Chapter: Biotechnology Applying the Genetic Revolution: Pathway Engineering
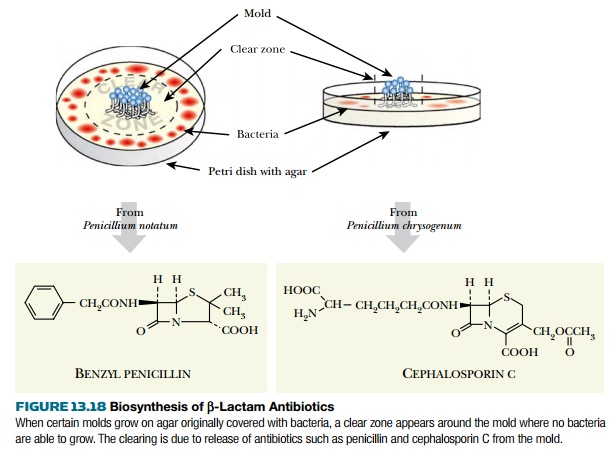
Biosynthesis of beta Lactam Antibiotics
BIOSYNTHESIS
OF β-LACTAM ANTIBIOTICS
Alexander Fleming discovered
the mold that makes penicillin in
the 1920s. The story is a classic case of chance favoring the prepared mind.
Fleming left Petri dishes containing bacterial cultures lying around long
enough to get moldy. He then noticed a clear zone, in which the bacteria had
been killed and had disintegrated, around a blue mold of the Penicillium group. He found that the
mold excreted a chemical toxic to bacteria but harmless to animals—penicillin. Fleming called the mold Penicillium
notatum. A related mold, Penicillium
chrysogenum, makes a related antibiotic called cephalosporin C. Both antibiotics are members of the b-lactam
family and are made by separate branches of the same biosynthetic pathway (Fig.
13.18).
The original β-lactams made by molds can be altered
chemically to give many different antibiotics. Although cephalosporin C itself
has only feeble antibacterial activity, it is the starting point for a vast
array of broad-spectrum antibiotics made by chemical modification. First,
cephalosporin C must be converted to 7-ACA (7- aminocephalosporanic acid),
which is not made by any known organism.
Originally this step was done
chemically and gave very low yields. Recently, a mold that makes cephalosporin
C was engineered to convert this to 7-ACA.
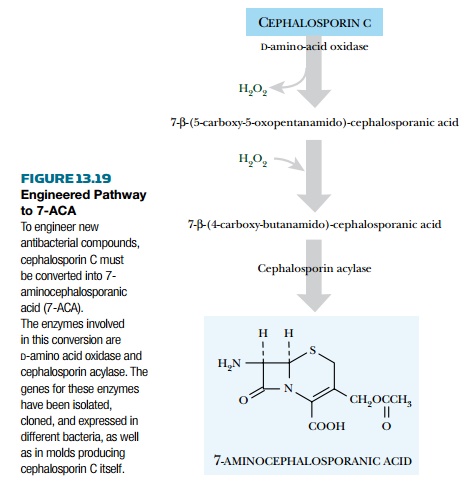
Two extra genes were inserted
to create the extended pathway (Fig. 13.19). The gene for D-amino-acid oxidase
was taken from a fungus (Fusarium solani) and the cephalosporin acylase gene
from a bacterium (Pseudomonas diminuta).
The 7-ACA is used as base
compound for a massive range of chemical modifications that provide antibiotics
with different properties. Among these are variants that are resistant to
bacterial β-lactamases and others that
penetrate bacterial cell walls better as well as antibiotics with better
pharmacological properties (e.g., superior absorption from the intestine).
Related Topics