Chapter: Introduction to Human Nutrition: Minerals and Trace Elements
Phosphorus: Absorption, transport, tissue distribution, Metabolic function, essentiality, Deficiency symptoms
Phosphorus
Phosphorus is never found free in nature, but is widely distributed in combination with minerals. Phosphate rock, which contains the mineral apatite, an impure tricalcium phosphate, is an important source of the element. Phosphorus is most commonly found in nature in its pentavalent form in combina-tion with oxygen as phosphate (PO43−). Phosphorus (as phosphate) is an essential constituent of all known protoplasm and is uniform across most plant and animal tissues. A practical consequence is that, as organisms consume other organisms lower in the food chain (whether animal or plant), they automati-cally obtain their phosphorus.
Absorption, transport, and tissue distribution
Phosphorus makes up about 0.65–1.1% of the adult body (~600 g). In the adult body 85% of phosphorus is in bone and the remaining 15% is distributed in soft tissues. Total phosphorus concentration in whole blood is 13 mmol/l, most of which is in the phospho-lipids of erythrocytes and plasma lipoproteins, with approximately 1 mmol/l present as inorganic phos-phate. This inorganic component, while constituting only a minute percentage of body phosphorus (<0.1%), is of critical importance. In adults, this com-ponent makes up about 15 mmol in total and is located mainly in the blood and ECF. It is into the inorganic compartment that phosphate is inserted on absorption from the diet and resorption from bone, and from this compartment that most urinary phosphorus and hydroxyapatite mineral phosphorus are derived (Figure 9.4). This compartment is also the primary source from which the cells of all tissues derive both structural and high-energy phosphate.
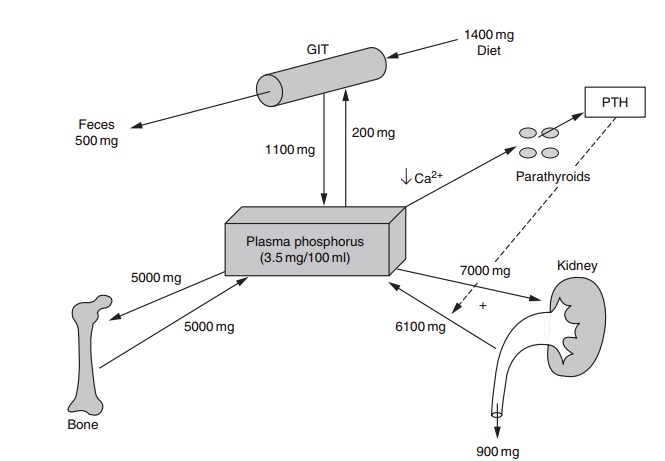
Figure 9.4 Homeostatic regulation of serum phosphorus. PTH, parathyroid hormone
Food phosphorus is a mixture of inorganic and organic forms. Intestinal phosphatases hydrolyze the organic forms contained in ingested protoplasm and, thus, most phosphorus absorption occurs as inor-ganic phosphate. On a mixed diet, absorption of total phosphorus ranges from 55% to 70% in adults. There is no evidence that this absorption varies with dietary intake. Furthermore, there appears to be no apparent adaptive mechanism that improves phosphorus absorption at low intakes. This situation is in sharp contrast to calcium, for which absorption efficiency increases as dietary intake decreases and for which adaptive mechanisms exist that improve absorption still further at habitual low intakes. While a portion of phosphorus absorption is by way of a saturable, active transport facilitated by 1,25(OH)2D3 the fact that fractional phosphorus absorption is virtually constant across a broad range of intakes suggests that the bulk of phosphorus absorption occurs by passive, concentration-dependent processes. Phosphorus absorption is reduced by ingestion of aluminum-containing antacids and by pharmacological doses of calcium carbonate. There is, however, no significant interference with phosphorus absorption by calcium at intakes within the typical adult range. Excretion of endogenous phosphorus is mainly through the kidneys. Inorganic serum phosphate is filtered at the glomerulus and reabsorbed in the proximal tubule. In the healthy adult, urine phosphorus is essentially equal to absorbed dietary phosphorus, minus small amounts of phosphorus lost in shed cells of skin and intestinal mucosa.
Metabolic function and essentiality
Structurally, phosphorus occurs as hydroxyapatite in calcified tissues and as phospholipids, which are a major component of most biological membranes, and as nucleotides and nucleic acid. Other functional roles of phosphorus include:
●buffering of acid or alkali excesses, hence helping to maintain normal pH
●the temporary storage and transfer of the energy derived from metabolic fuels
●by phosphorylation, and hence activation of many catalytic proteins.
As phosphorus is not irreversibly consumed in these processes and can be recycled indefinitely, the actual functions of dietary phosphorus are first to support tissue growth (either during individual development or through pregnancy and lactation), and second to replace excretory and dermal levels. In both processes, it is necessary to maintain a normal level of inorganic phosphate in the ECF, which would otherwise be depleted of phosphorus by growth and excretion.
Deficiency symptoms
Inadequate phosphorus intake is expressed as hypo-phosphatemia. Only limited quantities of phosphate are stored within cells, and most tissues depend on ECF inorganic phosphate for their metabolic phos-phate. When ECF inorganic phosphate levels are low, cellular dysfunction follows. At a whole organ-ism level, the effects of hypophosphatemia include anorexia, anemia, muscle weakness, bone pain, rickets and osteomalacia, general debility, increased suscep-tibility to infection, paresthesia, ataxia, confusion, and even death. The skeleton will exhibit either rickets in children or osteomalacia in adults. In both groups, the disorder consists of a failure to mineralize forming growth plate cartilage or bone matrix, together with impairment of chrondroblast and osteoblast func-tion. These severe manifestations are usually confined to situations in which ECF phosphate falls below approximately 0.3 mmol/l. Phosphorus is so ubiqui-tous in various foods that near total starvation is required to produce dietary phosphorus deficiency.
Related Topics