Chapter: Introduction to Human Nutrition: The Vitamins
Vitamin B12 - Human Nutrition
Vitamin B12
Dietary deficiency of vitamin B12 occurs only in strict vegans, since the vitamin is found almost exclusively in animal foods. However, functional deficiency (per-nicious anemia, with spinal cord degeneration) as a result of impaired absorption is relatively common, especially in older people with atrophic gastritis.
Structure and vitamers
The structure of vitamin B12 is shown in Figure 8.13. The term corrinoid is used as a generic descriptor for cobalt-containing compounds of this general struc-ture that, depending on the substituents in the pyrrole rings, may or may not have vitamin activity. The term “vitamin B12” is used as a generic descriptor for the cobalamins, that is, those corrinoids having the bio-logical activity of the vitamin. Some of the corrinoids that are growth factors for microorganisms not only have no vitamin B12 activity, but may be antimetabo-lites of the vitamin.
Although cyanocobalamin was the first form in which vitamin B12 was isolated, it is not an important naturally occurring vitamer, but rather an artifact due to the presence of cyanide in the charcoal used in the extraction procedure. It is more stable to light than the other vitamers, and hence is used in pharmaceuti-cal preparations. Photolysis of cyanocobalamin in solution leads to the formation of aquocobalamin or hydroxocobalamin, depending on pH. Hydroxoco-balamin is also used in pharmaceutical preparations, and is better retained after parenteral administration than is cyanocobalamin.
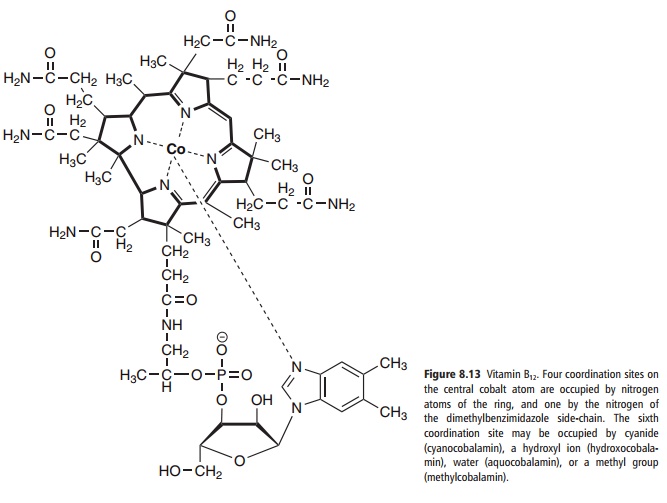
Vitamin B12 is found only in foods of animal origin, although it is also formed by bacteria. There are no plant sources of this vitamin. This means that strict vegetarians (vegans), who eat no foods of animal origin, are at risk of developing dietary vitamin B12 deficiency, although the small amounts of vitamin B12 formed by bacteria on the surface of fruits may be adequate to meet requirements. Prepa-rations of vitamin B12 made by bacterial fermentation that are ethically acceptable to vegans are readily available.
There are claims that yeast and some plants (espe-cially some algae) contain vitamin B12. This seems to be incorrect. The problem is that the officially recog-nized, and legally required, method of determining vitamin B12 in food analysis is a microbiological assay using organisms for which vitamin B12 is an essential growth factor. However, these organisms can also use some corrinoids that have no vitamin activity. There-fore, analysis reveals the presence of something that appears to be vitamin B12, but in fact is not the active vitamin and is useless in human nutrition. Biologi-cally active vitamin B12 has been identified in some preparations of algae, but this seems to be the result
Absorption and metabolism of vitamin B12
Absorption
Very small amounts of vitamin B12 can be absorbed by passive diffusion across the intestinal mucosa, but under normal conditions this is insignificant; the major route of vitamin B12 absorption is by attach-ment to a specific binding protein in the intestinal lumen.
This binding protein is intrinsic factor, so called because in the early studies of pernicious anemia it was found that two curative factors were involved: an extrinsic or dietary factor, which is now known to be vitamin B12, and an intrinsic or endogenously pro-duced factor. Intrinsic factor is a small glycoprotein secreted by the parietal cells of the gastric mucosa, which also secrete hydrochloric acid .
Gastric acid and pepsin play a role in vitamin B12 nutrition, serving to release the vitamin from protein binding, so making it available. Atrophic gastritis is a relatively common problem of advancing age; in the early stages there is failure of acid secretion but more or less normal secretion of intrinsic factor. This can result in vitamin B12depletion due to failure to release the vitamin from dietary proteins, although the absorption of free vitamin B12 (as in supplements or fortified foods) is unaffected. In the stomach, vitamin B12 binds to cobalophilin, a binding protein secreted in the saliva.
In the duodenum cobalophilin is hydrolyzed, releasing vitamin B12 to bind to intrinsic factor. Pan-creatic insufficiency can therefore be a factor in the development of vitamin B12 deficiency, since failure to hydrolyze cobalophilin will result in the excretion of cobalophilin-bound vitamin B12 rather than transfer to intrinsic factor. Intrinsic factor binds the various vitamin B12 vitamers, but not other corrinoids.
Vitamin B12 is absorbed from the distal third of the ileum. There are intrinsic factor–vitamin B12 binding sites on the brush border of the mucosal cells in this region; neither free intrinsic factor nor free vitamin B12 interacts with these receptors.
In plasma, vitamin B12 circulates bound to transco-balamin I, which is required for tissue uptake of the vitamin, and transcobalamin II, which seems to be a storage form of the vitamin.
Enterohepatic circulation of vitamin B12
There is a considerable enterohepatic circulation of vitamin B12. A third plasma vitamin B12 binding protein, transcobalamin III, is rapidly cleared by the liver, with a plasma half-life of the order of 5 min. This provides a mechanism for returning vitamin B12 and its metabolites from peripheral tissues to the liver, as well as for clearance of other corrinoids without vitamin activity, which may arise from either foods or the products of intestinal bacterial action, and be absorbed passively across the lower gut.
These corrinoids are then secreted into the bile, bound to cobalophilins; 3–8 μg (2.25–6 nmol) of vitamin B12 may be secreted in the bile each day, about the same as the dietary intake. Like dietary vitamin B12 bound to salivary cobalophilin, the biliary coba-lophilins are hydrolyzed in the duodenum, and the vitamin binds to intrinsic factor, so permitting reab-sorption in the ileum. Although cobalophilins and transcorrin III have low specificity, and will bind a variety of corrinoids, intrinsic factor binds only cobalamins, and so only the biologically active vitamin is reabsorbed.
Metabolic functions of vitamin B12
There are three vitamin B12-dependent enzymes in human tissues: methylmalonyl-CoA mutase (dis-cussed below under methylmalonic aciduria), leucine amino-mutase, and methionine synthetase.
Vitamin B12 deficiency: pernicious anemia
Vitamin B12 deficiency causes pernicious anemia; the release into the bloodstream of immature precursors of red blood cells (megaloblastic anemia). As dis-cussed below, vitamin B12 deficiency causes functional folate deficiency; this is what disturbs the rapid mul-tiplication of red blood cells, causing immature pre-cursors to be released into the circulation.
The other clinical feature of vitamin B12 deficiency, which is rarely seen in folic acid deficiency, is degen-eration of the spinal cord; hence the name “perni-cious” for the anemia of vitamin B12 deficiency. The spinal cord degeneration is due to a failure of the methylation of one arginine residue in myelin basic protein. About one-third of patients who present with megaloblastic anemia due to vitamin B12 deficiency also have spinal cord degeneration, and about one-third of deficient subjects present with neurological signs but no anemia.
The most common cause of pernicious anemia is failure of the absorption of vitamin B12, rather than dietary deficiency. Classical pernicious anemia is due to failure of intrinsic factor secretion, commonly the result of autoimmune disease, with production of anti-bodies against either the gastric parietal cells or intrin-sic factor. Atrophic gastritis with increasing age also leads to progressive failure of vitamin B12 absorption.
Dietary deficiency of vitamin B12 does occur, rarely, in strict vegetarians (vegans). The rarity of vitamin B12 deficiency among people who have no apparent dietary source of the vitamin suggests that bacterial contamination of water and foods with vitamin B12-producing organisms will provide minimally adequate amounts of the vitamin. The fruit bat develops vitamin B12 deficiency when fed on washed fruit under laboratory conditions, but in the wild micro-bial contamination of the outside of the fruit provides an adequate intake of the vitamin.
Vitamin B12 requirements
Most estimates of vitamin B12 requirements are based on the amounts given parenterally to maintain normal health in patients with pernicious anemia due to a failure of vitamin B12 absorption. This overestimates normal requirements, because of the enterohepatic circulation of vitamin B12; in people lacking intrinsic factor, or secreting anti-intrinsic factor antibodies, the vitamin that is excreted in the bile will be lost in the feces, whereas normally it is almost completely reabsorbed.
The total body pool of vitamin B12 is of the order of 2.5 mg (1.8 μmol), with a minimum desirable body pool of about 1 mg (0.3 μmol). The daily loss is about 0.1% of the body pool in subjects with normal entero-hepatic circulation of the vitamin; on this basis requirements are about 1–2.5 μg/day and reference intakes for adults range between 1.4 μg and 2.0 μg.
Assessment of vitamin B12 status
Measurement of plasma concentrations of vitamin B12 is the method of choice, and several simple and reliable radioligand binding assays have been devel-oped. A serum concentration of vitamin B12 below 110 pmol/l is associated with megaloblastic bone marrow, incipient anemia, and myelin damage. Below 150 pmol/l there are early bone marrow changes, abnormalities of the deoxyuridine monophosphate (dUMP) suppression test and methylmalonic aciduria after a valine load.
The Schilling test for vitamin B12 absorption
The absorption of vitamin B12 can be determined by the Schilling test. An oral dose of [57Co] or [58Co]-vitamin B12 is given with a parenteral flushing dose of 1 mg of non-radioactive vitamin to saturate body reserves, and the urinary excretion of radioactivity is followed as an index of absorption of the oral mate-rial. Normal subjects excrete 16–45% of the radioac-tivity over 24 h, whereas patients lacking the intrinsic factor excrete less than 5%.
The test can be repeated, giving the intrinsic factor orally together with the radioactive vitamin B12; if the impaired absorption was due to a simple lack of intrinsic factor, and not to anti-intrinsic factor anti-bodies in the saliva or gastric juice, then a normal amount of the radioactive material should be absorbed and excreted.
Methylmalonic aciduria
Methylmalonyl-CoA is formed as an intermediate in the catabolism of valine and by the carboxylation of propionyl-CoA arising in the catabolism of isoleu-cine, cholesterol, and (rare) fatty acids with an odd number of carbon atoms. Normally, it undergoes vitamin B12-dependent rearrangement to succinyl-CoA, catalyzed by methylmalonyl-CoA mutase. Vitamin B12 deficiency leads to an accumulation of methylmalonyl-CoA, which is hydrolyzed to methyl-malonic acid, which is excreted in the urine. Urinary excretion of methylmalonic acid, especially after a loading dose of valine, provides a means of assessing vitamin B12 nutritional status.
Related Topics