Chapter: 11th 12th std standard Class Organic Inorganic Physical Chemistry Higher secondary school College Notes
Electron affinity
Electron affinity
In modern terminology,
electron affinity is known as the electron gain enthalpy. Electron gain
enthalpy is the amount of energy released when an isolated gaseous atom accepts
an electron to form a monovalent gaseous anion.
Atom (g) +
Electron -> Anion (g)
+ energy
Example,
Cl (g) + e--> Cl-
(g) + EA
If an atom
has high tendency to accept an electron, large energy will be released.
Consequently, electron gain enthalpy will be high. On the other hand if an atom
has less tendency to accept the electron small amount of energy will be
released, leading to small value of electron gain enthalpy. The values of
electron gain enthalpy are expressed either in electron volt per atom or kilo
joules per mole of atoms. For example, electron gain enthalpy of
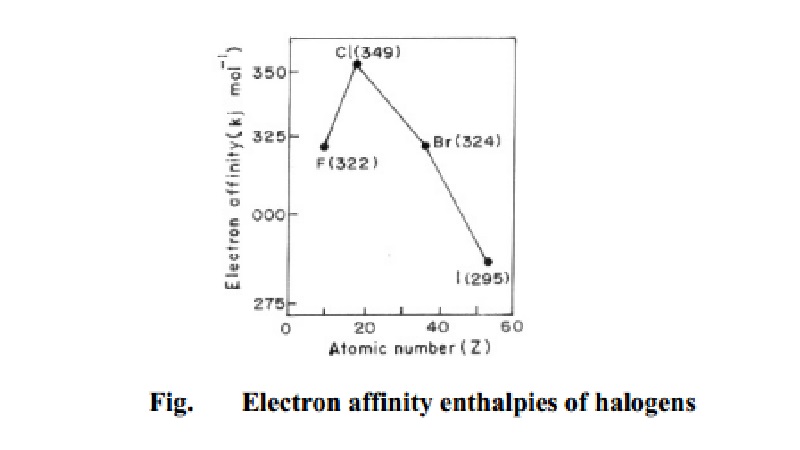
F
= 322 kJ mol-1
Cl = 349
kJ mol-1
Br = 324
kJ mol-1 and
I =295 kJ mol-1
Halogens (elements of group 17) can take up an
electron to acquire the stable noble gas configuration. Their values for
electron gain enthalpy are thus very high. Electron gain enthalpy values for
the halogens are as in Fig. 4.5.
Electron
gain enthalpies generally decrease on moving down the group. This is expected
on account of the increase in size of atoms, the effective nuclear attraction
for electrons decreases. As a result, there is less tendency to attract
additional electrons with an increase in atomic number down the group.
From the electron gain enthalpy data of halogens it is clear that,
contrary to expectation, the electron gain enthalpy of fluorine is lower than
that of chlorine. This is because the fluorine atom has a very compact
electronic shell due to its small size. The compactness of the fluorine shell
results in electron repulsion whenever an electron is introduced into its
2p-orbital. This is why its electron gain enthalpy is less than the expected
value. In Cl atom, 3p-orbitals are not as compact as the 2p-orbitals in
fluorine atom. The incoming electron is more readily accepted by the chlorine
atom because of weaker electron-electron repulsion. The electron gain enthalpy
of chlorine is, therefore, higher than that of fluorine.
In the case of noble gases, the outer s-and
p-orbitals are completely filled. No more electrons can be accommodated in
these orbitals. Noble gases, therefore, show no tendency to accept
electrons. Their electron gain
enthalpies are zero.
Electron gain enthalpies g The second electron gain enthalpy refers to a process in which the electron
is added to a negative ion. For example:
O- (g) + e- -> O2- (g)
Since a negative ion O- and an electron repel each other,
energy is required and not released by the process. Therefore the second
electron gain enthalpy is negative in this case.
Factors influencing the magnitude of electron
affinity
The magnitude of EA is influenced by a number of factors such as
(i) Atomic size; (ii) Effective nuclear charge; and (iii) Screening effect by inner electrons.
Example
Which of the following will have the most negative electron gain
enthalpy and which has the least negative? P, S, Cl, F.
Explain
your answer.
Solution
Electron gain enthalpy generally becomes more negative across a period
as we move from left to right. Within a group, electron gain enthalpy becomes
less negative down a group. However, adding an electron to the 2p orbital leads
to greater repulsion than adding an electron to the larger 3p orbital. Hence
the element with most negative electron gain enthalpy is chlorine; the one with
the least negative electron gain enthalpy is phosphorus.
enerally
increase as we move across a period from left to right. This is due to the
increase in the nuclear charge, which results in greater attraction for
electrons.
Related Topics