Chapter: The Diversity of Fishes: Biology, Evolution, and Ecology: A history of fishes
Subclass Chondrostei - Class Actinopterygii, Advanced jawed fishes : teleostomes (Osteichthyes)
Subclass Chondrostei
The origins of the Actinopterygii are once again obscure. Scale fragments appear in Late Silurian marine deposits, which may mean that the group is older than the sarcopterygians and as old as placoderms and elasmobranchs. Only the acanthodians among the bony, jawed fishes are of greater antiquity, supporting speculation of an acanthodian ancestry for modern bony fishes. However, complete fossil actinopterygians do not appear until the Mid to Late Devonian, when the group had expanded into a variety of marine, estuarine, and freshwater habitats. These early fishes, collectively known as palaeoniscoids (but see below) were relatively small (5–25 cm) and were distinguished from sarcopterygians by the presence of a single triangular dorsal fin, a forked heterocercal tail with no upper lobe above the unconstricted notochord, paired fins with narrow rather than fleshy bases, dermal bones lacking a cosmine layer, scales joined by a peg-and-socket arrangement and covered with ganoine (“ganoid” scales), relatively large eyes, and a blunt head (Fig. 11.19A, B). The term “ray-fin” refers to the parallel endoskeletal fin rays that were derived from scales. These rays supported the median and paired fins, which were moved by adjacent body musculature. In contrast, the fins of the Sarcopterygii had a thick, bony central axis and muscles contained in the fin itself (see Fig. 11.15C). Chondrosteans include one extant and 10 extinct orders, with relationships obscure. The most primitive group, the Devonian cheirolepidiforms, includes a species with the distinction of possessing the largest number of pelvic fin rays known among fishes, living or otherwise. Cheirolepis canadensis had 124 such rays versus six or fewer in living teleosts. The most diverse order, the Palaeonisciformes, contained four suborders and 17 families of wellrepresented fishes that showed tremendous morphological diversity (Figs 11.19, 11.20). The other orders of early chondrosteans are often lumped together as “palaeoniscoids” despite taxonomic differences, and palaeoniscoids are then treated as ancestral to later neopterygians and therefore teleosts.
Among the other orders are the Carboniferous tarrasiiforms, which were remarkably convergent with many modern eel-like forms, possessing an elongate body, dorsal and anal fins continuous with the caudal fin (the latter being diphycercal in this group), and pelvic fins and scales reduced or absent (Fig. 11.20). Saurichthyiforms converged on a needlefish body shape and are thought to have been similarlypredatory on small fishes, and phanerorhynchiforms bore a superficial resemblance to modern sturgeons. An advanced order from the Late Triassic, the perleidiforms, included Thoracopterus, a genus with expanded paired fins thought capable of biplane gliding, as occurs in modern exocoetid flyingfishes (Tintori & Sassi 1992). Thoracopterus possessed the enlarged pectoral and pelvic fins, reinforced rays in the paired fins, asymmetrical caudal fin, expanded caudal neural spines for muscle insertion, posterior position of dorsal and anal fins, and head shape of

Figure 11.19
Actinopterygian fishes at different grades of development. (A) Moythomasia and (B) Mimia, two primitive palaeoniscoid fishes from the Upper Devonian, with thick rhomboidal scales extending onto the fins, broadly triangular dorsal and anal fins, fulcral (ridge) scales along the back, a long mouth, and an asymmetrical heterocercal tail. (C) Parasemionotus, a pre-teleostean neopterygian from the Triassic, showing more flexible fins, shorter mouth, and abbreviate heterocercal tail. (D) Eolates, an advanced euteleost from the Lower Eocene, with characteristic teleostean diversified dorsal and anal fins, shortened vertebral column, premaxillary dominated upper jaw, and homocercal tail. (A) after Jessen (1966); (B) after Gardiner (1984); (C) after Lehman (1966); (D) after Sorbini (1975).

Figure 11.20
Morphological (and ecological) convergence in fish evolution. Palaeoniscoids were ancestral to early neopterygians, which were ancestral to modern teleosts. Certain body designs or plans have apparently been repeatedly favored in actinopterygians, leading to convergent designs among unrelated lineages. These striking convergences in body shape and presumably function are depicted for representative palaeoniscoids, early neopterygians, and teleosts. 1, Elongate piscivores with long, ooth-studded jaws and dorsal and anal fins placed posteriorly for rapid starts; 2, compressed-bodied, predatory, shallow water fishes with deeply forked tails and trailing fins; 3, broad-finned bottom feeders with subterminal mouths; 4, eel-like benthic forms; and 5, compressed, circular forms with large fins for maneuverability in shallow water habitats with abundant structure. Gliding fishes such as the Triassic chondrostean Thoracopterus (Fig. 11.21) can also be equated with modern teleostean flyingfishes. Adapted from Pough et al. (1989), not drawn to scale.
Palaeoniscoid trends
Palaeoniscoids fl ourished throughout the latter Paleozoic. Meanwhile, ostracoderms, acanthodians, and placoderms disappeared and sarcopterygians diminished in abundance. This correlation suggests ecological interaction among groups, and possible replacement of primitive jawed and jawless fishes with more advanced actinopterygian and chondrichthyan lineages. What innovations did the rayfinned fishes possess that might have given them ecological superiority? The available evidence strongly suggests that, once again, changes in jaw and fin structure leading to diversified feeding habits and increased mobility were critical to actinopterygian success and dominance.
Changes in the mechanics of jaw opening and closing during actinopterygian phylogeny have been the subject of intensive study (e.g., Lauder & Liem 1981; Lauder 1982; see also Carroll 1988, Pough et al. 2005 for reviews). The highly ossified braincase of the early actinopterygians makes it possible to determine the origins, insertions, and approximate sizes of the different muscle masses involved in jaw function, from which we can estimate the forces in operation.
During actinopterygian evolution, culminating in advanced teleosts, changes in the angles and connections between the skull case, dermal bones, muscles, and ligaments of the head and jaws have been most infl uential. In particular, the hyomandibula has been reoriented from oblique to vertical, the posterior end of the maxilla has been freed from the cheek bones, and the jaw musculature has increased in size and complexity. These changes increased the speed and strength of the bite. They also allowed for enlarging of the mouth both vertically and laterally. Hence when the mouth was opened, its volume increased and it assumed a more tubular shape. This changed the bite of a
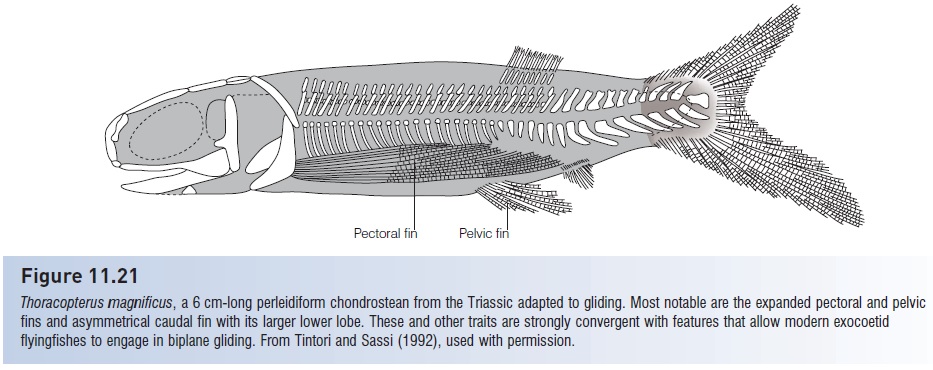
Figure 11.21
Thoracopterus magnificus, a 6 cm-long perleidiform chondrostean from the Triassic adapted to gliding. Most notable are the expanded pectoral and pelvic fins and asymmetrical caudal fin with its larger lower lobe. These and other traits are strongly convergent with features that allow modern exocoetid flyingfishes to engage in biplane gliding. From Tintori and Sassi (1992), used with permission.
In the modified condition, when the mouth is opened, water and prey are sucked in; when the mouth is closed, instead of water being pushed back out through the jaws, flow continues posteriorly through the gill slits, thereby trapping prey inside rather than pushing it back out of the mouth. Transport of water over the gills during breathing may also have been facilitated by these modifications.
Apparent improvements in other skeletal components are no less important. Palaeoniscoid scales changed from heavy, interlocking, diamond-shaped units to thinner, lighter, circular, cycloid structures. This reduction was accomplished by elimination of the dentine, vascular, and ganoine layers. Because palaeoniscoid fins consisted of jointed scales, reduction in scale thickness meant increased flexibility in fins; fins became mobile structures composed of dermal fin rays that could be erected or lowered and also moved laterally. Associated with scale reduction was increased ossifi cation of the vertebral column, leading to recognizable centra with dorsal and ventral accessory structures (neural and haemal arches). These accessory structures are closely related to modifications in the caudal region, where a major trend has been toward an increasingly symmetrical, homocercal tail (see Fig. 11.19). Caudal fin rays became supported by a series of ventral accessory,
hypural bones.
All these changes during palaeoniscoid phylogeny imply increased reliance on locomotion, integrated in both escape and prey capture. Heavy ganoid scales offer passive protection against predators but do not function until a predator has already captured a prey individual, a risky event that most potential prey would undoubtedly rather avoid. Lighter scales mean a lighter, more fl exible body, capable of more rapid swimming and quicker turns. Greater reliance on a gas bladder for attaining neutral buoyancy has also been suggested, which also frees fins to provide propulsion and maneuverability. Weight reduction of fins allows them to better serve as propellants, or as brakes and flaps for swimming, stopping, and turning (and gliding?). The correlation between dermal armor reduction and increased vertebral ossifi cation may indicate a shift from reliance on an external elastic/hydrostatic skeleton to an internal, muscular/ tendonous system (see Locomotion: movement and shape). Increased speed and mobility, combined with the already mentioned improvements in mouth structure, would mean that more advanced actinopterygians would not only be better at avoiding predators but also at capturing prey. These trends in palaeoniscoid evolution reoccur later in more advanced actinopterygian lineages (see Fig. 11.20).
Related Topics