Chapter: Biotechnology Applying the Genetic Revolution: Pathway Engineering
Sterol Synthesis and Modification
STEROL
SYNTHESIS AND MODIFICATION
The sterol nucleus consists of four fused rings: three six-membered
rings and one five-membered ring (Fig. 13.17 shows examples). Plants, animals,
and fungi make a wide variety of sterols. Most bacteria do not make sterols,
perhaps because synthesis of sterols involves molecular oxygen and many
bacteria can grow anaerobically. Nonetheless, many bacteria do possess enzymes
that can modify the structures of sterols made by other organisms.
Sterols are used both as
membrane components (e.g., cholesterol) and as hormones. (Steroid hormones
activate gene expression via receptor proteins. Note that steroids are sterol derivatives having keto rather than hydroxyl
groups.) Sterols made or modified by other organisms may exert major effects on
animals by interfering with the roles of the natural steroid hormones. In
addition, a variety of humanmade molecules also act as steroid mimics. Although
steroid hormones regulate a wide variety of processes, the effects of
environmental sterols on reproduction have been the most visible. Indeed, many
of them act by binding to estrogen receptors. Consequently, these compounds are
sometimes referred to as xenoestrogens.
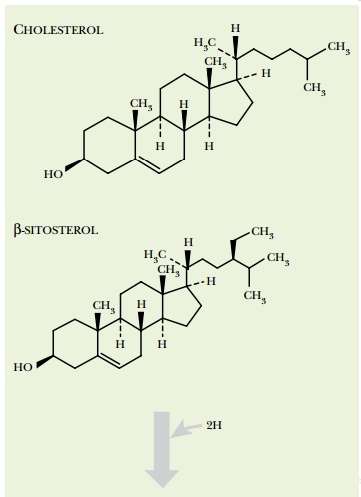
Cholesterol and its analogs also have significant effects on human health. For example, the level
of cholesterol in the blood has a major effect on the blocking of arteries by
fatty deposits (atherosclerosis). Absorption of cholesterol from food is
greatly inhibited by many plant sterols and especially by their hydrogenated
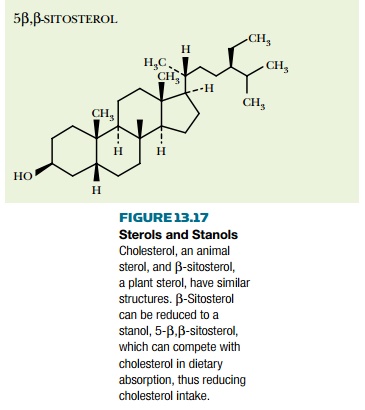
derivatives, or stanols. The sterol nucleus
has a double bond in the 5–6 position of the second ring (Fig. 13.17). When
this is reduced to a single bond, the resulting stanol has very different
biological properties. Eating plant material rich in such stanols reduces
dietary intake of cholesterol and so has a positive effect on health.
Consequently, plant sterols are sometimes hydrogenated to the corresponding
stanols during food processing. This may be done chemically or by using
microorganisms.
Many bacteria possess sterol-modifying
enzymes that carry out a wide variety of reactions. Some are able to reduce the
5–6 double bond, thus converting sterols into stanols. In addition, some
bacteria, especially species of Eubacterium,
can reduce cholesterol itself to coprostanol, which is poorly absorbed by the
intestine. Sterol-modifying enzymes are therefore of great potential use, both
in food processing and possibly in breaking down xenoestrogens for
decontamination of the environment. Several genes for sterol-modifying enzymes
from Eubacterium have been cloned and
expressed in E. coli, and the
corresponding enzymes have been purified and characterized.
Pathway engineering may also be of use in manufacturing sterols for therapeutic use. Steroids such as cortisone and prednisone are used to treat inflammatory diseases (arthritis, colitis, allergies, skin problems). Other uses for steroids include contraception and treating syndromes due to hormone deficiency. Today these steroids are made starting with plant sterols and using a mixture of chemical and microbial steps. Ultimately, the role of pathway engineering will be to provide a single microorganism that can carry out all the modification steps required in the manufacture of a particular product.
Related Topics