Chapter: 12th Chemistry : UNIT 12 : Carbonyl Compounds and Carboxylic Acids
Structure of carboxyl group
Structure of carboxyl group:
The carboxyl group represent a planar arrangement of atoms. In – COOH
group, the centre carbon atom and both the oxygen atoms are in sp2
hybridisation. The three sp2 hybrid orbitals of the carbon atom overlap.
The two sp2 – hybridised orbitals of the carboxyl carbon
overlap with one sp2 hybridised orbital of each oxygen atom while
the third sp2 hybridised orbital of carbon overlaps with either a s
-orbital of H – atom or a sp2 – hybridised orbital of C –atom of the
alkyl group to form three σ- bonds.
Each of the two oxygen atoms and the carbon atom are left with one unhybridised
p – orbital which is perpendicular to the σ -
bonding skeleton.
All these three p – orbitals being parallel overlap to form a π- bond
which is partly delocalized between carbon and oxygen atom on one side, and
carbon and oxygen of the OH group on the other side. In other words, RCOOH may
be represented as a resonance hybrid of the following two canonical structures.
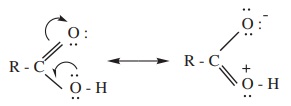
The carboxylic carbon is less electrophilic than carbonyl carbon because
of the possible resonance structure. i.e., delocalisation of lone pair
electrons from the oxygen in hydroxyl group.
Related Topics