Chapter: Basic & Clinical Pharmacology : The Gonadal Hormones & Inhibitors
Ovulation-Inducing Agents
OVULATION-INDUCING AGENTS
CLOMIPHENE
Clomiphene citrate, a
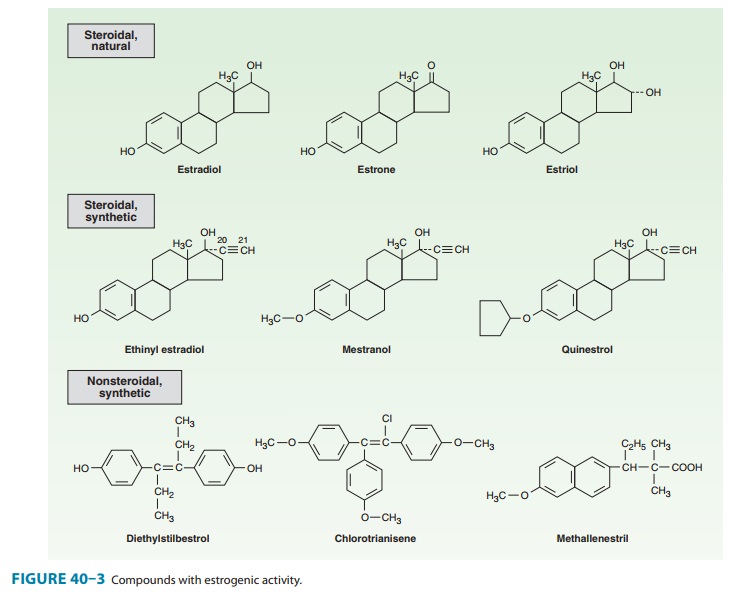
partial estrogen agonist, is closely related to the estrogen chlorotrianisene
(Figure 40–3). This compound is well absorbed when taken orally. It has a
half-life of 5–7 days and is excreted primarily in the urine. It exhibits
significant protein binding and enterohepatic circulation and is distributed to
adipose tissues.
Pharmacologic Effects
A. Mechanisms of Action
Clomiphene is a
partial agonist at estrogen receptors. The estro-genic agonist effects are best
demonstrated in animals with marked gonadal deficiency. Clomiphene has also
been shown to effectively inhibit the action of stronger estrogens. In humans
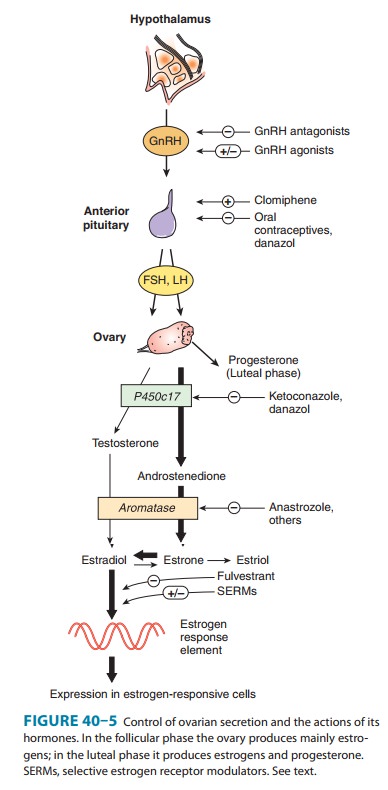
it leads to an increase in the secretion of gonadotropins and estrogens by
inhib-iting estradiol’s negative feedback effect on the gonadotropins (Figure
40–5).
B. Effects
The
pharmacologic importance of this compound rests on its abil-ity to stimulate
ovulation in women with oligomenorrhea or amenorrhea and ovulatory dysfunction.
The majority of patients suffer from polycystic ovary syndrome, a common
disorder affect-ing about 7% of women of reproductive age. The syndrome is
characterized by gonadotropin-dependent ovarian hyperandro-genism associated
with anovulation and infertility. The disorder is frequently accompanied by
adrenal hyperandrogenism. Clomiphene probably blocks the feedback inhibitory
influence of estrogens on the hypothalamus, causing a surge of gonadotropins,
which leads to ovulation.
Clinical Use
Clomiphene is used in
the treatment of disorders of ovulation in patients who wish to become
pregnant. Usually, a single ovulation is induced by a single course of therapy,
and the patient must be treated repeatedly until pregnancy is achieved, since
normal ovula-tory function does not usually resume. The compound is of no value
in patients with ovarian or pituitary failure.
When
clomiphene is administered in a dosage of 100 mg/d for 5 days, a rise in plasma
LH and FSH is observed after several days. In patients who ovulate, the initial
rise is followed by a second rise of gonadotropin levels just prior to
ovulation.
Adverse Effects
The
most common adverse effects in patients treated with this drug are hot flushes,
which resemble those experienced by menopausal patients. They tend to be mild,
and disappear when the drug is discontinued. There have been occasional reports
of eye symptoms due to intensification and prolongation of afterim-ages. These
are generally of short duration. Headache, constipation, allergic skin
reactions, and reversible hair loss have been reported occasionally.
The effective use of
clomiphene is associated with some stimu-lation of the ovaries and usually with
ovarian enlargement. The degree of enlargement tends to be greater and its
incidence higher in patients who have enlarged ovaries at the beginning of
therapy.
A
variety of other symptoms such as nausea and vomiting, increased nervous
tension, depression, fatigue, breast soreness, weight gain, urinary frequency,
and heavy menses have also been reported. However, these appear to result from
the hormonal changes associated with an ovulatory menstrual cycle rather than
from the medication. The incidence of multiple pregnancy is approximately 10%.
Clomiphene has not been shown to have an adverse effect when inadvertently
given to women who are already pregnant.
Contraindications & Cautions
Special
precautions should be observed in patients with enlarged ovaries. These women
are thought to be more sensitive to this drug and should receive small doses.
Any patient who complains of abdominal symptoms should be examined carefully.
Maximum ovarian enlargement occurs after the 5-day course has been com-pleted,
and many patients can be shown to have a palpable increase in ovarian size by
the seventh to tenth days. Treatment with clomiphene for more than a year may
be associated with an increased risk of low-grade ovarian cancer; however, the
evidence for this effect is not conclusive.
Special precautions
must also be taken in patients who have visual symptoms associated with
clomiphene therapy, since these symptoms may make activities such as driving
more hazardous.
OTHER DRUGS USED IN OVULATORY DISORDERS
In addition to clomiphene, a variety of other hormonal and non-hormonal agents are used in treating anovulatory disorders.
Related Topics