Chapter: Introduction to Human Nutrition: Minerals and Trace Elements
Selenium: Absorption, transport, tissue distribution, Metabolic function, essentiality, Deficiency symptoms
Selenium
Selenium is a nonmetallic element that has similar chemical properties to sulfur and has four natural oxidation states (0, −2, +4, +6). It combines with other elements to form inorganic selenides [sodium selenide (−2) Na2Se], selenites [sodium selenite (+4) Na2SeO3] and selenates [sodium selenate (+6) Na2SeO4], and with oxygen to form oxides [selenium (+4) dioxide SeO2] and oxyacids [selenic (+6) acid H2SeO4]. Selenium replaces sulfur to form a large number of organic selenium compounds, parti-cularly as selenocysteine, the twenty-first amino acid. Selenium is a component of selenoproteins, where it also occurs as selenides on the side-chains of seleno-cysteine at physiological pH. Selenium also displaces sulfur to form the amino acid selenomethionine.
Elemental selenium is stable and has three allotropic forms, deep red crystals, red amorphous powder, and the black vitreous form.
Selenium has many industrial uses, e.g., in elec-tronics, glass, ceramics, pigments, as alloys in steel, as catalysts in pharmaceutical production, in rubber vulcanization and in agriculture, as feed supplements and fertilizers. Because of its increasing use, selenium has become a potential health and environmental hazard. The primary pathway of exposure to selenium for the general population is food, followed by water (predominantly inorganic selenate and selenite), and air (mainly as elemental particulate selenium from combustion of fossil fuels and from volcanic gas).
Absorption, transport and tissue distribution
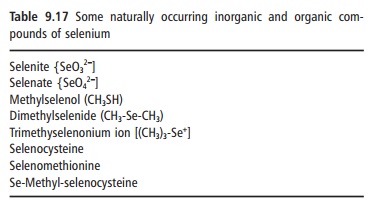
Absorption of dietary selenium takes place mainly in the small intestine, where some 50–80% is absorbed. Organic forms of selenium are more readily absorbed than inorganic forms and selenium compounds from plants are generally more bioavailable than those from animals, and particularly from fish. Some natu-rally occurring inorganic and organic compounds of selenium are given in Table 9.17.
The bioavailability of selenium from water (mainly inorganic selenates) and supplements is lower than from food. The overall bioavailability of selenium from the diet depends on a number of factors, includ-ing selenium status, lipid composition, and metals.
Inorganic forms of selenium are passively trans-ported across the intestinal brush border, whereas organic forms (selenomethionine and probably sele-nocysteine) are actively transported. On reaching the bloodstream, selenium is transported largely bound to protein (mainly very low-density β-lipoprotein with a small amount bound to albumin) for
Table 9.17 Some naturally occurring inorganic and organic com-pounds of selenium
deposition in various organs. Liver and kidney are the major target organs when selenium intake is high but, at lower intakes, the selenium content of the liver is decreased. Heart and muscle tissue are other target organs, with the latter, because of its total bulk, accounting for the greatest proportion of body selenium. The total body content of selenium can vary from about 3 mg to 15 mg depending on dietary intakes.
In the body, dietary selenium can be bound to sele-nium binding proteins but can also be directly incor-porated into selenoproteins during translation at the ribosome complex using a transfer RNA specific for the amino acid selenocysteine; thus, selenocysteine can be considered as the twenty-first amino acid in terms of ribosome-mediated protein synthesis.
The major excretion routes of selenium are in urine (mainly as trimethylselenonium ion), in feces (via biliary pancreatic and intestinal secretions, together with unabsorbed dietary selenium), and in breath (as volatile dimethylselenide). Unlike copper, and partic-ularly iron, which have inefficient excretion mecha-nisms, selenium is rapidly excreted in the urine. Figure 9.7 gives an overall view of selenium metabolism and excretion.
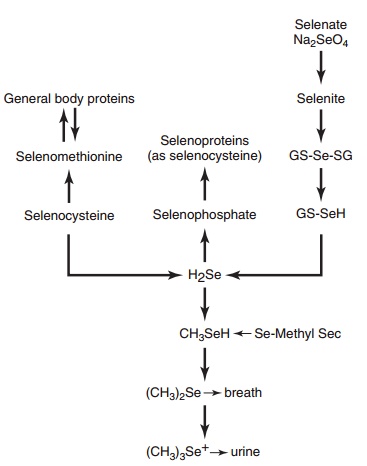
Figure 9.7 Selenium metabolism and excretion.
Metabolic function and essentiality
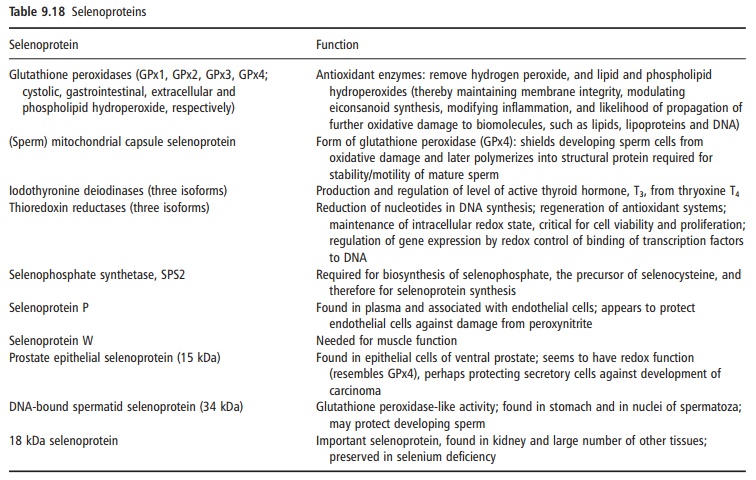
Selenocysteine is a component of at least 30 seleno-proteins, some of which have important enzymic functions (Table 9.18). Selenocysteine is generally at the active site of those selenoproteins with catalytic activity, and functions as a redox center for the selenium-dependent glutathione peroxidases (cys-tolic, phospholipid hydroperoxide, extracellular, and gastrointestinal), iodothyronine deiodinases (types I, II, and III), and thioredoxin reductases. The glutathi-one peroxidase isozymes, which account for about 36% of total body selenium, differ in their tissue expression and map to different chromosomes.
Deficiency symptoms
Keshan’s disease is a cardiomyopathy that affects chil-dren and women of child-bearing age and occurs in areas of China where the soil is deficient in selenium. Despite the strong evidence for an etiological role for selenium in Keshan’s disease (i.e., the occurrence of the disease only in those regions of China with low selenium soils and, hence, low amounts of selenium in the food chain, and only in those individuals with
poor selenium status together with the prevention of the disease in an at-risk population by supplementa-tion with selenium), there are certain epidemiological features of the disease that are not readily explained solely on the basis of selenium deficiency. A similar situation occurs with Kashin–Beck disease, a chronic osteoarthropathy that most commonly affects growing children and occurs in parts of Siberian Russia and in China, where it overlaps with Keshan’s disease. Although oral supplementation with selenium is effective in preventing the disease, it is likely that other factors, apart from selenium deficiency, are involved in the etiology of Kashin–Beck disease. There are also some selenium-responsive conditions with symptoms similar to Keshan’s disease that occur in patients receiving total parenteral nutrition.
One explanation for the complex etiology of sele-nium-responsive diseases in humans is that low selenium status may predispose to other deleterious conditions, most notably the increased incidence, virulence, or disease progression of a number of viral infections. For example, in a selenium-deficient animal model, harmless coxsackie virus can become virulent and cause myocarditis, not only in the
A coxsackie virus has been isolated from the blood and tissues of patients with Keshan’s disease and the infection may be responsible for the cardiomyopathy of that disease. It has been speculated that similar events linked with other RNA viruses may explain the emergence of new strains of influenza virus in China and the postulated crossing-over of the human immunodeficiency virus (HIV) to humans in the selenium-deficient popula-tion of Zaire. Many human viral pathogens (e.g., HIV, coxsackie, hepatitis, and measles viruses) can synthe-size viral selenoproteins and, thereby, lower the sele-nium available to the host. In any event, selenium deficiency is accompanied by loss of immunocompe-tence, with the impairment of both cell-mediated immunity and B-cell function. Covert suboptimal selenium status may be widespread in human popula-tions, as selenium supplementation in subjects considered to be selenium replete had marked immu-nostimulant effects, including increased proliferation of activated T-cells. Such immunostimulant effects or the production of antitumorigenic metabolites may explain the lowering of cancer incidence, particularly prostate cancer, after selenium supplementation in selenium-replete subjects (those who already had maximized selenoenzyme activity). Other proposed mechanisms for a cancer chemoprotective effect of selenium include antioxidant protection and reduc-tion of inflammation; inactivation of protein kinase C; altered carcinogen metabolism; reduction in DNA damage, stimulation of DNA repair (p53), and altera-tion in DNA methylation; cell cycle effects; enhanced apoptosis and inhibition of angiogenesis. Further evidence for any chemoprotective effect of selenium against cancer should arise from the Selenium and Vitamin E cancer Prevention Trial (SELECT), which is a large randomized controlled trial investigating the efficacy of selenium (200 μg of L-selenomethionine) and vitamin E (400 IU, dl-α-tocopherol acetate) alone and in combination for the prevention of prostate cancer in over 35 000 healthy men in 435 sites in the USA, Puerto Rico, and Canada and which should report sometime after 2008.
The evidence for suboptimal selenium status increasing the risk of cardiovascular disease is more fragmentary, but it has been proposed that optimiz-ing the activity of the seleno-dependent glutathione peroxidases and, thereby, increasing antioxidant activ-ity may be a factor. As selenium has well-recognized antioxidant and anti-inflammatory roles, other oxi-dative stress or inflammatory conditions (e.g., rheu-matoid arthritis, ulcerative colitis, pancreatitis, and asthma) may benefit from selenium supplementation. In addition, some, but certainly not all, studies have suggested beneficial (possibly antioxidant) effects of selenium on mood and reproduction in humans. The evidence, however, supporting a role for optimum selenium status preventing or ameliorating most inflammatory conditions is not strong and may be confounded by other dietary antioxidants, particu-larly vitamin E, compensating for low selenium status.
Related Topics