Chemistry - General methods of preparation of aldehydes and ketones | 12th Chemistry : UNIT 12 : Carbonyl Compounds and Carboxylic Acids
Chapter: 12th Chemistry : UNIT 12 : Carbonyl Compounds and Carboxylic Acids
General methods of preparation of aldehydes and ketones
General methods of preparation of aldehydes and ketones
A) Preparation of aldehydes and ketones
1) Oxidation and catalytic dehydrogenation of alcohols
We have already learnt that the oxidation of primary alcohol gives
aldehydes and secondary alcohol gives a ketone. Oxidising agents such as
acidified Na2 Cr2O7 , KMnO4 , PCC
are used for oxidation. Oxidation using PCC yield aldehydes . Other oxidising
agents further oxidise the aldhydes / ketones in to carboxylic acids (Refer
Unit No. - 11 Oxidation of alcohols)
When vapours of alcohols are passed over heavy metal catalyst such as
Cu, Ag, alcohols give aldehydes and ketons. (Refer Unit No. - 11 Catalytic
dehydrogenation of alcohols.
2) Ozonolysis of alkenes
We have already learnt in XI th standard that the reductive ozonolysis
of alkenes gives aldehydes and ketones.
Alkenes react with ozone to form ozonide which on subsequent cleavage
with zinc and water gives aldehydes and ketones. Zinc dust removes H2O2
formed, which otherwise can oxidise aldehydes / ketones.
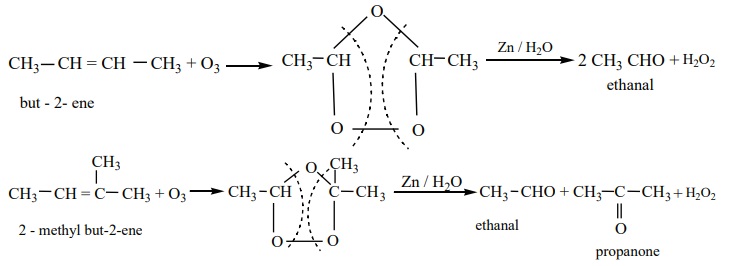
Terminal olefines give formaldehyde as one of the product.
Evaluate yourself – 1
What happens when the following alkenes are subjected to
reductive ozonolysis. 1) propene 2) 1 –
Butene 3) Isobutylene
3. Hydration of alkynes
We have already learnt in XI standard that the hydration of alkynes in
presence of 40% dilute sulpuric acid and 1% HgSO4 to give the
corresponding aldehydes / ketones.
a) Hydration of acetylene yields acetaldehyde
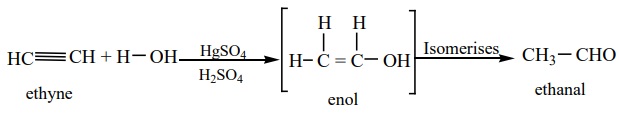
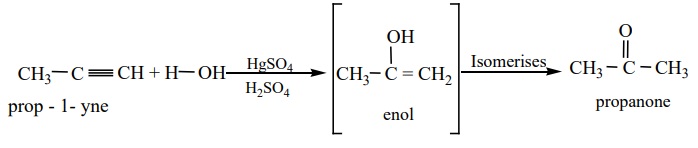
b) Hydration of alkynes, other than acetylene gives ketones
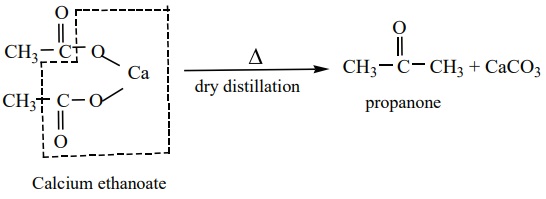
4. From calcium salts of carboxylic acids
Aldehydes and ketones may be prepared by the dry distillation of calcium
salts of carboxylic acids.
a) Aldehydes
are
obtained when the mixture of calcium salt of carboxylic acid and calcium formate is subjected to dry
distillation.
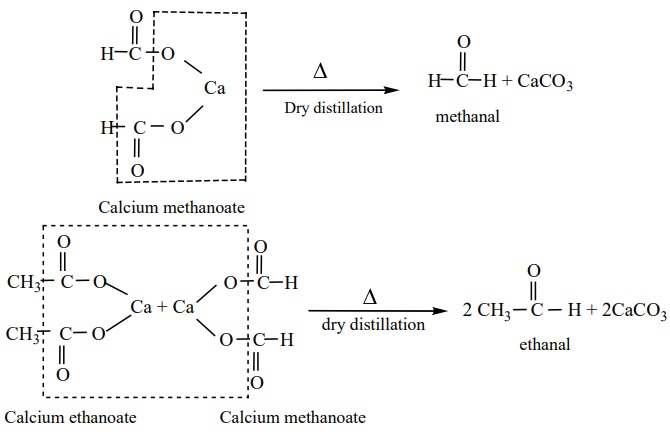
b) Symmetrical
ketones can be obtained by dry distillation of the calcium salt of carboxylic acid (except formic acid)
B) Preparation of aldehydes
1) Rosenmund reduction
a) Aldehydes
can be
prepared by the hydrogenation of acid chloride, in the presence of palladium supported by barium
sulphate. This reaction is called Rosenmund
reduction.
Example
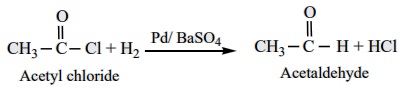
In this reaction, barium sulphate act as a catalytic poison to palladium
catalyst, so that aldehyde cannot be further reduced to alcohol.
Formaldehyde and ketones cannot be prepared by this method.
2. Stephen's reaction
When alkylcyanides are reduced using SnCl2 / HCl , imines are
formed, which on hydrolysis gives corresponding aldehyde.
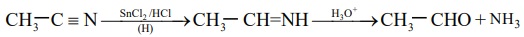
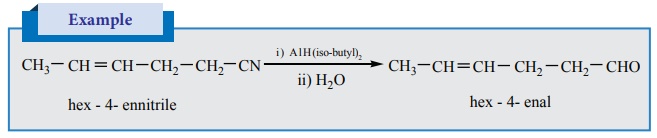
3. Selective reduction of cyanides
Diisobutyl aluminium hydride (DIBAL –H) selectively reduces the alkyl
cyanides to form imines which on hydrolysis gives aldehydes.
Example
C) Preparation of benzaldehyde
1. Side chain oxidation of toluene and its derivatives by strong oxidising
agents such as KMnO4 gives benzoic acid.
When chromylchloride is used as an oxidising agent, toluene gives
benzaldehyde. This reaction is called Etard
reaction. Acetic anhydride and CrO3 can also be used for this
reaction.
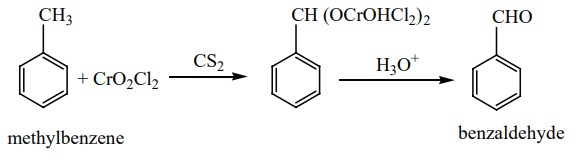
Oxidation of toluene by chromic oxide gives benzylidine diacetate which
on hydrolysis gives benzaldehyde.
2) Gattermann – Koch reaction
This reaction is a variant of Friedel – Crafts acylation reaction. In
this method, reaction of carbon monoxide and HCl generate an intermediate which
reacts like formyl chloride.
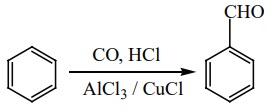
3) Manufacture of benzaldehyde from toluene
Side chain chlorination of toluene gives benzal chloride, which on
hydrolysis gives benzaldehyde.
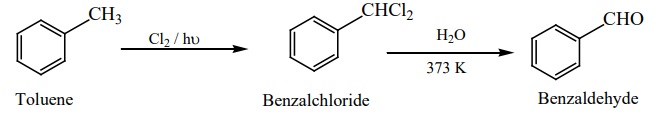
This is the commercial method for the manufacture of benzaldehye.
D) Preparation of ketones
1) Ketones can be prepared by the action of acid
chloride with dialkyl cadmium.
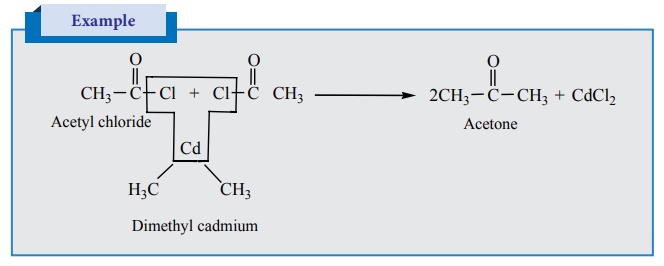
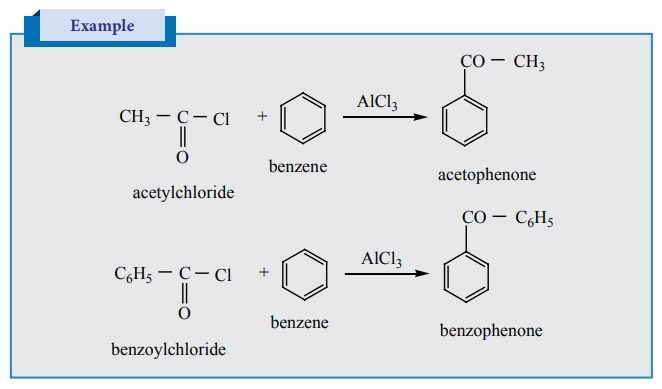
2) Preparation of phenyl ketones
Friedel – Crafts acylation
It is the best method for preparing alkyl aryl ketones or diaryl
ketones. This reaction succeeds only with benzene and activated benzene
derivatives.
Example
Related Topics