Chapter: Biochemical Pharmacology : Drugs that act on sodium and potassium channels
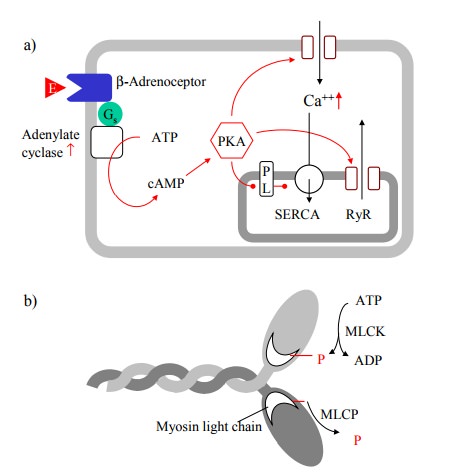
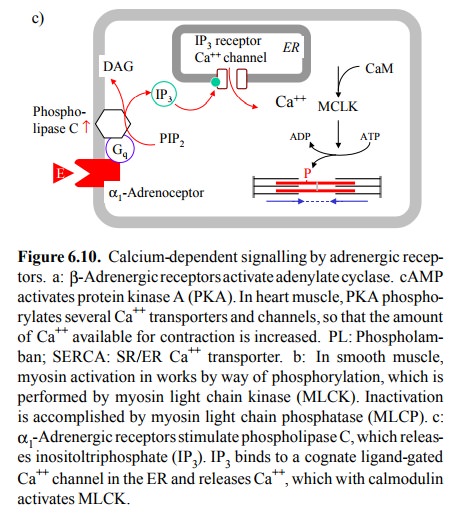
Calcium-dependent signaling by adrenergic receptors
Calcium-dependent signaling
by adrenergic receptors
Calcium is also involved in
the cellular effects of media-tors such as epinephrine and norepinephrine. In
the heart, the predominant adrenergic receptors are of the type β1, and
both agonists and antagonists of β receptors are being used in cardiac therapy. β-Adrenoceptors always activate adenylate cyclase and, through cAMP,
protein kinase A (PKA). However, the downstream effectors may differ de-pending
on the cell type. In the heart, PKA changes the ac-tivity of several target
proteins including RyR, DHPR, and the regulatory ER membrane protein
phospholamban (Fig-ure 6.10a). While the effect of phospholamban
phosphory-lation is a disinhibition of SERCA (SR/ER calcium trans-porter, an
ATP-dependent uniporter) that will tend to reduce cytosolic Ca++,
the first two will increase the availability of Ca++, which seems to
be the net effect.
In smooth muscle, contraction
is slower and longer lasting than in striated muscle. Regulation of actin and
myosin does not work by way of troponin / tropomyosin but by phosphorylation of
the regulatory myosin light chain (Fig-ure 6.10b). This is catalyzed by myosin
light chain kinase (MLCK), which is calmodulin-dependent and, hence, again
under the control of calcium. However, less calcium is nec essary in this
regulatory mechanism, because MLCK pro-vides an extra amplification stage not
present in the direct binding of calcium to troponin.
In smooth muscle, β-adrenoceptors decrease
contractility: PKA phosphorylates MLCK, which thereby becomes in-activated. In
contrast, α1-adrenoceptors
increase smooth muscle contractility.
They activate phospholipase C, which in turn releases inositoltriphosphate (IP3)
from the endo-plasmic reticulum by binding to a cognate receptor chan-nel
(Figure 6.10c). Ca++ then binds to calmodulin, which in turn
activates myosin light chain kinase.
Related Topics