Chapter: Biotechnology Applying the Genetic Revolution: Pathway Engineering
Removal of Halogen, Nitro, and Sulfonate Groups
REMOVAL
OF HALOGEN, NITRO, AND SULFONATE GROUPS
A wide variety of aromatic
compounds are found in the environment, some naturally, others due to human
pollution. For almost all such compounds, bacteria can be isolated that degrade
them. Polychlorinated biphenyls (PCBs) are industrial pollutants. Other
chlorinated aromatics include selective herbicides such as 2,4-D
(2-4-dichlorophenoxyacetate). Many nitro and sulfonate derivatives are used in
the pharmaceutical, dye, and detergent industries. The chlorine, nitro, and
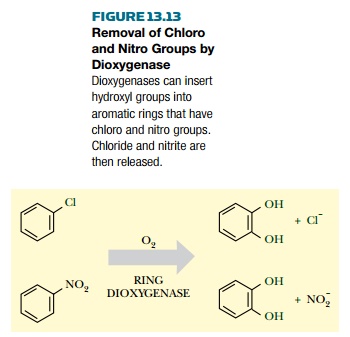
sulfonate groups may be removed during the dioxygenase reaction to release
chloride, nitrite, or bisulfite (Fig. 13.13). Ring dioxygenases from certain
bacteria will work on substituted rings, whereas chloro, nitro, or sulfonate
groups inhibit others. About 10% of the organic pollution in the river Rhine is
aromatic sulfonates from the German dye industry. Not surprisingly, many
bacteria isolated from the Rhine possess dioxygenases that are good at knocking
off sulfonate and nitro groups.
Related Topics