Chapter: 11th 12th std standard Class Organic Inorganic Physical Chemistry Higher secondary school College Notes
The structure of the three isotopes of hydrogen
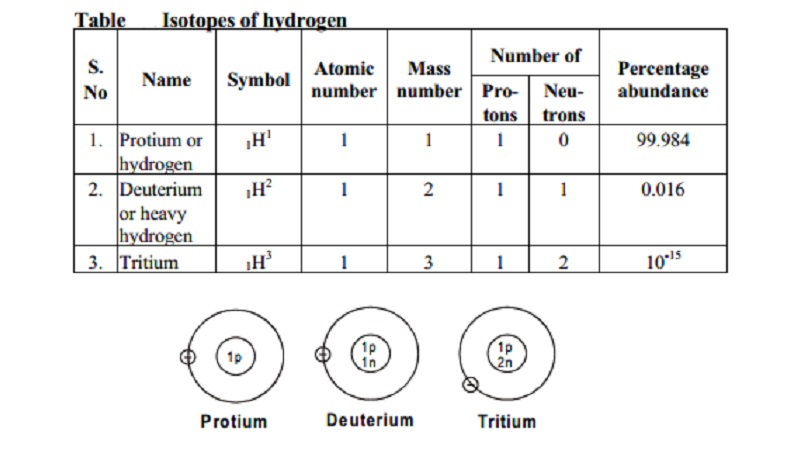
Isotopes
of hydrogen
Hydrogen is the first element in the periodic table. It has the simplest
electronic configuration 1s1. It contains one proton in the nucleus
and one electron.
Isotopes:- Atoms of the same element having same atomic number but
different mass number are called isotopes.
There are three isotopes for hydrogen with mass numbers 1, 2 and 3, each
possessing an atomic number of one.
The
structure of the three isotopes of hydrogen are
1. Protium or ordinary
hydrogen: It is the common form of hydrogen. It consists of one proton in its nucleus
and one electron revolving around it. It constitutes 99.984% of total hydrogen
available in nature. Its mass number is one.
2. Deuterium or heavy
hydrogen: 1H2 or 1D2. It
occurs naturally in very small traces.
The proportion present in naturally occurring hydrogen is in the approximate
ratio: D: H~ 1:6000. It's nucleus consists of a proton and a neutron. However
only a solitary electron is revolving around the nucleus. Its chemical
properties are similar to those of protium but their reaction rates are
different.
3.
Tritium, 1H3 or 1T3: It occurs in the upper atmosphere only where it is continuously formed by nuclear reactions induced by cosmic
rays. Unlike deuterium, it is radioactive, with a half-life of ~ 12.3 years.
It's nucleus consists of one proton and two neutrons.
They will have same similar chemical properties, however, their reaction
rates will be different and their physical properties differ appreciably.
Isotopes of hydrogen :
S. Atomic Mass Number
of Percentage
Name Symbol
No number number Pro- Neu- abundance
tons trons
1. Protium
or 1H1 1 1 1 0 99.984
hydrogen
2. Deuterium 1H2 1 2 1 1 0.016
or
heavy
hydrogen
3. Tritium 1H3 1 3 1 2 10-15
Related Topics