Chapter: Biochemistry: Lipid Metabolism
Acetyl-CoA Carboxylase-A New Target in the Fight against Obesity?
Acetyl-CoA
Carboxylase—A New Target in the Fight against Obesity?
Malonyl-CoA has two very important functions in metabolism. First,
it is the committed intermediate in fatty-acid synthesis. Second, it strongly
inhibits carnitine palmitoyltransferase I and therefore fatty-acid oxidation.
The level of malonyl-CoA in the cytosol can determine whether the cell will be
oxidizing fats or storing fats. The enzyme that produces malonyl-CoA is
acetyl-CoA carboxylase, or ACC. There are two isoforms of this enzyme encoded
by separate genes. ACC1 is found in the liver and adipose tissue, while ACC2 is
found in cardiac and skeletal muscle. High glucose concentrations and high
insulin concentrations lead to stimulation of ACC2. Exercise has the opposite
effect. During exercise, an AMP-dependent protein kinase phosphorylates ACC2
and inactivates it.
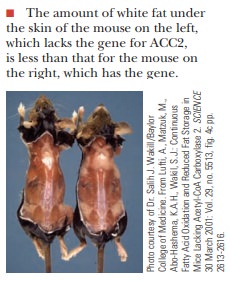
Some recent studies looked at the nature of weight gain and weight
loss with respect to ACC2. The investigators created a strain of mice lack-ing
the gene for ACC2. These mice ate more than their wild-type counterparts but
had significantly lower stores of lipids (30%– 40% less in skeletal muscle and
10% less in cardiac muscle). Even the adipose tissue, which still had ACC1,
showed a reduction in stored triacylglycerols of up to 50%. The mice showed no
other abnormalities. They grew and reproduced normally and had nor-mal life
spans. The investigators concluded that reduced pools of malonyl-CoA due to the
lack of ACC2 results in increased β-oxidation via removal of the block on
carnitine palmitoyltransfer-ase I, and a decrease in fatty-acid synthesis. They
speculate that ACC2 would be a good target for drugs used to combat obesity.
Related Topics