Chapter: Introduction to Human Nutrition: The Vitamins
Toxicity of vitamin A
Toxicity of vitamin A
There is only a limited capacity to metabolize vitamin A. Excessively high intakes lead to accumulation in the liver and other tissues, beyond the capacity of normal binding proteins, so that free, unbound, vitamin A is present. This leads to liver and bone damage, hair loss, vomiting, and headaches. Single doses of 60 mg of retinol are given to children in developing countries as a prophylactic against vitamin
A deficiency: an amount adequate to meet the child’s needs for 4–6 months. About 1% of children so treated show transient signs of toxicity, but this is considered an acceptable risk in view of the high prevalence and devastating effects of deficiency.
The chronic toxicity of vitamin A is a more general cause for concern; prolonged and regular intake of more than about 7.5–9 mg/day by adults (and signifi-cantly less for children) causes signs and symptoms of toxicity affecting:
●the central nervous system: headache, nausea, ataxia and anorexia, all associated with increased cerebro-spinal fluid pressure
●the liver: hepatomegaly with histological changes in the liver, increased collagen formation and hyperlipidemia
●bones: joint pains, thickening of the long bones, hypercalcemia and calcification of soft tissues the skin: excessive dryness, scaling and chapping of the skin, desquamation and alopecia.
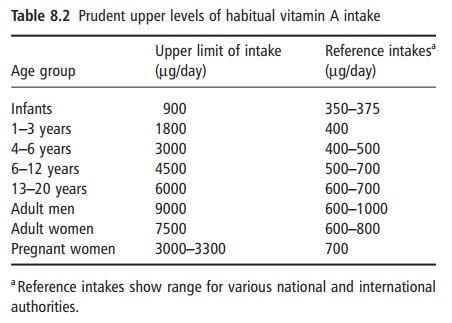
The recommended upper limits of habitual intake of retinol, compared with reference intakes, are shown in Table 8.2. As discussed above, habitual high intakes of vitamin A, albeit below these prudent upper levels of intake, may be associated with impaired respon-siveness to vitamin D, poor mineralization of bone and the early development of osteoporosis.
Teratogenicity of vitamin A
The synthetic retinoids (vitamin A analogues) used in dermatology are highly teratogenic. After women have been treated with them, it is recommended that contraceptive precautions be continued for 12 months, because of their retention in the body.
By extrapolation, it has been assumed that retinol is also teratogenic, although there is little evidence. In case– control studies, intakes between 2400 μg/day and 3300 μg/day during pregnancy have been associated with birth defects. Other studies have not demon-strated any teratogenic effect at this level of intake, and it has been suggested that the threshold plasma concentration associated with teratogenic effects is unlikely to be reached with intakes below 7500 μg/ day. Nevertheless, pregnant women are advised not to consume more than 3000 μg/day (American Pediatric Association recommendation) or 3300 μg (UK Department of Health recommendation).
Related Topics