Chapter: Psychology: Development
Prenatal Development
PRENATAL DEVELOPMENT
The
story of how we grow and change across the life span is one of the most
interesting in all of psychology. But as with any story, we must begin at the
beginning, with a consideration of how genetic and environmental factors
interact to shape us before we are even born.
From Conception to Birth
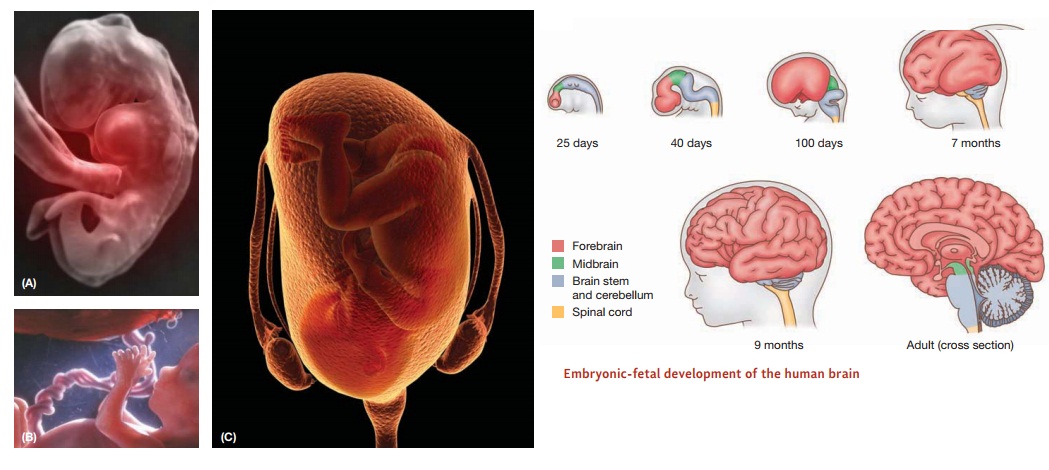
The
voyage toward becoming a human being begins at conception, when a sperm and egg
cell unite to form the fertilized egg or zygote
(Figure 14.1). Within hours of this union of sperm and egg, the nuclei of the
two cells merge, creating a novel combination of 23 pairs of chromosomes, half
from the mother and half from the father. The zygote begins a process of
dividing and redividing, producing a blastocyst, a mass of identical cells.
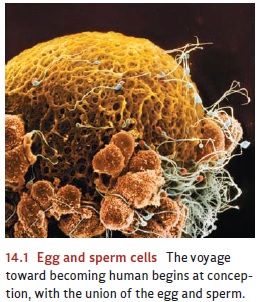
About
10 to 14 days after fertilization, the blastocyst attaches itself to the wall
of the uterus. Then, the embryonic stage
begins (Figure 14.2). During this phase, critical genes turn on and produce chemical
signals that induce a process of differentiation among the proliferating cells.
The mass of cells (now called an embryo)
soon has three distinct cell types—those that will become the nervous system
and the outer skin; those that will form the skeletal system and voluntary
muscles; and those that will form the gut and digestive organs.
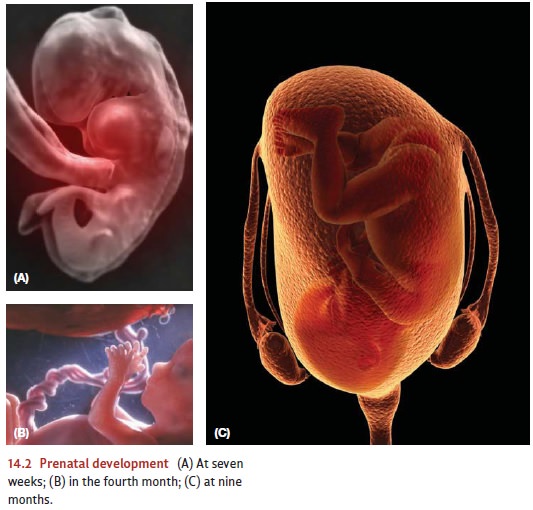
One
month after conception, the placenta and umbilical cord have developed, and the
embryo begins to develop the major organ systems of the body (including the
heart and lungs), as well as arms, legs, and facial features. At about this
same time, we can detect the beginnings of a nervous system: a structure called
a neural tube with three
identifiable subparts, one that will develop into the brain stem and spinal
cord, and two others that will develop into the midbrain and forebrain (Figure
14.3).
Two
months after conception, the fetal stage
begins. By this point, the mass of cells (now called a fetus) has grown to 1 inch in length and the heart has begun to
beat. The nervous system continues to grow at a remarkable pace. New nerve
cells are generated at a rate that can approach 250,000 new cells per minute
(Kolb & Whishaw, 2009; Mueller, 1996), and these cells start to form
themselves into a network. Several mecha-nisms contribute to this networking,
including specific genes that produce chemical sig-nals that serve as
“beacons,” attracting connections that sprout from other nerve cells.
Even
at this early stage, the fetus is capable of simple behaviors, and so will show
a sucking reflex if its lips are touched. It’s not long before other—more
sophisticated— capacities come into view, including the capacity for learning.
For example, in one now-classic study, DeCasper and Spence (1986) asked
pregnant mothers to read aloud to their unborn infants twice a day for the last
6 weeks of their pregnancy from one of two Dr. Seuss books. Once the children
were born, researchers set up an apparatus that was controlled by the way the
newborns sucked on a special pacifier; if they sucked in one way, the apparatus
played the story their mothers had read before they were born; if they sucked
in another way, the apparatus played an unfamiliar story. The researchers found
that infants adjusted their sucking pattern so that they could listen to the
story to which they had been exposed in utero, indicating that the infants had
“learned” one story and preferred it to the story they did not know.
The Prenatal Environment
A
great deal of prenatal development is powerfully guided by the genome.
Environmental factors are just as important, however, as we have seen in the
capacity of the fetus to learn from its experiences. But what in general does
“environment” mean in this early stage of development?
Consider
the earliest stages of embryonic growth. Some of the cells in the embryo will
eventually become the brain; others will become the gall bladder or the bones
of the foot. But every cell in the embryo has the same genes, and so presumably
all receive the same genetic instructions. How does each cell manage to develop
appropriately?
The
answer seems to be that the fate of each cell is determined in part by its
cellular neighbors—the cells that form its physical environment. Evidence comes
from studies of salamander embryos. Early in their development, salamanders
have an outer layer of tissue that gradually differentiates, and cells in this
layer will become teeth only if they make contact with certain other cells in
the embryo’s mouth region. Without this contact, cells in this layer become
skin.
In
humans, the cells that will become the brain initially show no distinction
between neurons and glia. As the cells reproduce and differentiate, though, these
two types become distinct, and the newly created neurons actually migrate
toward their appropriate posi-tions. This migration process is guided by glia
that act as guidewires. Various chemicals also guide the process by attracting
some types of nerve cells and repelling other types (Hatten, 2002). In all
cases, the migrating neurons approach the surface of the develop-ing cortex,
but the first-arriving neurons stop short of the surface. Later-arriving
neurons pass these now-stationary cells, and these late arrivals in turn are
passed by even later arrivals. As a result, the cortex literally develops from
the inside out, with layers closer to the surface established later than deeper
layers.
Of
course, it’s not enough that the nerve cells end up in the right places; they
also need to end up connected in the right way, so that each nerve cell sends
its messages to the right target. How does each developing nerve cell come to
know its eventual target? The answer, of course, begins with the genes. Early in
development, genetic specifica- tions lead neurons to form protomaps, providing a rough “wiring diagram” for the brain’s
circuits. The areas mapped in this way seem to attract connections from the
appropriate inputs, so that, for example, the protomap in the projection area
for vision attracts afferent fibers from the thalamus, with the result that the
visual cortex comes to receive the right input signals (e.g., Rakic, 1995; for
some complexities, though, see Sur & Rubenstein, 2005).
Inevitably,
there are some wiring errors, but there is a safeguard in place to deal with
these. Many more neurons are created than are needed, and each neuron tries to
form far more connections than are required. If a neuron’s connections prove
either wrong or redundant, that neuron can withdraw its connections and find
better targets, or it can be given a message to die (Kuan, Roth, Flavell, &
Rakic, 2000; Rubenstein & Rakic, 1999). In fact, it is normal for between
20 and 80% of neurons to die as the brain develops, depending upon the region
of the brain. This decimation primarily occurs early in development—in humans,
about 4 to 6 months after conception (Rosenzweig, Leiman, & Breedlove,
1996)—but according to some investigators, it continues at a slower rate for
much longer, perhaps even a decade.
So
far, we have focused on how the local environment surrounding each neuron
guides its differentiation and migration. More global features of the
environment also play a major role, namely, the organism’s own bodily fluids,
especially its blood. Thus, for example, hormones circulating in the fetus’s
blood have a powerful influence on the development of the child’s external
anatomy, the development of the nervous system, and even later sex-typical play
(Auyeung et al., 2009). Moreover, the bloodstream of mammalian embryos is
intimately connected to the mother’s blood supply, and so her blood, too,
becomes part of the embryo’s environment.
The
maternal blood supplies oxygen and nutrition to the developing fetus, and this
is one of the reasons why normal development depends on the nutritional state
of the mother. But the mother’s blood supply also plays another role—it
provides a conduit through which factors in the external environment can
influence the fetus. Unfortunately, many of these external factors are teratogens—factors that can disrupt
development. The long list of teratogens includes environmental toxins such as
lead and mercury, as well as alcohol, cigarette smoke, X-rays, and diseases
such as rubella (German measles). Teratogens can have a number of negative
effects, depending on the type, timing, and amount of exposure. For example,
when a pregnant woman drinks alcohol, the alcohol enters both her bloodstream
and that of her fetus. Even light drink-ing can affect the brain of the
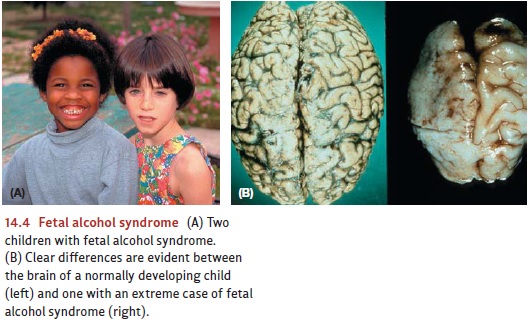
developing fetus (Ikonomidou et al., 2000), and heavy drinking can lead to fetal alcohol syndrome, which is
characterized by both psycholog-ical problems (learning disorders and behavior
difficulties) and physical abnormalities (smaller stature and a characteristic
pattern of facial abnormalities; Figure 14.4).
Related Topics