Chapter: 11th 12th std standard Class Organic Inorganic Physical Chemistry Higher secondary school College Notes
Imperfections In Solids
IMPERFECTIONS IN SOLIDS
Almost all the crystals encountered in practice suffer
from imperfections or defects of various
kinds. An ideally perfect crystal is one which has the same unit cell and contains the same lattice points throughout the
crystal. The term imperfection or
defect is generally used to describe any deviation of the ideally perfect crystal from the periodic arrangement of its
constituents.
Point
Defects
If the deviation occurs due to missing atoms, displaced
atoms or extra atoms, the imperfection
is named as a point defect. Such defects arise due to imperfect packing during the original crystallisation or they may
arise from thermal vibrations of atoms at
elevated temperatures. The most common point defects are the Schottky defect and Frenkel defect. Comparatively less
common point defects are the metal
excess defect and the metal deficiency defect.
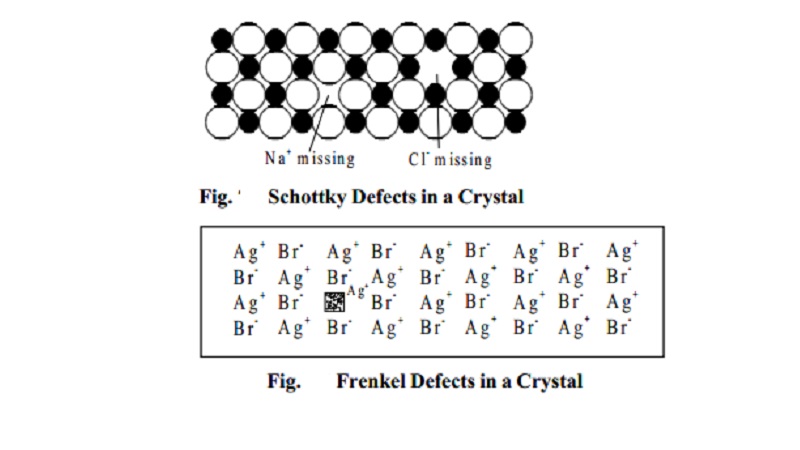
Schottky defects
This defect is caused if some of the lattice points are
unoccupied. The points which are
unoccupied are called lattice vacancies. The number of missing positive and negative ions is the same in this case and thus, the
crystal remains neutral. The existence of
two vacancies, one due to a missing Na+ ion and the other due to a missing Cl- ion in a crystal of NaCl is shown in Fig.
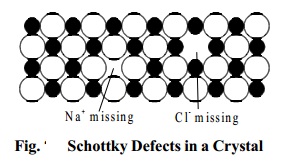
Schottky defects appears generally in ionic crystals in
which the positive and negative ions do
not differ much in size.
Frenkel defects
This defect arise when an ion occupies an interstitial
position between the lattice points. This defect occurs generally in ionic
crystals in which the anion is much larger in
size than the cation. AgBr is an example for this type of defect. One of the Ag+ ion
occupies a position in the interstitial space rather than its own appropriate site in the lattice is shown in Fig.

The crystal remains neutral since the number of positive
ions is the same as the number of
negative ions.
Metal excess defects
If a crystal of NaCl is heated in sodium vapour, it
acquires a yellow colour. This yellow colour is due to the formation of a
non-stoichiometric compound of NaCl in which
there is a slight excess of sodium ions. This defect is called the metal excess defect.
Metal deficiency defects
In certain cases, one of the positive ions is missing
from its lattice site and the extra
negative charge is balanced by some nearby metal ion acquiring additional charges instead of original charge. This type of defect
is generally found in compounds of
transition metals which can exhibit variable valency. FeO and FeS show this type of defects.
Related Topics