Chapter: Introduction to Human Nutrition: Minerals and Trace Elements
Calcium: Absorption, transport, tissue distribution, Metabolic function, essentiality, Deficiency symptoms
Calcium
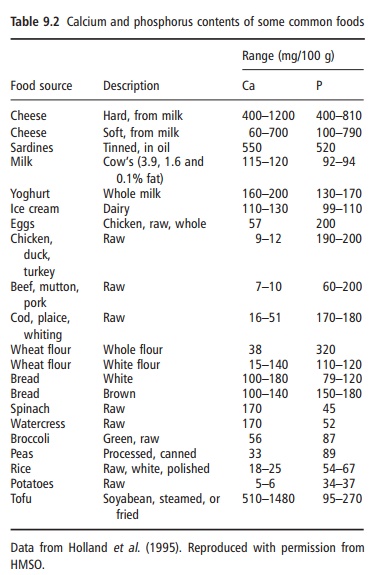
Calcium is a metallic element, fifth in abundance in the Earth’s crust, of which it forms more than 3%. Calcium is never found in nature uncombined; it occurs abundantly as chalk, granite, eggshell, sea-shells, “hard” water, bone, and limestone. The metal is used as a reducing agent in preparing other metals such as thorium, uranium, and zirconium, and is used as a deoxidizer, disulfurizer, or decarburizer for various ferrous and nonferrous alloys. It is also used as an alloying agent for aluminum, beryllium, copper, lead, and magnesium alloys. Calcium was among the first materials known to be essential in the diet. All foods of vegetable origin contain small but useful amounts of calcium. Animals concentrate calcium in milk, and milk and dairy products are the most important food sources of calcium for many human populations.
Absorption, transport, and tissue distribution
The adult human body contains about 1200 g of calcium, which amounts to about 1–2% of body weight. Of this, 99% is found in mineralized tissues, such as bones and teeth, where it is present as calcium phosphate (together with a small component of calcium carbonate), providing rigidity and structure. The remaining 1% is found in blood, extracellular fluid (ECF), muscle, and other tissues.
Calcium is under close homeostatic control, with processes such as absorption, excretion and secretion, and storage in bone being involved in maintaining the concentration of ionized calcium in the plasma within a tightly regulated range. This tight regulation of plasma calcium concentration is achieved through a complex physiological system comprising the interac-tion of the calcitropic hormones, such as parathy-roid hormone (PTH), 1,25-dihydroxycholecalciferol [1,25(OH)2D3] and calcitonin, with specific target tissues (kidney, bone, and intestine) that serve to increase or to decrease the entry of calcium into the extracellular space (plasma) (Figure 9.2). Only in extreme circumstances, such as severe malnutrition or hyperparathyroidism, is the serum ionized calcium concentration below or above the normal range. The secretion of these hormones is governed wholly, or in part, by the plasma concentration of ionized calcium, thus forming a negative feedback system. PTH and 1,25(OH)2D3 are secreted when plasma calcium is low, whereas calcitonin is secreted when plasma calcium is high.
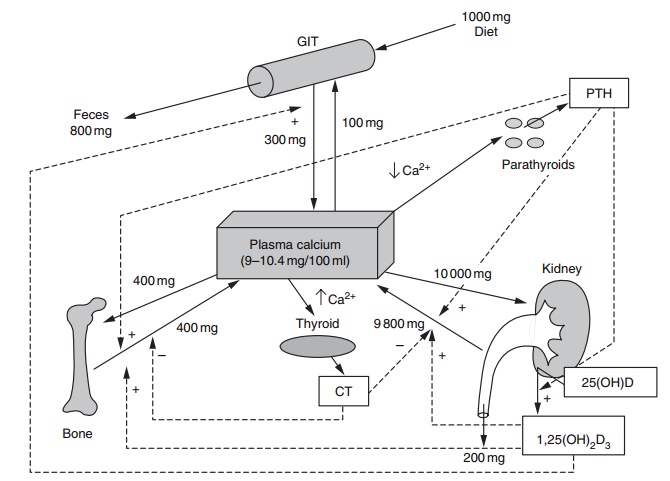
Figure 9.2 Homeostatic regulation of serum calcium, showing the integration of hormone action at the tissue level. CT, calcitonin; PTH, para-thyroid hormone; 1,25(OH)2D3, 1,25-dihydroxycholecalciferol (to convert from mg/day to mmol/day multiply by 40).
Calcium in food occurs as salts or associated with other dietary constituents in the form of complexes of calcium ions. Calcium must be released in a soluble, and probably ionized, form before it can be absorbed.
Calcium is absorbed in the intestine by two routes, transcellular and paracellular (Figure 9.3). The trans-cellular route involves active transport of calcium by the mucosal calcium transport protein, calbindin, and is saturable and subject to physiological and nutri-tional regulation via vitamin D. The paracellular route involves passive calcium transport through the tight junctions between mucosal cells; it is nonsaturable, essentially independent of nutritional and physiologi-cal regulation, and concentration dependent. Most calcium absorption in humans occurs in the small intestine, but there is some evidence for a small colonic component. Transcellular calcium absorption responds to calcium needs, as reflected by changes in plasma calcium concentration, by hormone-mediated up- or down-regulation of calbindin in mucosal cells; for example, reduced plasma calcium evokes a PTH-mediated increase in plasma 1,25(OH)2D3, which stimulates increased calbindin synthesis in intestinal mucosal cells.
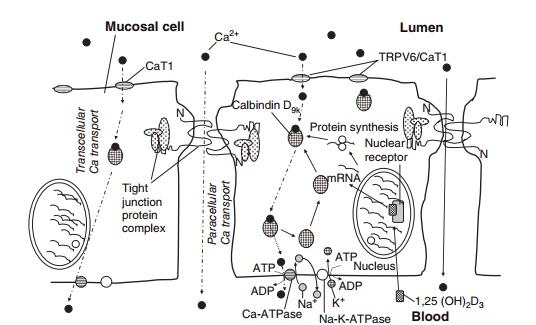
Figure 9.3 Calcium transport across the intestinal mucosal lining: paracellular calcium transport (between mucosal cells) and transcellular calcium transport (across the mucosal cell).
On average, between 10% and 30% of the calcium is absorbed from a mixed diet by healthy adults.
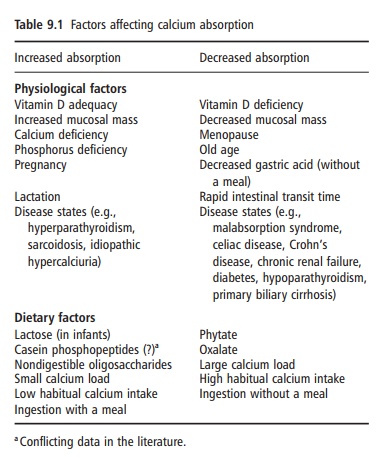
However, the efficiency of intestinal calcium absorp-tion is influenced by a variety of physiological factors (Table 9.1). Calcium absorption may also be influ-enced by a number of dietary factors (Table 9.1).
Metabolic function and essentiality
Calcium is required for normal growth and develop-ment of the skeleton. During skeletal growth and maturation, i.e., until the early twenties in humans, calcium accumulates in the skeleton at an average rate of 150 mg/day. During maturity, the body, and there-fore the skeleton, is more or less in calcium equilib-rium. From the age of about 50 years in men and from the menopause in women, bone balance becomes negative and bone is lost from all skeletal sites. This bone loss is associated with a marked rise in frac-ture rates in both sexes, but particularly in women. Adequate calcium intake is critical to achieving optimal peak bone mass (PBM) and modifies the rate of bone loss associated with aging. Extraskeletal calcium (representing around 1% of total body calcium) plays a role in mediating vascular contrac-tion and vasodilatation, muscle contraction, nerve transmission, glandular secretion, and as an impor-tant second messenger molecule.
Deficiency symptoms
Because of the small metabolic pool of calcium (less than 0.1% in the ECF compartment) relative to the large skeletal reserve, for all practical purposes meta- bolic calcium deficiency probably never exists, at least not as a nutritional disorder. An inadequate intake or poor intestinal absorption of calcium causes the circulating ionized calcium concentration to decline acutely, which triggers an increase in PTH synthesis and release. PTH acts on three target organs (either directly or indirectly) to restore the circulating calcium
concentration to normal (Figure 9.2). At the kidney, PTH promotes the reabsorption of calcium in the distal tubule. PTH affects the intestine indirectly by stimulating the production of 1,25(OH)2D3 (in the kidney), which, in turn, leads to increased calcium absorption. PTH also induces bone resorption (by signaling osteoclasts), thereby releasing calcium into blood. Owing to the action of PTH and 1,25(OH)2D3 on the target tissues, plasma calcium concentrations are restored within minutes to hours.
If, however, there is a continual inadequate intake or poor intestinal absorption of calcium (e.g., because of vitamin D deficiency), circulating calcium concen-tration is maintained largely at the expense of skeletal mass, that is, from an increased rate of bone resorp-tion. This PTH-mediated increase in bone resorp-tion is one of several important causes of reduced bone mass and osteoporosis. The cumulative effect of calcium depletion (by whatever mechanism) on the skeleton over many years contributes to the increas-ing frequency of osteoporotic fractures with age. Prolonged inadequate calcium intake in younger people reduces the rate of accretion of the skeleton and may prevent the attainment of the genetically determined maximal PBM. This may increase the risk of osteoporosis as the PBM in adulthood is predictive of bone mass in later life. Chronic inadequate intake or poor intestinal absorption of calcium may also play some role in the etiologies of hypertension, including pre-eclampsia and colon cancer. Calcium intake may also play a role in body weight regulation; however, this requires further investigation.
Related Topics