestone (flux). As a result of reduction, iron is obtained in fused
or molten state.
Fe2O3 + 3C - > 2Fe + 3CO
CaCO3 - > CaO + CO2 Fe2O3 + 3CO - > 2Fe + 3CO2 CaO + SiO2 - > CaSiO3
Flux + Gangue - > Slag
Similarly,
in the extraction of copper from copper
pyrites, the ore is mixed with coke and heated in blast furnace (smelted).
Infusible impurity FeO is converted to FeSiO3(slag) and is removed.
A mixture containing sulphide of copper and iron, called matte is formed in the molten state.
FeO (Gangue) SiO2 (Flux) - > - > FeSiO3
(Slag)
Other Examples
ZnO + C - > Zn + CO
SnO2 + 2C - >Sn + 2CO
MnO2 + 2C - > Mn + 2CO
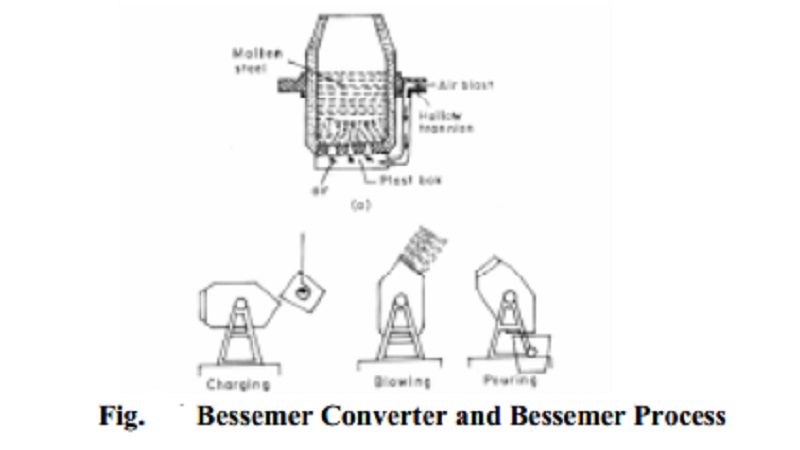
3 Bessemerisation
It is the process used for the manufacture of
steel from pig iron. Steel is an alloy of carbon and iron and contains
0.15-1.5% of carbon with traces of sulphur, phosphorus, manganese and silicon
as impurities. Depending upon the carbon content, steel are classified into
three classes namely mild carbon steel (0.15-0.3%), medium carbon steel
(0.3-0.8%) and high carbon steel (0.8-1.50).
The
process was discovered by Henry Bessemer in England (1856). The principle
involved in this process is that cold air blowed through refractory lined
vessel known as converter containing molten pig iron at about 2 atmospheric
pressure, oxidizing the impurities and simultaneously converting pig iron to
steel.
This process mainly differs in the use of acidic
and basic refractory linings of the converters. In this process low phosphorus
pig iron (below 0.09%) is treated by acidic Bessemer process and high
phosphorus pig iron (more than 1.5%) is treated in basic.
The converter is a pear shaped furnace about 6m high and 3m in diameter.
It is made of steel plates and is lined inside with silica or magnesia (MgO),
depending upon the nature of impurities present in the pig iron. If the
impurities present in the pig iron are basic, e.g., manganese, a lining of
silica brick is used and the process is known as acid Bessemer process. If impurities are acidic, e.g., sulphur, phosphorus etc., a basic lining of
lime (CaO) or magnesia (MgO) is used in the converter and process is then known
as basic Bessemer process. The
capacity of the converter is from 10-25 tonnes of charge at a time. The
converter is mounted on shafts or trunnions, one of which is hollow and serves
as a wind pipe and upon which the converter can rotate in any position. The
converter is also provided with a number of holes at the bottom through which a
hot blast of air can be introduced.
The molten
pig iron is mixed in mixers and then charged into converter. About 15-16 tonnes
of iron can be charged at a time. The converter is first set in the horizontal
position and after charging the converter is adjusted in vertical position.
After charging a blast of cold air is admitted through the hole provided at the
bottom at a pressure of about 2-3 kg/cm3. The blast is continued for
about 15 minutes during which the impurities are oxidized. Mn is oxidized to
MnO and Si is oxidized to SiO2. Carbon is also oxidized to CO. The
resulting oxides of Mn and Si (MnO and SiO2) combine together to
form slag of manganese silicate Fig..
Related Topics