Chapter: Biotechnology Applying the Genetic Revolution: Pathway Engineering
Degradation of Aromatic Ring Compounds
DEGRADATION
OF AROMATIC RING COMPOUNDS
Many bacteria contain
plasmids allowing the metabolism of materials they would otherwise be unable to
use. Of major interest are a variety of pathways allowing the degradation of
both linear and cyclic hydrocarbons and related compounds. Many industrial
chemicals that are derived from petroleum, including various pesticides and
herbicides, contain aromatic rings. These chemicals, together with oil residues
or spills, are often responsible for significant pollution. Consequently, their
biodegradation by bacteria is of considerable practical importance.
Chemical compounds that
possess significant biological activity, but are foreign to the environment and
were introduced artificially by human industrial activity, are sometimes
referred to as xenobiotics.
Many species of Pseudomonas and related bacteria contain plasmids conferring the ability to grow using aromatic hydrocarbons as an energy source. Pseudomonas is widely distributed in soil, water, etc., and is an obligate aerobe. Different pseudomonads can use a wide range of substrates including alkanes, mono- and polycyclic aromatics, heterocyclics, phenols, terpenes, halogenated compounds, and so forth. Many of the degradation systems are of wide specificity and can handle many related substrates. The earliest pathways discovered were those for toluene/ xylene and naphthalene. The plasmids were originally named after their substrates (e.g., the TOL plasmid, now known as pTOL, degrades toluene) and are mostly large and capable of self-transfer between different related bacterial strains.
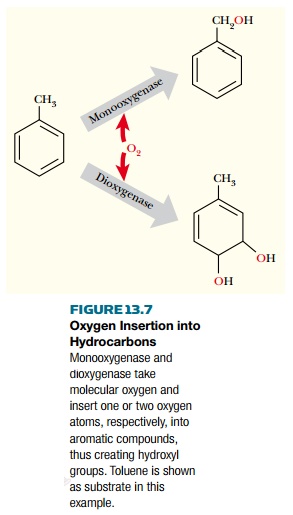
Most of these pathways
involve direct insertion of oxygen by oxygenases
(Fig. 13.7) as a key step. Monooxygenases insert a single oxygen atom to give one -OH group, whereas dioxygenases insert two oxygen atoms
and yield diols. When attacking
aromatic compounds, the monooxygenases generally attack the side chains,
whereas the dioxygenases attack the aromatic rings themselves.
Related Topics