Chapter: Essentials of Anatomy and Physiology: Nervous System
Electrical Signals and Neural Pathways
ELECTRICAL SIGNALS AND NEURAL PATHWAYS
A. Describe a resting membrane potential, and explain how an action potential is generated and propagated. Compare the roles of leak and gated ion channels.
B. Describe the structure and function of a synapse.
C. List the parts of a reflex arc, and describe its function.
D. Describe a converging and a diverging circuit and the role of summation in neural pathways.
Resting Membrane Potential
All cells exhibit electrical properties. The inside of most cell mem-branes is negatively charged compared to the outside of the cell mem-brane, which is positively charged. This uneven distribution of charge means the cell membrane is polarized. In an unstimulated (or resting) cell, the uneven charge distribution is called the resting membrane potential. The outside of the cell membrane can be thought of as the positive pole of a battery and the inside as the negative pole. Thus, a small voltage difference, called a potential, can be measured across the resting cell membrane.
The resting membrane potential is generated by three main fac-tors: (1) a higher concentration of K+ immediately inside the cell membrane, (2) a higher concentration of Na+ immediately outside the cell membrane, and (3) greater permeability of the cell membrane to K+ than to Na+ (figure 8.7, step 1). Thus, the resting membrane potential results from differences in the concentration of ions across the membrane and the permeability characteristics of the membrane.
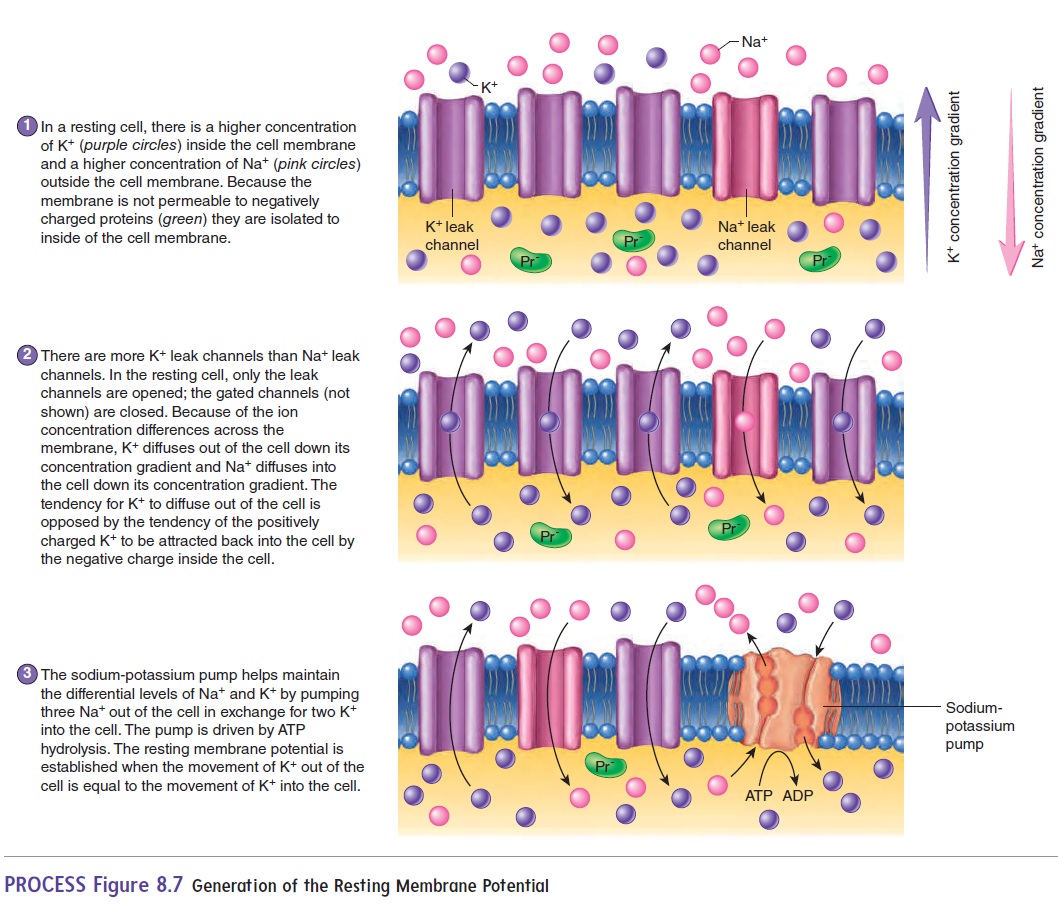
The difference in membrane permeability is due to the differ-ence in the number of open ion channels. Recall that ions cannot move freely across the cell membrane; instead, ions must flow through ion channels, which are proteins embed-ded in the cell membrane. Ions flow through channels due to the differences in their concentration across the membrane. There are two basic types of ion channels: leak channels and gated channels (see figure 3.5). Leak channels are always open. Thus, as the name suggests, ions can “leak” across the membrane down their concentration gradient. When a cell is at rest, the membrane potential is established by diffusion of ions through leak channels.

Because there are 50–100 times more K+ leak channels than Na+ leak channels, the resting membrane has much greater perme-ability to K+ than to Na+; therefore, the K+ leak channels have the greatest contribution to the resting membrane potential. Gatedchannels are closed until opened by specific signals. Chemically gated channels are opened by neurotransmitters or other chemi-cals, whereas voltage-gated channels are opened by a change in membrane potential. When opened, the gated channels can change the membrane potential and are thus responsible for the action potential, described next.
As mentioned earlier, there is a net negative charge inside and a net positive charge outside a resting cell. The primary source of negative charge inside the cell is a high concentration of negatively charged molecules, such as proteins, that cannot diffuse out of the cell because the cell membrane is impermeable to them (figure 8.7, step 2). Consequently, as positively charged K+ leaks out of the cell via the leak channels, the charge inside the cell membrane becomes even more negative. The negatively charged molecules inside the cell tend to attract the positive K+ back into the cell. The resting membrane potential is the point of equilibrium at which the tendency for K+ to move down its concentration gradient out of the cell is balanced by the negative charge within the cell, which tends to attract the K+ back into the cell (figure 8.7, step 2).
To compensate for the constant leakage of ions across the membrane, the sodium-potassium pump (Na+–K+ pump) is required to maintain the greater concentration of Na+ outside the cell mem-brane and K+inside. The pump actively transports K+ into the cell and Na+ out of the cell (figure 8.7, step 3). The importance of this pump is indicated by the astounding amount of energy it consumes. It is estimated that the sodium-potassium pump consumes 25% of all the ATP in a typical cell and 70% of the ATP in a neuron.
Action Potentials
Muscle and nerve cells are excitable cells, meaning that the resting membrane potential changes in response to stimuli that activate gated ion channels. The opening and closing of gated channels can change the permeability characteristics of the cell membrane and hence change the membrane potential.
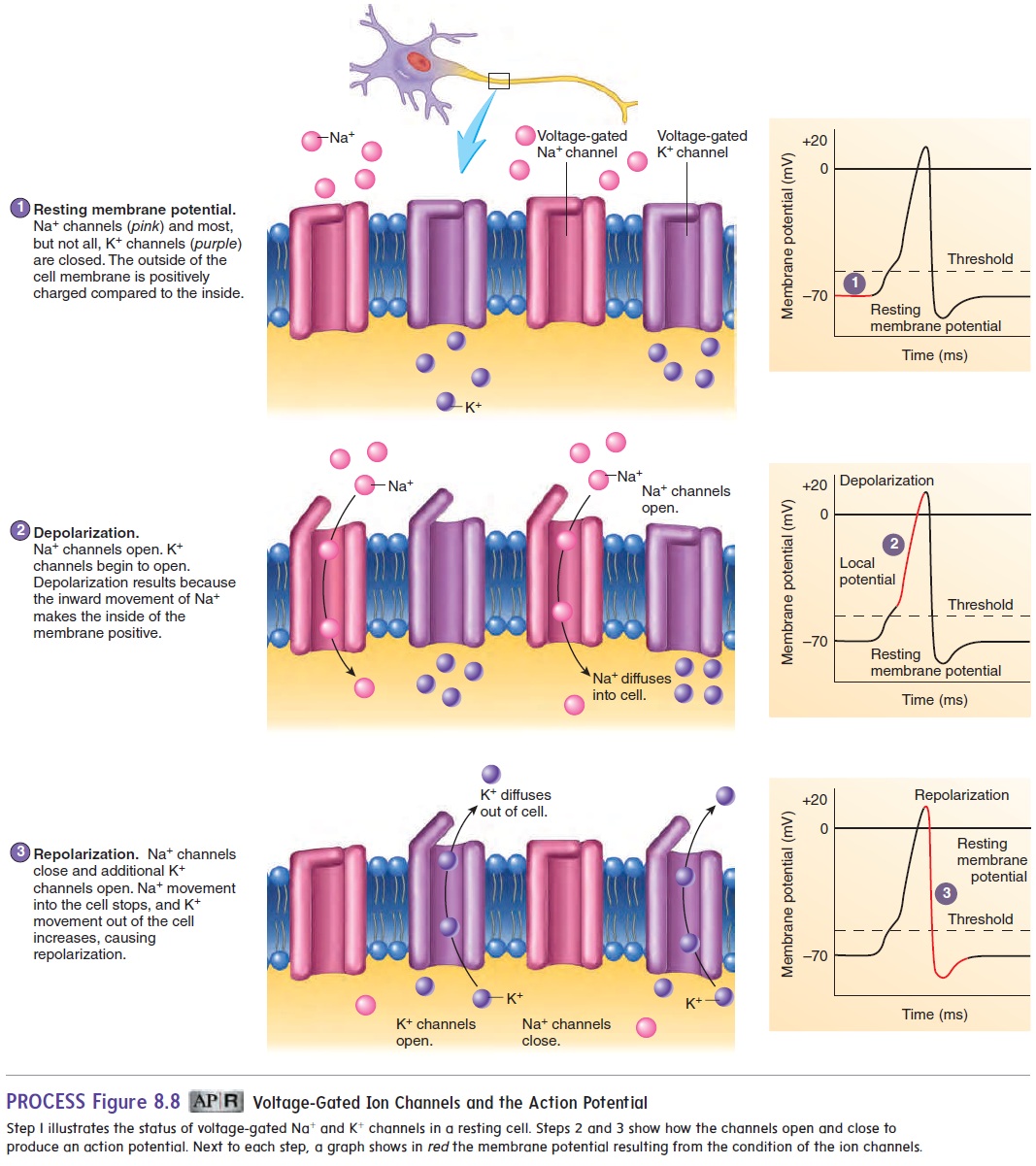
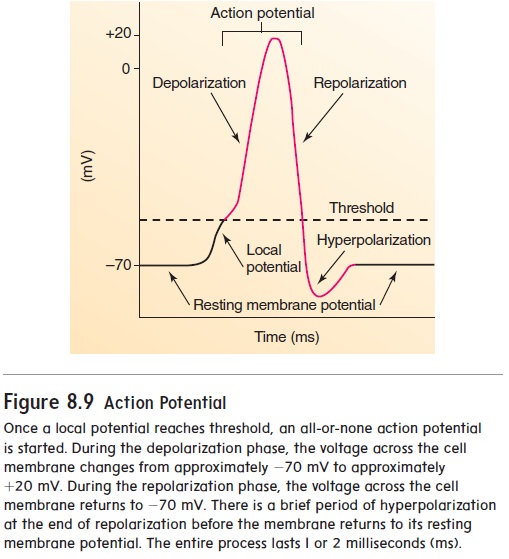
The channels responsible for the action potential are voltage-gated Na+ and K+ channels. When the cell membrane is at rest, the voltage-gated channels are closed (figure 8.8, step 1). When a stimulus is applied to a muscle cell or nerve cell, following neurotransmitter activation of chemically gated channels, Na+ channels open very briefly, and Na+ diffuses quickly into the cell (figure 8.8, step 2). This movement of Na+, which is called a localcurrent, causes the inside of the cell membrane to become posi-tive, a change called depolarization. This depolarization results in a local potential. If depolarization is not strong enough, the Na+ channels close again, and the local potential disappears without being conducted along the nerve cell membrane. If depolariza-tion is large enough, Na+ enters the cell so that the local potential reaches a threshold value. This threshold depolarization causes voltage-gated Na+ channels to open. Threshold is most often reached at the axon hillock, near the cell body. The opening of these channels causes a massive, 600-fold increase in membrane permeability to Na+. Voltage-gated K+ channels also begin to open. As more Na+ enters the cell, depolarization occurs until a brief reversal of charge takes place across the membrane—the inside of the cell membrane becomes positive relative to the outside of the cell membrane. The charge reversal causes Na+ channels to close and more K+ channels to open. Na+ then stops entering the cell, and K+ leaves the cell (figure 8.8, step 3). This repolarizes the cell membrane to its resting membrane potential. Depolarization and repolarization constitute an action potential (figure 8.9). At the end of repolarization, the charge on the cell membrane briefly becomes more negative than the resting mem-brane potential; this condition is called hyperpolarization. The elevated permeability to K+ lasts only a very short time.
In summary, the resting membrane potential is set by the activity of the leak channels. On stimulation, chemically gated channels are opened and initiate localpotentials. If sufficiently strong, the local potentials activate voltage-gated channels to initi-ate an action potential.
Action potentials occur in an all-or-none fashion. That is, if threshold is reached, an action potential occurs; if the threshold is not reached, no action potential occurs. Action potentials in a cell are all of the same magnitude—in other words, the amount of charge reversal is always the same. Stronger stimuli produce a greater frequency of action potentials but do not increase the size of each action potential. Thus, neural signaling is based on the number of action potentials.
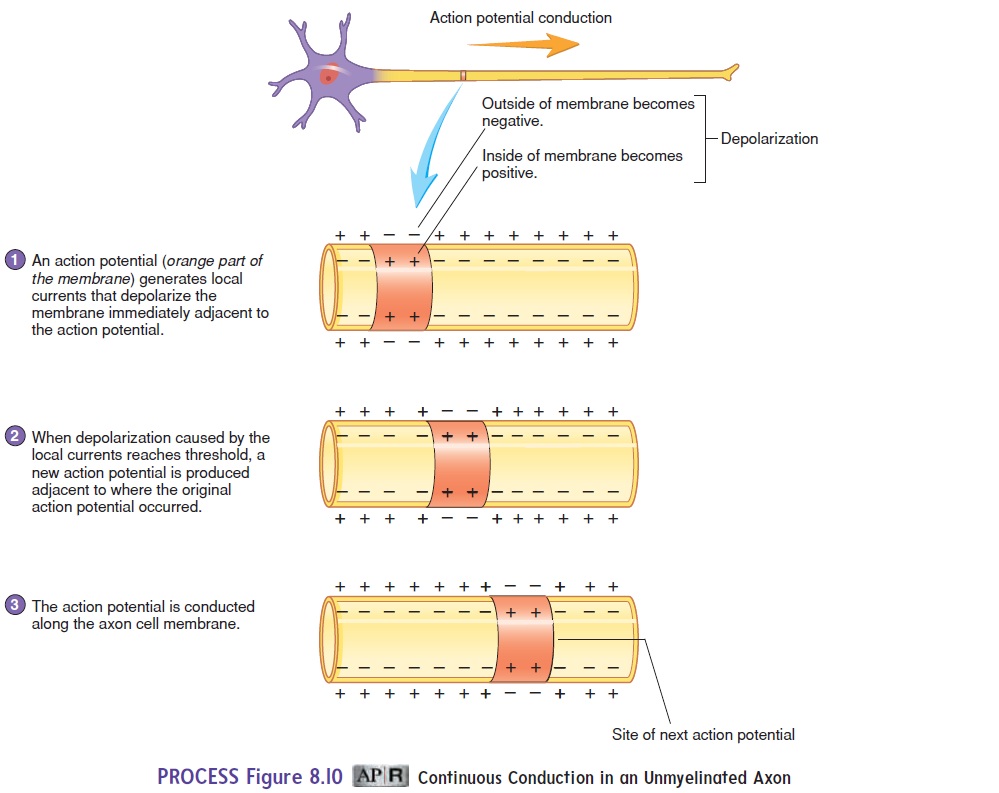
Action potentials are conducted slowly in unmyelinated axons and more rapidly in myelinated axons. In unmyelinated axons, an action potential in one part of a cell membrane stimulates local currents in adjacent parts of the cell membrane. The local cur-rents in the adjacent membrane produce an action potential. By this means, the action potential is conducted along the entire axon cell membrane. This type of action potential conduction is called continuous conduction (figure 8.10).
In myelinated axons, an action potential at one node of Ranvier causes a local current to flow through the surrounding extracellular fluid and through the cytoplasm of the axon to the next node, stimulat-ing an action potential at that node of Ranvier. By this means, action potentials “jump” from one node of Ranvier to the next along the length of the axon. This type of action potential conduction is calledsaltatory (sal′ tă-tōr-ē; to leap) conduction (figure 8.11).
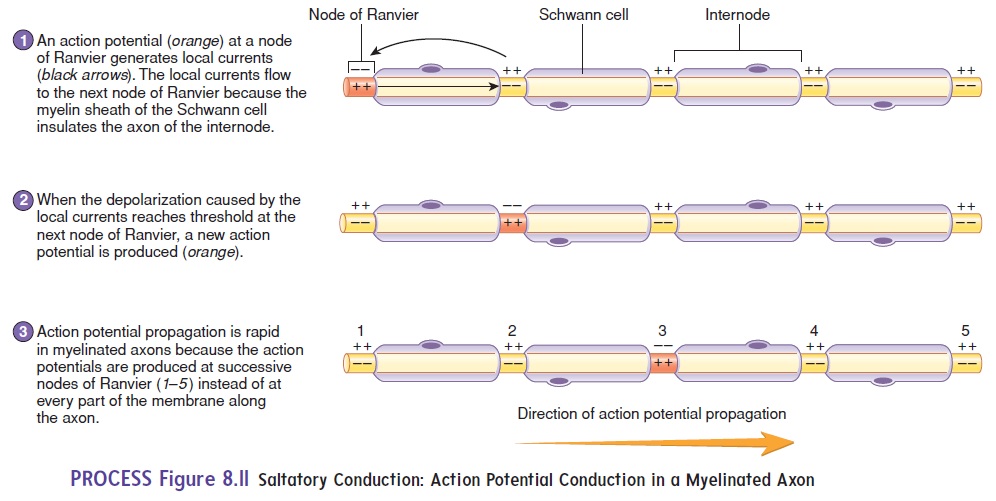
Saltatory conduction greatly increases the conduction velocity because the nodes of Ranvier make it unnecessary for action potentials to travel along the entire cell membrane. Action potential conduc-tion in a myelinated fiber is like a child skipping across the floor, whereas in an unmyelinated axon it is like a child walking heel to toe across the floor.
Medium-diameter, lightly myelinated axons, characteristic of autonomic neurons, conduct action potentials at the rate of about 3–15 meters per second (m/s), whereas large-diameter, heavily myelinated axons conduct action potentials at the rate of 15–120 m/s. These rapidly conducted action potentials, carried by sensory and motor neurons, allow for rapid responses to changes in the external environment. In addition, several hundred times fewer ions cross the cell membrane during conduction in myelinated cells than in unmyelinated cells. Much less energy is therefore required for the sodium-potassium pump to maintain the ion distribution.
The Synapse
A synapse (sin′ aps) is a junction where the axon of one neuron interacts with another neuron or with cells of an effector organ, such as a muscle or gland (figure 8.12). The end of the axon forms a pre-synaptic terminal. The membrane of the dendrite or effector cell isthe postsynaptic membrane, and the space separating the presyn-aptic and postsynaptic membranes is the synaptic cleft. Chemical substances called neurotransmitters (noor′ ō-trans-mit′ erz; neuro-, nerve + transmitto, to send across) are stored in synaptic vesicles in the presynaptic terminal. When an action potential reaches the presynaptic terminal, voltage-gated Ca2+ channels open, and Ca2+ moves into the cell. This influx of Ca2+ causes the release of neurotransmitters by exocytosis from the presynaptic terminal. The neurotransmitters diffuse across the synaptic cleft and bind to specific receptor molecules on the postsynaptic membrane. The binding of neurotransmitters to these membrane receptors causes chemically gated channels for Na+, K+, or Cl− to open or close in the postsynaptic membrane, depending on the type of neurotrans-mitter in the presynaptic terminal and the type of receptors on the postsynaptic membrane. The response may be either stimulation or inhibition of an action potential in the postsynaptic cell. For example, if Na+ channels open, the postsynaptic cell becomes depo-larized, and an action potential will result if threshold is reached. If K+ or Cl− channels open, the inside of the postsynaptic cell tends to become more negative, or hyperpolarized (hı̄′ per-pō′ lăr-ı̄-zed), and an action potential is inhibited from occurring.
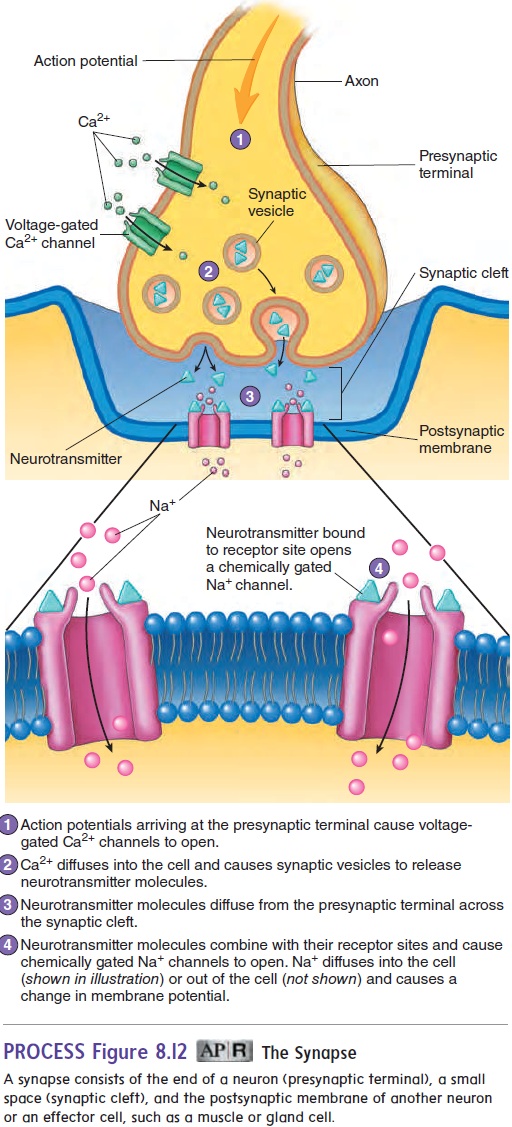
Of the many neurotransmitters or suspected neurotransmitter substances, the best known are acetylcholine (ACh) (as′ e-til-kō′ lēn) and norepinephrine (nōr′ ep-i-nef′ rin). Other neurotrans-mitters include serotonin (sēr- ō-tō′ nin), dopamine (dō′ pă-mēn), γ (gamma)-aminobutyric (gam′ă ă-mē′nō-bū-tēr′ik) acid (GABA),glycine, and endorphins (en′ dōr-finz; endogenous morphine) (table 8.2). Neurotransmitter substances are rapidly broken down by enzymes within the synaptic cleft or are transported back into the presynaptic terminal. Consequently, they are removed from the synaptic cleft, so their effects on the postsynaptic membrane are very short-term. In synapses where acetylcholine is the neurotrans-mitter, such as in the neuromuscular junction , an enzyme called acetylcholinesterase (as′ e-til-kō-lin-es′ - ter-ās) breaks down the acetylcholine. The breakdown products are then returned to the presynaptic terminal for reuse. Norepinephrine is
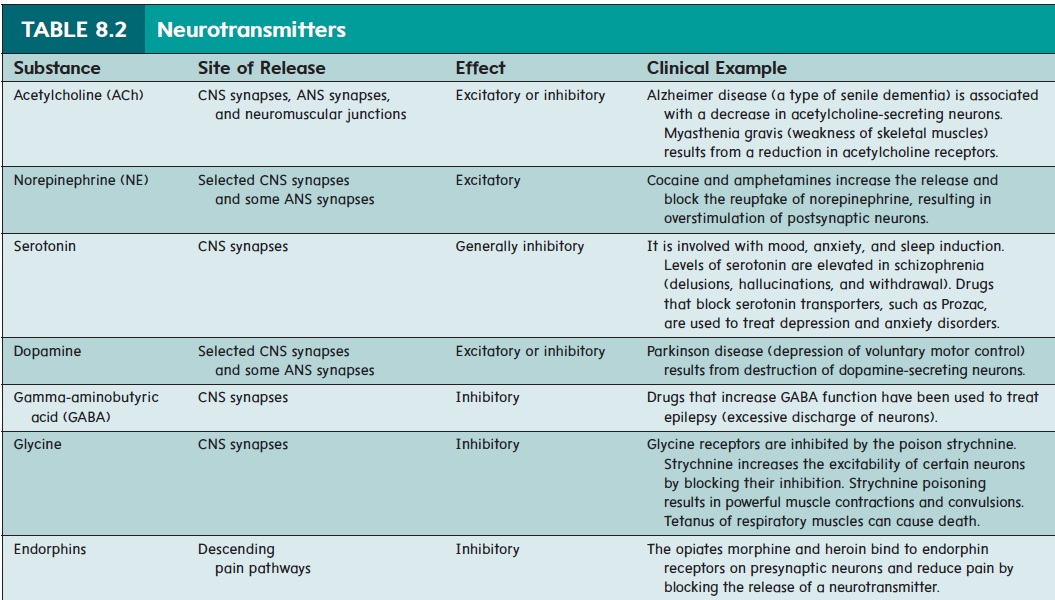
either actively transported back into the presynaptic terminal or broken down by enzymes. The release and breakdown or removal of neurotransmitters occurs so rapidly that a postsynaptic cell can be stimulated many times a second. Drugs can modulate the action of neurotransmitters at the synapse. Cocaine and amphetamines increase the release and block the reuptake of norepinephrine, resulting in overstimulation of postsynaptic neurons and deleteri-ous effects on the body. Drugs that block serotonin reuptake are particularly effective at treating depression and behavioral disorders.
Reflexes
A reflex is an involuntary reaction in response to a stimulus applied to the periphery and transmitted to the CNS. Reflexes allow a person to react to stimuli more quickly than is possible if conscious thought is involved. A reflex arc is the neuronal pathway by which a reflex occurs (figure 8.13). The reflex arc is the basic functional unit of the nervous system because it is the smallest, simplest pathway capable of receiving a stimulus and yielding a response. A reflex arc generally has five basic components:(1) a sensory receptor; (2) a sensory neuron; (3) in some reflexes, interneurons, which are neurons located between and communi-cating with two other neurons; (4) a motor neuron; and (5) an effector organ (muscles or glands). The simplest reflex arcs donot involve interneurons. Most reflexes occur in the spinal cord or brainstem rather than in the higher brain centers.
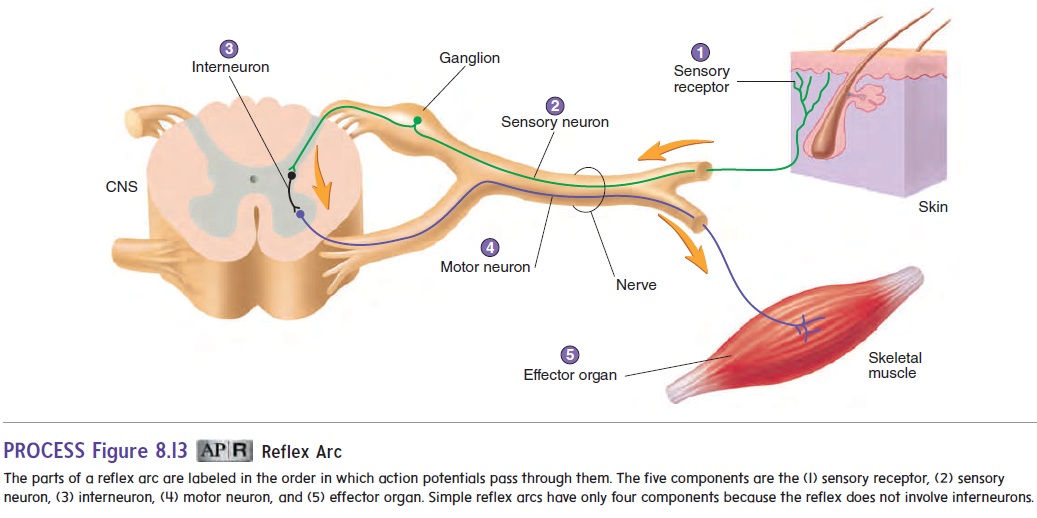
One example of a reflex occurs when a person’s finger touches a hot stove. The heat stimulates pain receptors in the skin, and action potentials are produced. Sensory neurons conduct the action potentials to the spinal cord, where they synapse with interneurons. The interneurons, in turn, synapse with motor neurons in the spi-nal cord that conduct action potentials along their axons to flexor muscles in the upper limb. These muscles contract and pull the finger away from the stove. No conscious thought is required for this reflex, and withdrawal of the finger from the stimulus begins before the person is consciously aware of any pain.
Neuronal Pathways
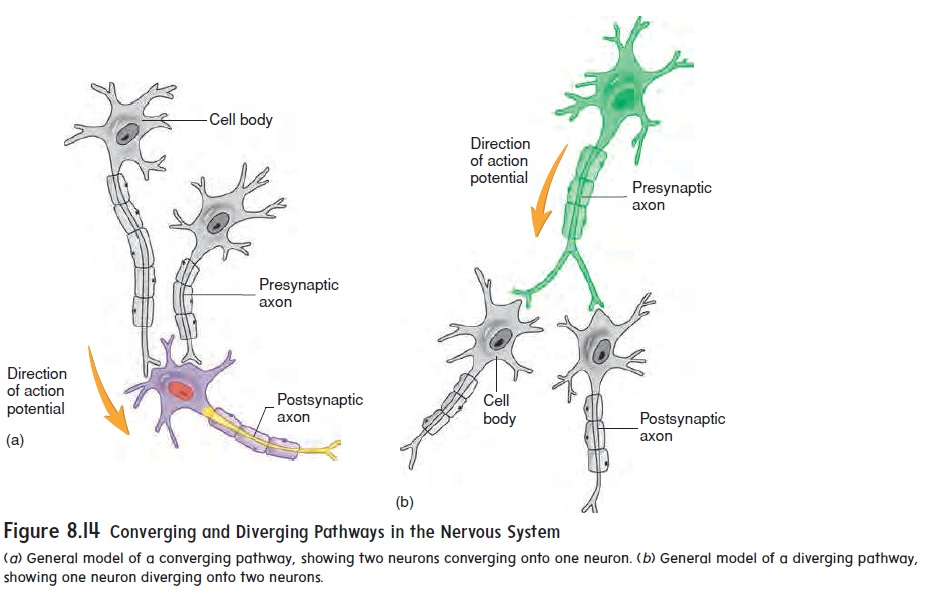
Neurons are organized within the CNS to form pathways ranging from relatively simple to extremely complex. The two simplest pathways are converging and diverging pathways. In a convergingpathway,two or more neurons synapse with (converge on) the sameneuron (figure 8.14a). This allows information transmitted in more than one neuronal pathway to converge into a single pathway. In a diverging pathway, the axon from one neuron divides (diverges) and synapses with more than one other neuron (figure 8.14b). This allows information transmitted in one neuronal pathway to diverge into two or more pathways.
Within the CNS and in many PNS synapses, it takes more than a single action potential to have an effect. A single presyn-aptic action potential usually does not cause a sufficiently large postsynaptic local potential to reach threshold and produce an action potential in the target cell. Instead, many presynaptic action potentials are needed in a process called summation. Summation of signals in neuronal pathways allows integration of multiple sub-threshold local potentials. Summation of the local potentials can bring the membrane potential to threshold and trigger an action potential. Spatial summation occurs when the local potentials originate from different locations on the postsynaptic neuron— for example, from converging pathways. Temporal summation occurs when local potentials overlap in time. This can occur from a single input that fires rapidly, which allows the resulting local potentials to overlap briefly. Spatial and temporal summation can lead to stimulation or inhibition, depending on the type of signal. Collectively, this integration of multiple inputs determines whether the postsynaptic neuron will fire an action potential.
Related Topics