Chapter: 12th Chemistry : UNIT 12 : Carbonyl Compounds and Carboxylic Acids
Chemical properties of aldehydes and ketones
Chemical properties of aldehydes and ketones
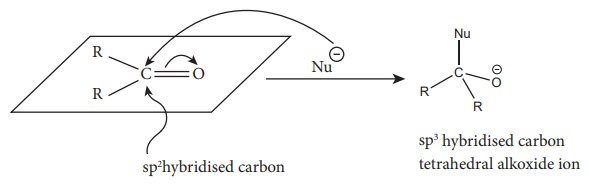
A) Nucleophilic addition reactions
This reaction is the most common reactions of aldehydes and ketones. The
carbonyl carbon carries a small degree of positive charge. Nucleophile such as
CN− can
attack the carbonyl carbon and uses its lone pair to form a new carbon –
nucleophile ' σ' bond,
at the same time two electrons from the carbon – oxygen double bond move to the
most electronegative oxygen atom. This results in the formation of an alkoxide
ion. In this process, the hybridisation of carbon changes from sp2
to sp3 .
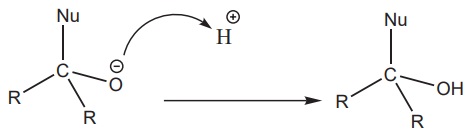
The tetrahedral intermediate can be protonated by water or an acid to
form an alcohol.
In general, aldehydes are more reactive than ketones towards
nucleophilic addition reactions due to +I and steric effect of alkyl groups.
Examples
1) Addition of HCN
Attack of CN− on carbonyl carbon followed by protonation gives cyanohydrins.
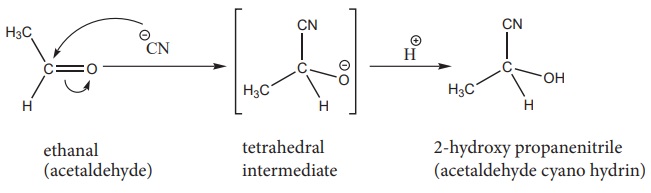
The cyanohydrins can be converted into hydroxy acid by acid hydrolysis.
Reduction of cyanohydrins gives hydroxy amines
2) Addition of NaHSO3
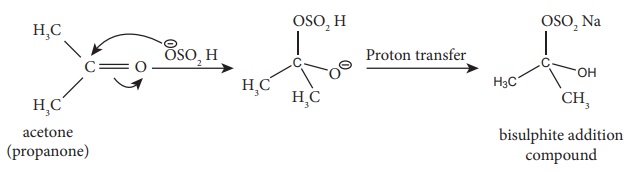
This reaction finds application in the separation and purification of
carbonyl compound. The bisulphate addition compound is water soluble and the
solution is treated with mineral acid to regenerate the carbonyl compounds .
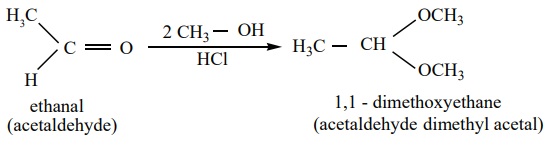
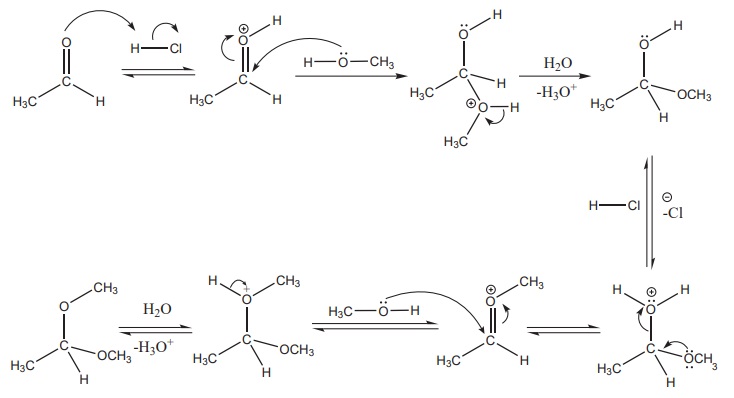
3) Addition of alcohol
When aldehydes / ketones is treated with 2 equivalents of an alcohol in
the presence of an acid catalyst to form acetals.
example
When acetaldehyde is treated with 2 equivalent of methanol in presence
of HCl, 1,1, - dimethoxy ethane is obtained.
Mechanisum
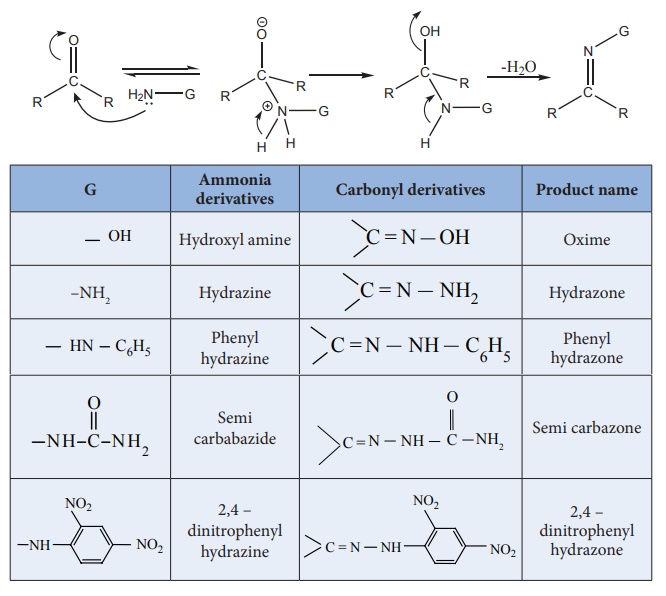
4) Addition of ammonia and its derivatives
When the nucleophiles, such as ammonia and its derivative 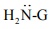 is treated with carbonyl compound, nuceophilic addition takes place, the
carbonyl oxygen atom is protonated and then elimination takes place to form
carbon – nitrogen double bond
is treated with carbonyl compound, nuceophilic addition takes place, the
carbonyl oxygen atom is protonated and then elimination takes place to form
carbon – nitrogen double bond 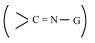
When G – alkyl, aryl, OH, NH2 , C6H5 NH, NHCONH2 etc...
i) Reaction
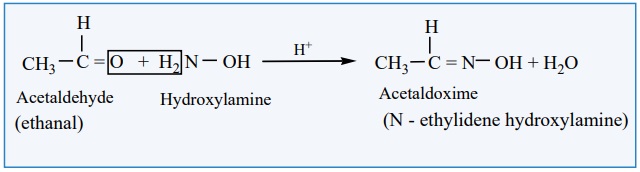
with hydroxyl amine
Aldehyde and ketones react with hydroxylamine to form oxime.
Example:
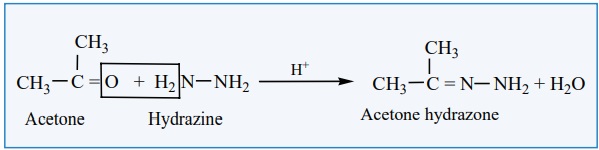
ii) Reaction
with hydrazine
Aldehydes and ketones react with hydrazine to form hydrazone.
Example:
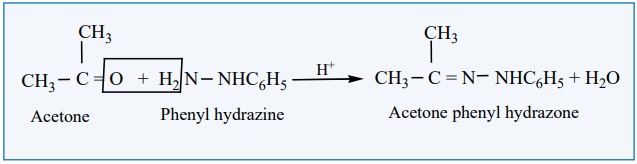
iii) Reaction
with phenyl hydrazine
Aldehydes and ketones react with phenyl hydrazine to form phenyl hydrazone.
Example:
5) Reaction with NH3
i) Aliphatic aldehydes (except formaldehyde) react with an ethereal
solution of ammonia to form aldimines.
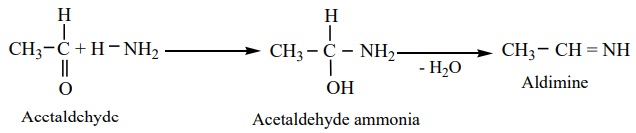
ii) Formaldehyde reacts with ammonia to form hexa methylene tetramine,
which is also known as Urotropine.
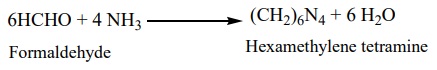
Structure
Uses
(i) Urotropine is used as a medicine to treat
urinary infection.
(ii) Nitration of Urotropine under controlled condition gives an
explosive RDX (Research and development explosive). It is also called cyclonite
or cyclotri methylene trinitramine.
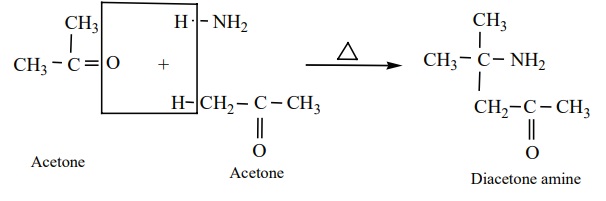
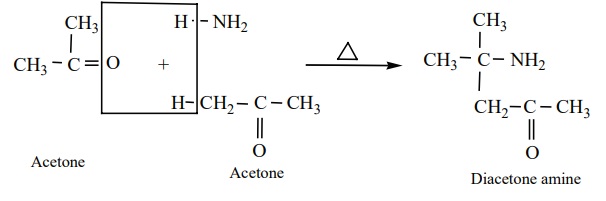
iii) Acetone reacts with ammonia to form diacetone amine.
iv) Benzaldehyde form a complex condensation product with ammonia.
B) Oxidation of aldehydes and ketones
a) Oxidation of aldehydes
Aldehydes are easily oxidised to carboxylic acid containing the same
number of carbon atom, as in parent aldehyde. The common oxidising agents are
acidified K2Cr2O7, acidic or alkaline KMnO4
or chromic oxide.
Example
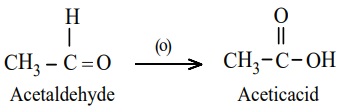
b) Oxidation of ketone
Ketones are not easily oxidised. Under drastic condition or with
powerful oxidising agent like Con.HNO3, H+/KMnO4,
H+/K2Cr2O7, cleavage of
carbon-carbon bond takes place to give a mixture of carboxylic acids having
less number of carbon atom than the parent ketone.
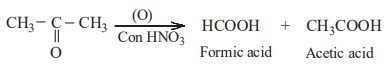
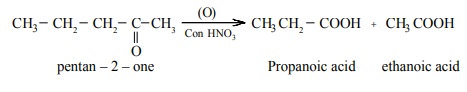
The oxidation of unsymmetrical ketones is governed by Popoff’s rule. It states that during
the oxidation of an unsymmetrical ketone, a (C–CO) bond is cleaved in such a
way that the keto group stays with the smaller alkyl group.
C) Reduction reactions
(i) Reduction to alcohols
We have already learnt that aldehydes and ketones can be easily reduced
to primary and secondary alcohols respectively. The most commonly used reducing
agents are Lithium Aluminium hydride (LiAlH4), and Sodium
borohydride (NaBH4).
a) Aldehyde
are reduced to primary alcohols.
Example
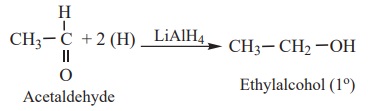
b) Ketone
are reduced to Secondary alcohols.
Example
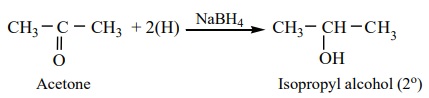
The above reactions can also be carried out with hydrogen in the
presence of metal catalyst like Pt, Pd, or Ni. LiAlH4 and NaBH4
do not reduce isolated carbon – carbon double bonds and double bond of benzene
rings. In case of α, β unsaturated aldehyde and ketones, LiAlH4
reduces only C = O group leaving C = C bond as such.
ii) Reduction to hydrocarbon
The carbonyl group of aldehydes and ketones can be reduced to methylene
group using suitable reducing agents to give hydrocarbons.
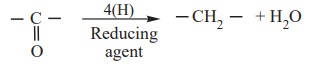
a) Clemmensen
reduction
Aldehydes and Ketones when heated with zinc amalgam and concentrated
hydrochloric acid gives hydrocarbons.
Example
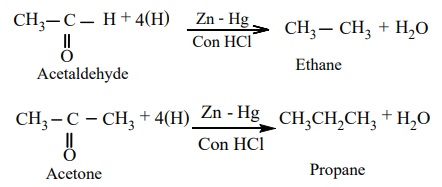
b) Wolf
Kishner reduction Aldehydes and Ketones when heated with hydrazine
(NH2NH2) and
sodium ethoxide, hydrocarbons are formed Hydrazine acts as a reducing agent and
sodium ethoxide as a catalyst.
Example
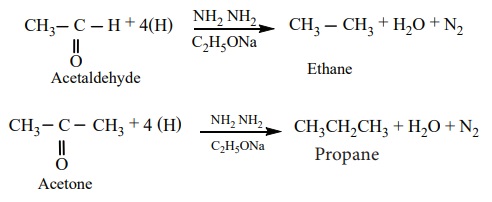
Aldehyde (or) ketones is first converted to its hydrazone which on
heating with strong base gives hydrocarbons.
(iii) Reduction to pinacols: Ketones, on reduction
with magnesium amalgam and water, are reduced to symmetrical diols known
as pinacol.
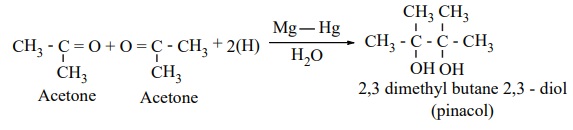
D) Haloform reaction
Acetaldehyde and methyl ketones, containing 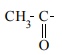 group, when treated with halogen and alkali give the corresponding haloform.
This is known as Haloform reaction.
group, when treated with halogen and alkali give the corresponding haloform.
This is known as Haloform reaction.
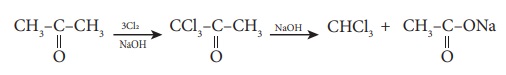
E) Reaction involving alkylgroup
i) Aldol condensation
The carbon attached to carbonyl carbon is called α - carbon and the hydrogen atom attached
to α - carbon
is called α -
hydrogen.
In presence of dilute base NaOH, or KOH, two molecules of an aldehyde or
ketone having α-hydrogen
add together to give β-
hydroxyl aldehyde (aldol) or β -
hydroxyl ketone (ketol). The reaction is called aldol condensation reaction. The aldol or ketol readily loses water
to give α,β – unsaturated compounds which are aldol condensation products.
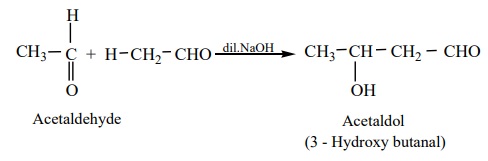
a) Acetaldehyde when warmed with dil NaOH gives β -
hydroxyl butyraldehyde (acetaldol)
Mechanism
The mechanism of aldol condensation of acetaldehyde takes place in three
steps.
Step 1 :
The carbanion is formed as the α - hydrogen
atom is removed as a proton by the base.

Step 2 :
The carbanion attacks the carbonyl carbon of another unionized aldehyde
to form an alkoxide ion.
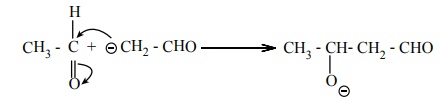
Step 3 :
The alkoxide ion formed is protonated by water to form aldol.
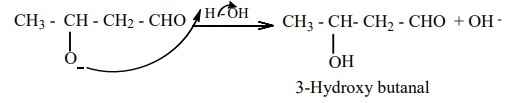
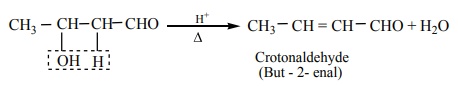
The aldol rapidly undergoes dehydration on heating with acid to form α - β
unsaturated aldehyde.
ii) Crossed aldol condensation
Aldol condensation can also take place between two different aldehydes
or ketones or between one aldehyde and one ketone such an aldol condensation is
called crossed or mixed aldol condensation. This reaction is
not very useful as the product is usually a mixture of all possible condensation products and cannot be separated easily.
Example :
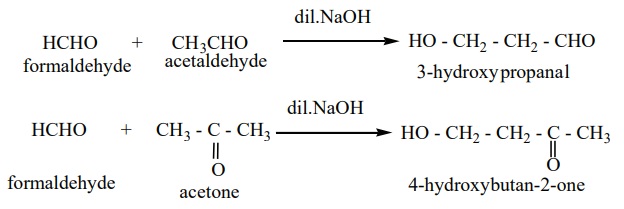
F) Some important reactions of benzaldehyde
i) Claisen – Schmidt Condensation
Benzaldehye condenses with aliphatic aldehyde or methyl ketone in the
presence of dil. alkali at room temperature to form unsaturated aldehyde or
ketone. This type of reaction is called Claisen – Schmidt condensation.
Example
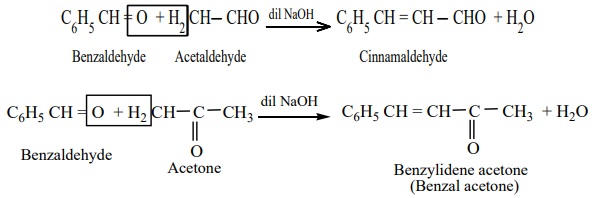
ii) Cannizaro reaction
In the presence of concentrated aqueous or alcoholic alkali, aldehydes
which do not have α -
hydrogen atom undergo self oxidation and reduction (disproportionation) to give
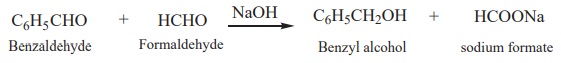
a mixture of alcohol and a salt of carboxylic acid. This reaction is called Cannizaro reaction.
Benzaldehyde on treatment with concentrated NaOH (50%) gives benzyl
alcohol and sodium benzoate.
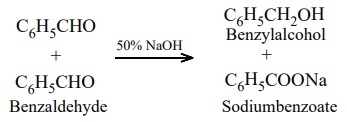
This reaction is an example disproportionation reaction
Mechanism of Cannizaro reaction
Cannizaro reaction involves three steps.
Step 1 : Attack of OH- on the carbonyl
carbon.
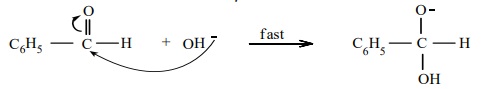
Step 2 : Hydride ion transfer
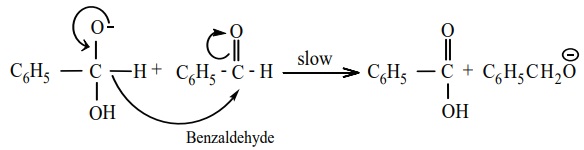
Step 3 : Acid – base reaction.
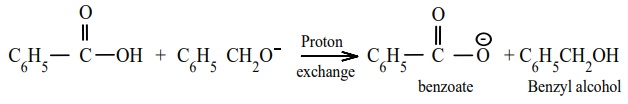
Cannizaro reaction is a characteristic of aldehyde having no α –
hydrogen.
Crossed Cannizaro reaction
When Cannizaro reaction takes place between two different aldehydes
(neither containing an α hydrogen atom), the reaction is called as crossed
cannizaro reaction.
In crossed cannizaro reaction more reactive aldehyde is oxidized and
less reactive aldehyde is reduced.
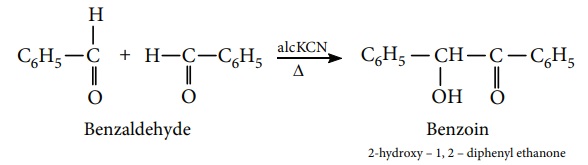
3) Benzoin condensation
The Benzoin condensation involves the treatment of an aromatic aldehyde
with aqueous alcoholic KCN. The products are a hydroxy ketone.
Example
Benzaldehyde reacts with alcoholic KCN to form benzoin
4) Perkins’ reaction
When an aromatic aldehyde is heated with an aliphatic acid anhydride in
the presence of the sodium salt of the acid corresponding to the anhydride, condensation
takes place and an α, β unsaturated
acid is obtained. This reaction is known as Perkin’s
reaction.
Example:
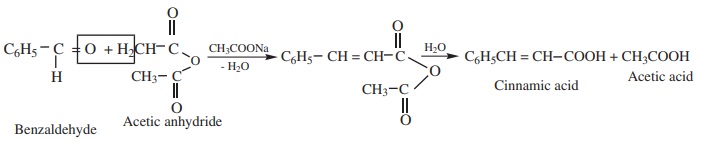
5) Knoevenagal reaction
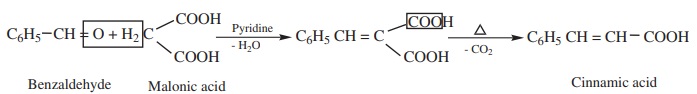
Benzaldehyde condenses with malonic acid in presence of pyridine forming
cinnamic acid, Pyridine act as the basic catalyst.
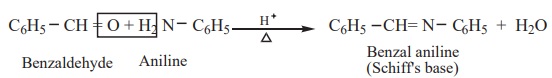
6) Reaction with amine
Aromatic aldehydes react with primary amines (aliphatic or aromatic) in
the presence of an acid to form schiff’s
base.
Example
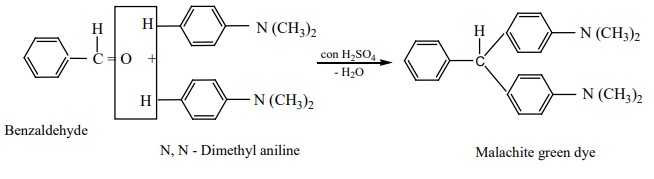
7) Condensation with tertiary aromatic amines
Benzaldehyde condenses with tertiary aromatic amines like N, N –
dimethyl aniline in the presence of strong acids to form triphenyl methane dye.
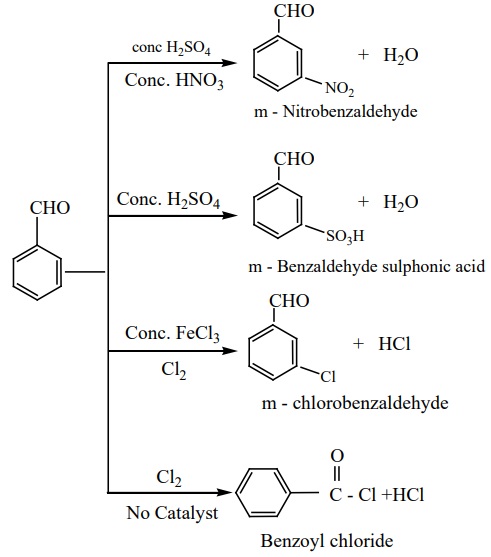
8) Electrophilic substitution reactions of benzaldehyde
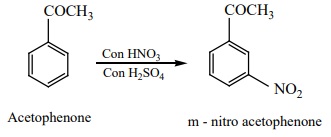
Electrophilic substitution reaction of acetophenone
Acetophenone reacts with Nitrating mixture to form m –
nitroacetophenone.
Related Topics