Chapter: Basic & Clinical Pharmacology : The Gonadal Hormones & Inhibitors
Disturbances in Ovarian Function
THE OVARY ESTROGENS, PROGESTINS, OTHER OVARIAN HORMONES, ORAL
CONTRACEPTIVES, INHIBITORS & ANTAGONISTS, & OVULATION INDUCING AGENTS
The ovary has
important gametogenic functions that are inte-grated with its hormonal
activity. In the human female, the gonad is relatively quiescent during
childhood, the period of rapid growth and maturation. At puberty, the ovary
begins a 30- to 40-year period of cyclic function called the menstrual cycle because of the regular
episodes of bleeding that are its most obvi-ous manifestation. It then fails to
respond to gonadotropins secreted by the anterior pituitary gland, and the
cessation of cyclic bleeding that occurs is called menopause.
The mechanism
responsible for the onset of ovarian function at the time of puberty is thought
to be neural in origin, because the immature gonad can be stimulated by
gonadotropins already present in the pituitary and because the pituitary is
responsive toexogenous hypothalamic gonadotropin-releasing hormone. The
maturation of centers in the brain may withdraw a childhood-related inhibitory
effect upon hypothalamic arcuate nucleus neurons, allowing them to produce gonadotropin-releasing hormone(GnRH) in
pulses with the appropriate amplitude, which stimu-lates the release of follicle-stimulating hormone (FSH) and luteinizing hormone (LH) . At first,
smallamounts of the latter two hormones are released during the night, and the
limited quantities of ovarian estrogen secreted in response start to cause
breast development. Subsequently, FSH and LH are secreted throughout the day
and night, causing secretion of higher amounts of estrogen and leading to
further breast enlargement, alterations in fat distribution, and a growth spurt
that culminates in epiphysial closure in the long bones. The change of ovarian
function at puberty is called gonadarche.
A year or so after
gonadarche, sufficient estrogen is produced to induce endometrial changes and
periodic bleeding. After the first few irregular cycles, which may be
anovulatory, normal cyclic function is established.
At the beginning of
each cycle, a variable number of follicles (vesicular follicles), each
containing an ovum, begin to enlarge in response to FSH. After 5 or 6 days, one
follicle, called the dominant
ACRONYMS
CBG Corticosteroid-binding
globulin (transcortin)
DHEA Dehydroepiandrosterone
DHEAS Dehydroepiandrosterone
sulfate
ERE Estrogen
response element
FSH Follicle-stimulating
hormone
GnRH Gonadotropin-releasing
hormone
HDL High-density
lipoprotein
HRT Hormone
replacement therapy (also called HT)
LDL Low-density
lipoprotein
LH Luteinizing
hormone
PRE Progesterone
response element
SERM Selective
estrogen receptor modulator
SHBG Sex
hormone-binding globulin
TBG Thyroxine-binding
globulin
follicle, begins to
develop more rapidly. The outer theca and inner granulosa cells of this
follicle multiply and, under the influence of LH, synthesize and release
estrogens at an increasing rate. The estrogens appear to inhibit FSH release
and may lead to regression of the smaller, less mature follicles. The mature
dominant ovarian follicle consists of an ovum surrounded by a fluid-filled
antrum lined by granulosa and theca cells. The estrogen secretion reaches a
peak just before midcycle, and the granulosa cells begin to secrete
progesterone. These changes stimulate the brief surge in LH and FSH release
that precedes and causes ovulation. When the follicle ruptures, the ovum is
released into the abdominal cavity near the opening of the uterine tube.
Following the above
events, the cavity of the ruptured follicle fills with blood (corpus
hemorrhagicum), and the luteinized theca and granulosa cells proliferate and
replace the blood to form the corpus luteum. The cells of this structure
produce estrogens and progester-one for the remainder of the cycle, or longer
if pregnancy occurs.
If pregnancy does not
occur, the corpus luteum begins to degenerate and ceases hormone production,
eventually becoming a corpus albicans. The endometrium, which proliferated
dur-ing the follicular phase and developed its glandular function during the
luteal phase, is shed in the process of menstruation. These events are
summarized in Figure 40–1.
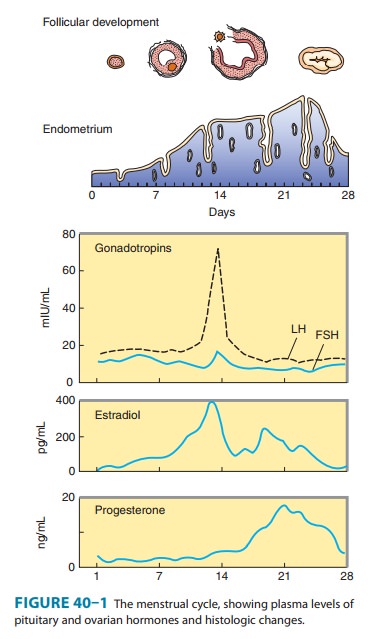
The ovary normally
ceases its gametogenic and endocrine func-tion with time. This change is
accompanied by a cessation in uter-ine bleeding (menopause) and occurs at a
mean age of 52 years in the USA. Although the ovary ceases to secrete estrogen,
significant levels of estrogen persist in many women as a result of conversion
of adrenal and ovarian steroids such as androstenedione to estrone and
estradiol in adipose and possibly other nonendocrine tissues.
Disturbances in Ovarian Function
Disturbances of cyclic
function are common even during the peak years of reproduction. A minority of
these result from inflammatory or neoplastic processes that influence the
functions of the uterus,ovaries, or pituitary. Many of the minor disturbances
leading to periods of amenorrhea or anovulatory cycles are self-limited. They
are often associated with emotional or physical stress and reflect temporary
alterations in the stress centers in the brain that control the secretion of
GnRH. Anovulatory cycles are also associated with eating disorders (bulimia,
anorexia nervosa) and with severe exercise such as distance running and
swimming. Among the more com-mon organic causes of persistent ovulatory
disturbances are pituitary prolactinomas and syndromes and tumors characterized
by excessive ovarian or adrenal androgen production. Normal ovarian function
can be modified by androgens produced by the adrenal cortex or tumors arising
from it. The ovary also gives rise to androgen-produc-ing neoplasms such as
arrhenoblastomas, as well as to estrogen-pro-ducing granulosa cell tumors.
Related Topics