Chapter: Biotechnology Applying the Genetic Revolution: Pathway Engineering
Ethanol, Elephants, and Pathway Engineering
ETHANOL,
ELEPHANTS, AND PATHWAY ENGINEERING
Humans weren’t the first to
appreciate alcohol. Elephants, monkeys, and other wild animals deliberately
consume fruit that has naturally fermented, yielding alcohol. Indeed, elephants
in both Africa and Asia may run amok after consuming fermented fruit.
Occasionally, elephants will even raid local villages and knock over houses to
“recover” fermented liquids from their human competitors! There is even a
species of the fruit fly Drosophila,
from the sherry-producing regions of Spain, that relies on sherry as its sole
source of nutrition. These insects spend their lives circling around in the
caves where sherry is processed and presumably do not need to fly straight. The
earliest cultural remains from human alcohol consumption date to about 5000 B.C. Analysis of a yellowish
residue found in Neolithic pottery from Iran showed that it was derived from
wine.
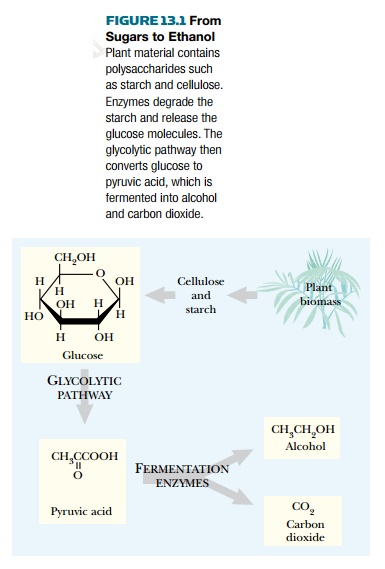
Alcohol is made from sugar
(Fig. 13.1). Sugars are components of the carbohydrates making up much of the
bulk of plant matter. So, in principle, alcohol can be made from almost any
plant-derived material. Yeast is used to ferment sugars derived from grain or
grapes, which produces an alcoholic liquid—the basis of beer or wine,
respectively. Distillation is then used to make concentrated liquors such as
whiskey or vodka. The lone exception to yeast is the use of a bacterium, Zymomonas, which ferments sugar from the
sap of the agave plant to give a liquid known as pulque. Distillation converts
this into tequila.
There is little need for
genetic engineering in the area of alcoholic drinks. However, alcohol may be
blended with gasoline to give “gasohol,” which works well in most internal combustion
engines. Thus conversion of waste biomass to fuel alcohol would not only get
rid of large amounts of waste material but would also reduce gasoline
consumption. If the United States converted the 100 million tons of waste paper
it generates each year into fuel-grade alcohol, this could replace 15% of the
gasoline used. Ethanol can also be made from corn, which is very economical
because many acres of corn are grown each year in the United States and a large
surplus is generated. Unlike wood pulp for paper, corn can be regenerated in 1
year.
The advantage of using Zymomonas and yeast is that they make
only alcohol during fermentation, whereas most microorganisms generate mixtures
of fermentation products. For example, Escherichia
coli makes a mixture of ethanol, acetate, succinate, lactate, and formate.
Although many fermentation products are potentially useful, purification is an
expensive drawback. The problem with Zymomonas
is that it lives entirely on glucose and lacks the enzymes to break down other
sugars, let alone those needed to degrade carbohydrate polymers such as starch
and cellulose. Yeast is almost as narrow in its growth requirements. Zymomonas grows faster than yeast and
makes alcohol faster as well. On the other hand, yeasts are more alcohol resistant and are therefore capable of
accumulating higher concentrations of ethanol in the medium before growth is
halted.
Genetic engineering is being
used to make improved strains of both yeast and Zymomonas that can use a wider range of sugars. In addition, genes
for enzymes capable of breaking down starch, cellulose, or other plant
polysaccharides can be inserted (see later discussion). Finally, these
organisms can also be engineered for improved resistance to alcohol or for
other properties that optimize growth and production under industrial
conditions.
Xylose is a five-carbon sugar
that is a major component of various polysaccharides (xylans) found in plant
cell walls (see later discussion). Vast amounts of waste material from plants
are available for possible biodegradation. Breakdown of the polysaccharide
polymers would release large amounts of xylose. Consequently, it is worthwhile
to develop strains of Zymomonas that efficiently ferment xylose to ethanol.
This has been done in two stages. First
the genes for metabolism of xylose itself must be introduced, because Zymomonas
does not naturally use this sugar. The xylA and xylB genes encode the enzymes
xylose isomerase and xylulose kinase, respectively, which convert xylose to
xylulose and then to xylulose 5-phosphate. These two genes were placed on a
shuttle vector that carries replication origins for both E. coli, in which the
genetic engineering was done, and Zymomonas (Fig. 13.2).
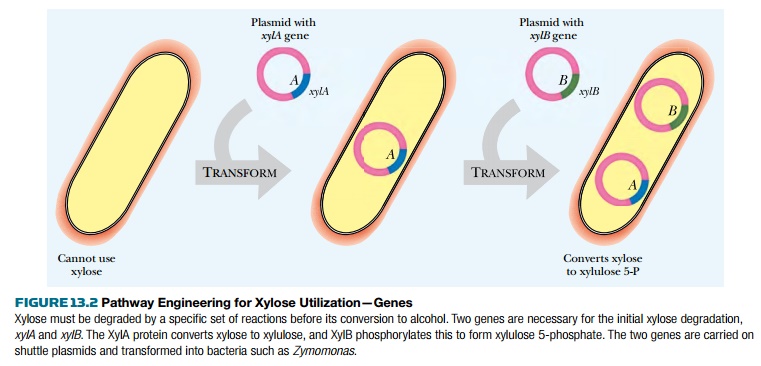
The strain with just the
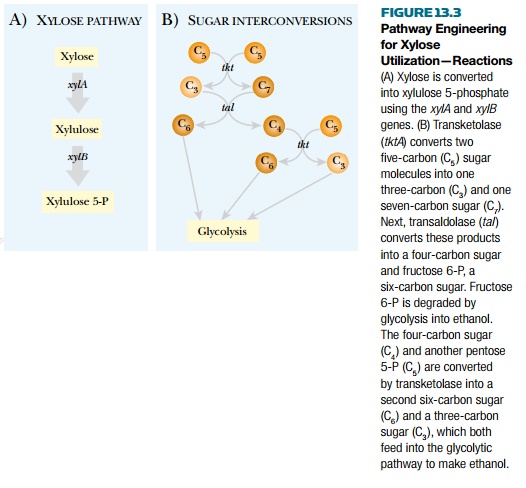
extra xylAB genes grew poorly because
it accumulated xylulose 5-phosphate as well as the phosphates of other pentose
sugars, including ribose 5-phosphate. The genes for transketolase (tktA) and transaldolase (tal), two enzymes that convert pentose
phosphates back into hexose phosphates, were then included on the plasmid,
under control of a separate promoter. The resulting Zymomonas was then able to convert xylose first to xylulose
5-phosphate and then to fructose 6-phosphate and glyceraldehyde 3-phosphate.
Finally, these central intermediates were fermented efficiently to ethanol
(Fig. 13.3).
Related Topics