Chapter: The Diversity of Fishes: Biology, Evolution, and Ecology: A history of fishes
Trends during teleostean phylogeny
Trends during teleostean phylogeny
Although numerous derived traits characterize teleostean groups (Fig. 11.23), trends in five areas can be readily linked to functional improvements that contributed to teleostean success. These trends include reduction in bony elements, repositioning and elaboration of the dorsal fin, change in placement and function of paired fins, structural modifications to and interaction between the caudal fin and gas bladder, and jaw improvements.
Reduction of bony elements Teleosts show a general reduction in bony elements as compared to pre-teleostean groups (see Nelson 1994, 2006). This reduction occurred through fusion or actual loss of bones. For example, higher teleosts have the following features.
1 There are fewer, more ossified vertebrae (in general 60–80 in many elopomorphs and clupeomorphs, 30– 40 in ostariophysans, 30–70 in protacanthopterygians, 20–35 in paracanthopterygians, and 20–30 in most percomorphs). A shorter, more ossified axial skeleton would allow for attachment of stronger trunk musculature, thus enhancing locomotion.
2 There are fewer vertebral accessories, such as intermuscular bones and ribs, and the replacement of numerous small intermuscular bones with fewer, thicker zygopophyses (compare the “boniness” of fillets from a herring or trout with that from a tuna or flatfish).
3 There are fewer bones in the skull (e.g., the orbitosphenoid is missing in perciforms; there are 10–20 branchiostegals in osteoglossomorphs, elopomorphs, and clupeomorphs, 5–20 in ostariophysans and protacanthopterygians, and 4–8 in paracanthopterygians and acanthopterygians).
4 There is a reorganization and reduction in the number of bones of the tail, including fusion of the supporting
Table 11.1
Repeated trends in fish evolution. Although fishes represent diverse and heterogeneous assemblages assigned to at least five different classes, certain repeated trends have characterized the evolution of these groups or of major, successful taxa within them. The following list summarizes traits or characteristics common to the evolution of several groups.

5 There is a reduction of the number of biting bones in the upper jaw from two to one. The maxilla becomes excluded from the gapein paracanthopterygians and acanthopterygians. In more primitive groups, it is a tooth-bearing bone, whereas in the two spiny superorders, it pivots with the elongate premaxilla to create a tubular mouth (see below).
6 There is a reduction in the number of fin rays in paired fins (six or more soft pelvic rays in most lower teleosts, six or fewer in most paracanthopterygians, and one spine with five or fewer rays in most acanthopterygians).
7 There is a reduction in the amount of bone in the scales (compare the heavy cycloid scale of a tarpon, Megalopidae, or arapaima, Osteoglossidae, with the thin ctenoid scales of most paracanthopterygians and acanthopterygians). A trend toward reduction in armor is familiar by now, as it also occurred during the evolution of several groups (Table 11.1). One possible
interpretation is that mechanical protection against predators was of paramount importance when several of these taxa arose, but a premium on mobility soon developed because lighter, quicker fishes with improvements in both predator avoidance and food getting were favored.
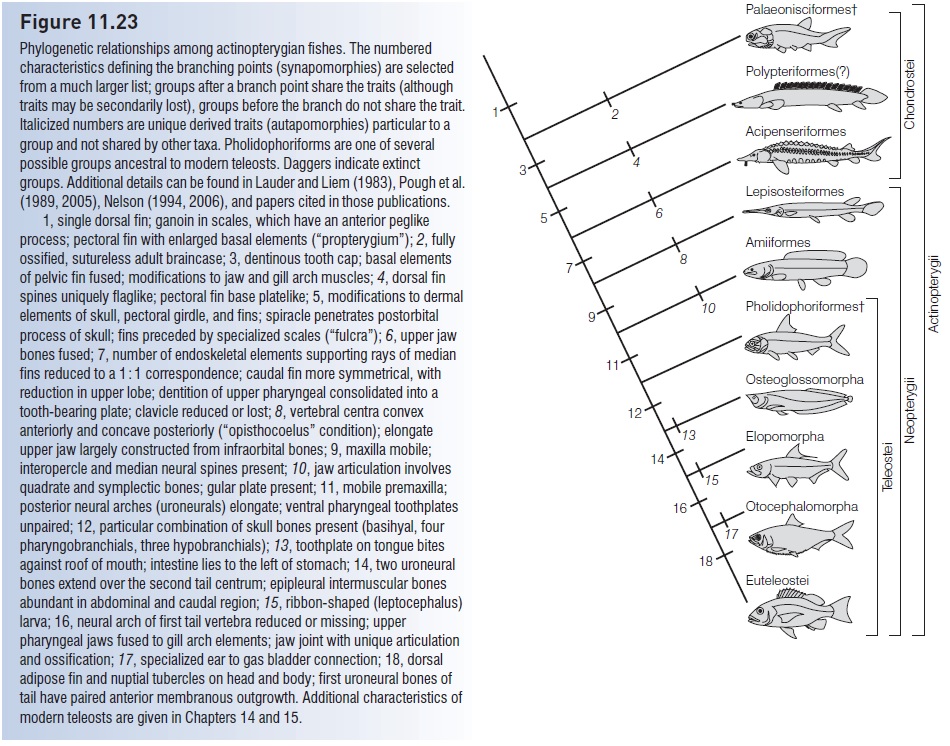
Shifts in position and use of the dorsal fin The dorsal fin in primitive teleosts is a simple, spineless, fixed, single, midbody keel that prevents rolling and serves as a pivot point for fishes that typically swim in open water situations (e.g., Mooneye, tarpon, bonefish, herrings, minnows, trouts) (Fig. 11.24). In higher teleosts, the trend is for the dorsal fin to become elongate and diversified. This is usually manifested as two fins, the anterior portion spinous and the posterior portion soft-rayed. Diversifi cation of a fin into an anterior, hardened spinous portion and posterior, fl exible portion maintains the protective function of the fin without sacrifi cing its role in maneuverability. Stability is still provided when the fin is erect, but many other functions can be served. The erected spiny dorsal provides protection from predators by increasing the body dimensions of the fish; folding the spinous dorsal against the body enhances streamlining. Rapid raising and lowering of the dorsal serves as a social signal in many fishes (similar diversifi cation and actions in the anal fin serve the same purposes).
The soft dorsal, through its fl exibility, can function as a rudder when slightly curved and as a brake when greatly curved. It can also provide mobility if sinusoidal waves are passed down its length (various knifefishes) or if it is fl apped in conjunction with the anal fin (triggerfishes, ocean sunfish) (see Locomotory types).
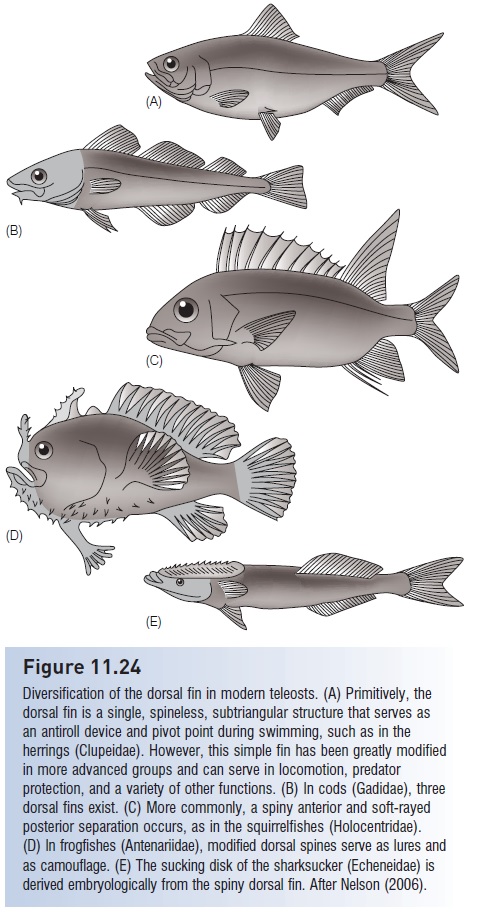
Figure 11.24
Diversification of the dorsal fin in modern teleosts. (A) Primitively, the dorsal fin is a single, spineless, subtriangular structure that serves as an antiroll device and pivot point during swimming, such as in the herrings (Clupeidae). However, this simple fin has been greatly modified in more advanced groups and can serve in locomotion, predator protection, and a variety of other functions. (B) In cods (Gadidae), three dorsal fins exist. (C) More commonly, a spiny anterior and soft-rayed posterior separation occurs, as in the squirrelfishes (Holocentridae). (D) In frogfishes (Antenariidae), modified dorsal spines serve as lures and as camouflage. (E) The sucking disk of the sharksucker (Echeneidae) is derived embryologically from the spiny dorsal fin. After Nelson (2006).
Truly bizarre modifications of the dorsal fin are seen in many higher teleosts. In paracanthopterygian anglerfishes, the first spiny ray is modified into an elongate, ornamented lure to attract prey, whereas filaments and fleshy growths increase the resemblance that some fishes show to seaweed or other structures (e.g., Sargassumfish, Antennariidae). Scorpionfishes (Acanthopterygii) use the spiny dorsal as a venom delivery system for protection against predators. Long, trailing filaments of probable social function (mate attraction, school maintenance) characterize many acanthopterygian fishes (e.g., carangids, angelfishes, cichlids). The familiar suction disk of the sharksucker, another acanthopterygian, is derived from the first dorsal fin.
Placement and function of paired fins In basal teleosts, pectoral fins are oriented horizontally and located in thethoracic position, below the edge of the gill cover; pelvic fins occur at mid-body in an abdominal location (Fig. 11.25). In this confi guration, both fins act primarily as planes that help stabilize movement up and down (pitch) or from side to side (roll), as well as providing some braking force. During teleostean phylogeny, pectoral fins move up onto the sides of the body and their base assumes a vertical orientation; pelvic fins move into thoracic or even jugular (throat) position. These relocations have several apparent functions (see Webb 1982). Pectorals on the side can be sculled for fine movement and positioning, such as slow
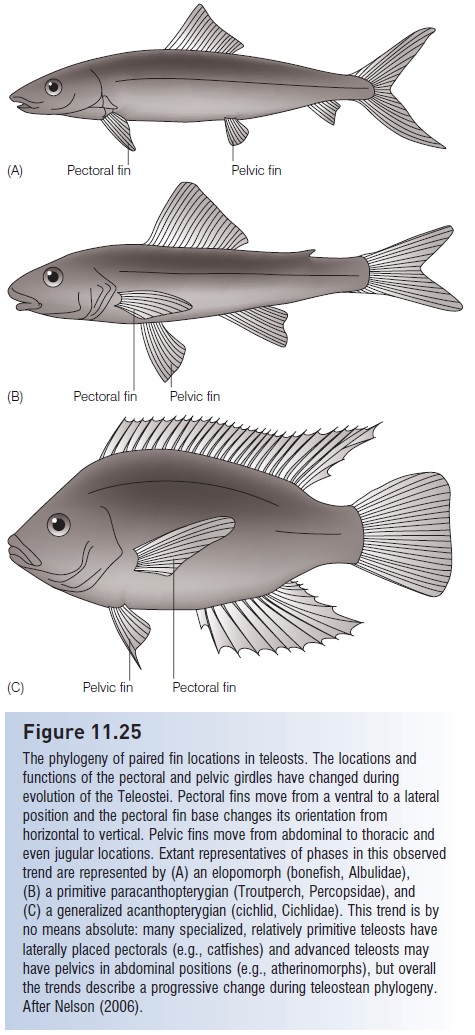
Figure 11.25
The phylogeny of paired fin locations in teleosts. The locations and functions of the pectoral and pelvic girdles have changed during evolution of the Teleostei. Pectoral fins move from a ventral to a lateral position and the pectoral fin base changes its orientation from horizontal to vertical. Pelvic fins move from abdominal to thoracic and even jugular locations. Extant representatives of phases in this observed trend are represented by (A) an elopomorph (bonefish, Albulidae), (B) a primitive paracanthopterygian (Troutperch, Percopsidae), and (C) a generalized acanthopterygian (cichlid, Cichlidae). This trend is by no means absolute: many specialized, relatively primitive teleosts have laterally placed pectorals (e.g., catfishes) and advanced teleosts may have pelvics in abdominal positions (e.g., atherinomorphs), but overall the trends describe a progressive change during teleostean phylogeny. After Nelson (2006).
swimming, and hovering and backing in midwater. As these fins are often transparent, their use in locomotion might be less obvious to a potential prey animal than would be lateral undulations of the body. Placement of the pelvics forward helps in braking and reduces pitching; their location under the spinous dorsal, in combination with spinous armament, increases the effective body depth of a fish at the point at which it is most likely to be attacked by a predator (Webb & Skadsen 1980).
Caudal fin and gas bladder modifications Actinopterygian evolution is characterized by a progressive increase in symmetry of the tail fin (see Fig. 11.19). Tail fins became externally and functionally homocercal fairly early in the group’s history. Fossil impressions of the tails of late Paleozoic palaeoniscoids show that the upper and lower lobes were equal, in contrast to the heterocercal and abbreviate heterocercal tails of earlier palaeoniscoids. Symmetry becomes more pronounced in the teleosts, reflecting the internal modifications that followed. These internal changes include notochord and body shortening and the reworking of large bones and sets of bones that support the caudal fin rays. In particular, teleosts developed a series of hypural bones from several haemal arches. Some of these bones fused to form a ventral hypural plate, continuing a trend evident in palaeonisciforms. Fusion and reduction of number of vertebrae, reduction of intermuscular bones, and increased tail symmetry all correlate with a greater role of the caudal region in locomotion. The trend is toward an increased dependence on high power caudal swimming, culminating in steadily swimming fishes with lunate tails, such as jacks (Carangidae), tunas (Scombridae), and billfishes (Istiophoridae) (Webb 1982). Primitive teleosts used sequential contraction of trunk musculature throughout the body, producing a wave of contraction from head to tail. By focusing muscle contraction on the tail and its supporting structures, advanced teleosts could swim faster and more efficiently than more primitive teleosts that depended on sinusoidal movement of the body.

In apparent conjunction with tail and paired fin modifications, an additional teleostean trend is added control over gas bladder function. It can be debated whether gas bladders arose initially as breathing or buoyancy control structures (see Buoyancy regulation), but the latter function has taken precedence in teleosts. Living pre-teleosteans and primitive teleosteans have aphysostomous gas bladder, in which a pneumatic duct connects the gas bladder with the gut and ultimately the mouth. The gas bladder is filled with gas by gulping air; gas is expelled largely via the same route. Fine adjustments are difficult in this system, and the fish is somewhat dependent on access to the surface. More advanced teleosts (paracanthopterygians, acanthopterygians) arephysoclistous, having lost the pneumatic duct and the link to atmospheric air. They instead rely more on internally generated and absorbed gases to fill and empty the gas bladder and are capable of finer control of buoyancy (physostomous fishes have gas secretion capabilities, but are usually not as refined as in physoclists; see Fig. 5.8).
Although gas bladders do not normally fossilize, the codevelopment of a gas-filled, internally controlled gas bladder, a homocercal tail, and paired, multifunctional, fl exible fins is taken as a strong indication that they evolved as a suite of characters. A gas bladder makes an otherwise dense fish neutrally buoyant, which means that small, precise adjustments in body orientation and movement will not be counteracted by continuous sinking. Thus a fish can remain at a fixed point in the water column and turn on its body axis without moving forward. This trend toward a combination of a functionally homocercal tail and some sort of internal hydrostatic organ was previously evident in the three major sarcopterygian groups. Lungfishes, osteolepidimorphs/ elpistostegalians, and coelacanths evolved symmetrical, diphycercal tails. The first two groups had functional gas bladders (lungs), and early coelacanths possessed a gas bladder (Lund & Lund 1985), although it is small and fat-filled in the living coelacanths. Interestingly, the feeding mode of the living coelacanths involves hovering relatively still in open water by a combination of paired fin movements and buoyancy control (Fricke et al. 1987).
Feeding apparatus modifications Two major changes have characterized the anatomy of foraging in teleosts.
1 Teleosts continue a trend seen in neopterygians with respect to increasing suction capabilities. Teleosts developed a protrusiblepipette mouth, capable of generating powerful, directed, negative pressures (see Figs 8.4, 8.5). The pipette mouth results from enlargement of the muscles and modifications to the bones in the jaw apparatus, most notably the maxilla and hyomandibula, but also involves connections with the mandibular, opercular, and pectoral bone series. Early in teleostean evolution, the rear portion of the maxilla was freed from its connection with other cheek bones, allowing it to swing forward and allowing the reoriented hyomandibula to move outward, thus increasing mouth volume. Skin folds developed along the lateral margins of the jaw bones, creating a hole-free tubular apparatus that prevents lateral escape by small prey. In the more advanced groups (Paracanthopterygii and particularly the Acanthopterygii), the premaxilla develops an ascending process which is basically a vertical extension at its anterior tip that slides along the front of the skull, thus allowing the premaxilla to shoot forward at prey as the mouth is opened (see Jaw protrusion: the great leap forward).
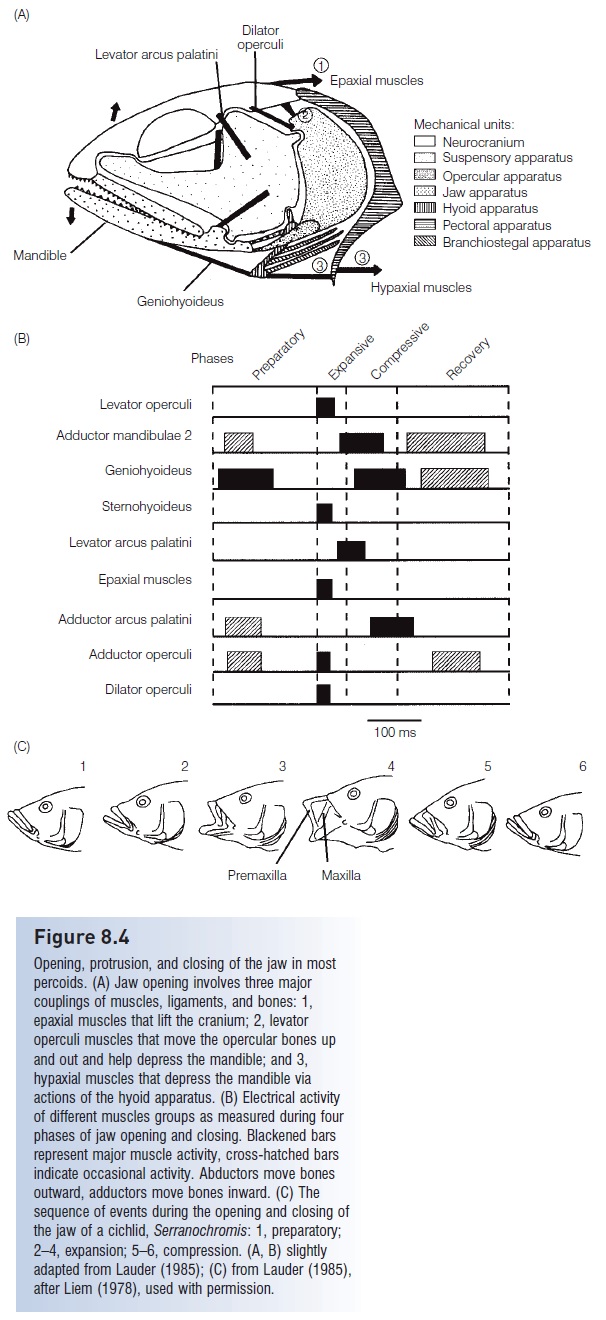
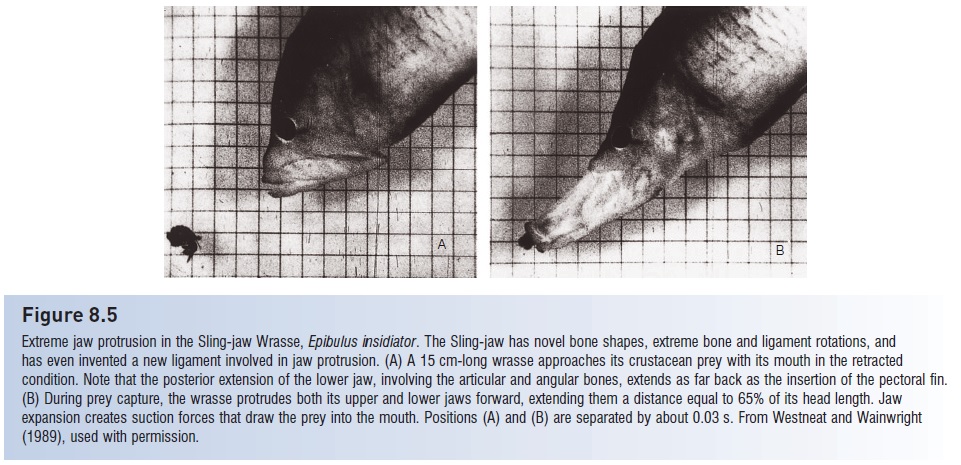
The end product of action in this complex of bones, muscles, ligaments, and pivot points is very rapid expansion of the orobranchial chamber. In paracanthopterygian anglerfishes, mouth volume can increase 13-fold over the course of 7 ms (7/1000 of a second) (Pietsch & Grobecker 1987). Maximal expansion of the gape is one direction these changes take, particularly in predators on other fishes. In feeders on zooplankton and other small prey, many advanced teleosts have, if anything, reduced gape width to increase suction power. And some groups have maximized the speed of mout extension without suction. For example, zooplanktivorousChromis damselfish (Pomacentridae) can fully protrude their jaws in as little as 6 ms to capture evasive copepods (Coughlin & Strickler 1990). During jaw protrusion in the pikehead, Luciocephalus pulcher (Luciocephalidae), head length is increased by one-third at a rate of 51 cm/s, thus increasing the speed of attack by almost 40%, with no appreciable suction force generated; velocity increases of up to 89% have been recorded in Largemouth Bass (Nyberg 1971; Lauder & Liem 1981). Observations of mouth extension without suction have fueled a debate as to whether the pipette mouth developed primarily to generate suction power for inhaling prey or to rapidly extend the anterior portion of the body to overtake prey (Lauder & Liem 1981; Lauder 1982). Regardless, the primary selection pressure driving these modifications in the jaw was undoubtedly facilitation of prey capture.
2 Once prey are captured, they are passed back into the mouth to be manipulated. For soft-bodied prey, including most small fishes, manipulation primarily involves positioning the prey to facilitate head-first swallowing, thus avoiding the improved teleostean fin spines that might cause choking or blockage if prey are swallowed tail first. Later digestion of fish prey requires little more than chemical breakdown in the gut. However, many potential prey have extremely effective physical defenses that are relatively impervious to gut chemistry. The hard, calcareous shells of mollusks, the chitinous exoskeletons of crustaceans, and the cell walls of plants all require mechanical rupturing before digestive enzymes can have much effect. A protrusible mouth is effective for initial capture of prey, but, because of the emphasis on fore–aft movement, protrusibility evolved at a sacrifi ce in up-and-down chewing motion. Therefore mechanical rupturing must occur elsewhere. In teleosts, it has been the dentition and musculature of thepharyngeal “jaws” that have diversified to serve this chewing, crushing, and grinding function (Lauder 1982).
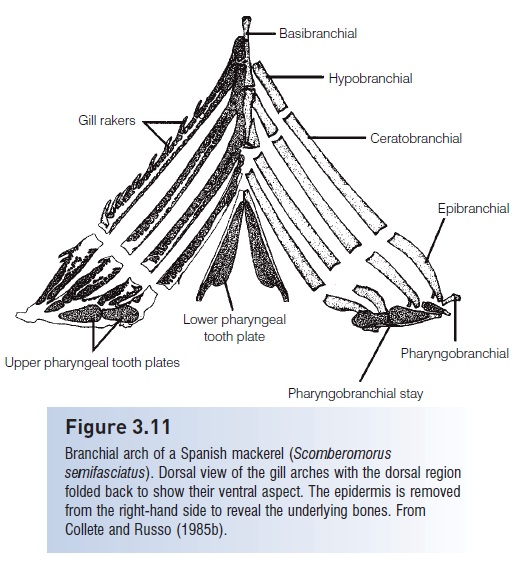
Pharyngeal pads lie posterior to the marginal jaws, just anterior to the esophagus (see Fig. 3.11), and are derived from dermal tooth plates in the pharynx. During teleostean phylogeny, the function of these pads has elaborated from simple holding of prey prior to swallowing to manipulation and preparation that facilitates digestion. The pads have become armed with a variety of dentition types and have fused to dorsal and ventral elements of the gill arches. The branchial musculature has been reworked and a new muscular connection from the anterior vertebral column has been made, to bring upper and lower plates together in complex, powerful movements. Hence in acanthopyterygian groups with this pharyngognathous condition, we find the molluskfeeding croakers and drums (Sciaenidae) with molariform dentition, parrotfishes (Scaridae) with pharyngeal jaws capable of grinding up coral rock to expose the algae contained therein, and the highly successful cichlids with a variety of pharyngeal tooth and jaw arrangements that allow their food to be “crushed, triturated, macerated, compacted or in other ways prepared”. The development and diversifi cation of pharyngeal jaws and dentition has undoubtedly broadened the diet of teleosts to include hard-bodied prey and, more importantly, plant material; herbivory is essentially unknown in non-teleostean fishes. This diversification probably extended teleost foraging capabilities far beyond what was possible with the early actinopterygian dependence on more anterior jaw elements. It is more than coincidence that several of the most successful modern teleostean families (cyprinids, cichlids, labrids) have both highly protrusible front jaws and diversified pharyngeal jaws.
Although our emphasis above has been on identifying five general areas that changed during teleostean phylogeny, it is important to remember that these traits changed in concert, that anatomical trends during teleostean phylogeny represent a suite of adaptations. Modification of one trait probably enhanced the effectiveness of and was affected by the other traits. The greatest manifestation of the trends is evident in the acanthopterygian fishes, with their ctenoid scales, diversified yet spiny fins, symmetrical tails, fine maneuverability via pectoral fin sculling, physoclistous gas bladders, greatly expandable mouth volumes, and effective pharyngeal teeth. The end result “has been increased swimming speed combined with maneuverability . . . without significant loss of defensive structures” (Gosline 1971, p. 152). In other words, higher teleosts represent quick, spiny fish with a highly efficient feeding apparatus that can catch and eat small, hard prey items.
Also note that these trends generally describe different taxonomic groups but in no way preclude the possibility of a primitive group deriving specializations characteristic of a more advanced taxon. For example, true eels (a relatively primitive teleostean order) as well as other eel-like fishes, regardless of taxonomic position, have expanded dorsal and anal fins, greatly reduced or absent scales, and missing pelvic and even pectoral fins. Elaborate median fins are not found solely in advanced superorders. Many osteoglossomorphs are highly derived, specialized fishes that use their own electrical output to locate objects and locomote via an elongate dorsal (gymnarchids) or anal (gymnotids) fin. Many adult deepsea fishes (Stenopterygii, Cyclosquamata, Scopelomorpha), although belonging to relatively primitive superorders that should be characterized by physostomous gas bladders, are instead secondarily physoclistous, probably to prevent gas loss via the gut and because they never go to the surface to gulp air. Elaborate pharyngeal dentition, a hallmark of the Acanthopterygii, is used widely in the relatively primitive minnows and suckers (Ostariophysi). A protrusible mouth – brought on by an ascending, sliding premaxillary process – characterizes acanthopterygians and the closely related paracanthopterygians, but was evolved independently and differently in a more primitive group, the ostariophysan cypriniforms, as well as in elasmobranchs and sturgeons. Environmental conditions determine the selection pressures operating on a lineage; groups that evolve more effective adaptations will be favored whether or not they are “breaking the rules” of teleostean phylogeny.
Related Topics