Chapter: Pharmaceutical Biotechnology: Fundamentals and Applications : Formulation of Biotech Products, Including Biopharmaceutical Considerations
Primary Drying - Excipients Used in Parenteral Formulations of biotech Products
Primary Drying
In the primary drying stage (Fig. 6) sublimation of the water mass in the vial is initiated by lowering the pressure. The water vapor is collected on a condenser, with a (substantially) lower temperature than the shelf with the vials. Sublimation costs energy (about 2500 kJ/gram ice). Temperature drops are avoided by
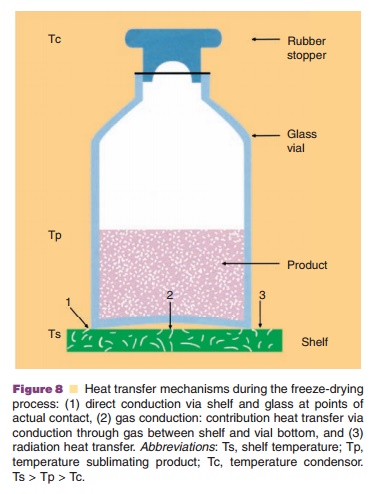
Heat is transferred to the vial through (i) direct shelf-vial contact (conductance), (ii) radiation, and (iii) gas conduction (Fig. 8). Gas conduction depends on the pressure: if one selects relatively high gas pressures, heat transport is promoted because of a high conductivity. But, it reduces mass transfer, because of a low driving force: the pressure between equilibrium vapor pressure at the interface between the frozen mass/dried cake and the chamber pressure (Pikal, 1990a).
During the primary drying stage one transfers heat from the shelf through the vial bottom and the frozen mass to the interface frozen mass/dry powder, to keep the sublimation process going. During this drying stage the vial content should never reach or exceed the eutectic temperature or glass transition temperature range. Typically a safety margin of 2 C to 5 C is used, otherwise the cake will collapse. Collapse causes a strong reduction in sublimation rate and poor cake formation. Heat transfer resistance decreases during the drying process as the transport distance is reduced by the retreating interface. With the mass transfer resistance (transport of water vapor), however, the opposite occurs. Mass transfer resistance increases during the drying process as the dry cake becomes thicker.
This situation makes it clear that parameters such as chamber pressure and shelf heating are not necessarily constant during the primary drying
process. They should be carefully chosen and adjusted as the drying process proceeds.
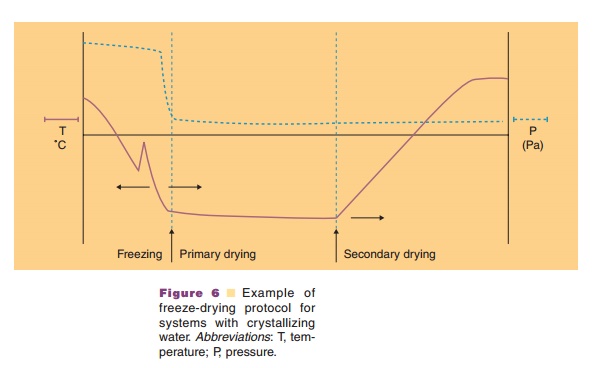
The eutectic temperature or glass transition temperature are parameters of great importance to develop a rationally designed freeze-drying protocol. Information about these parameters can be obtained by microscopic observation of the freeze-drying process, differential scanning calorimetry (DSC), or electrical resistance measurements.
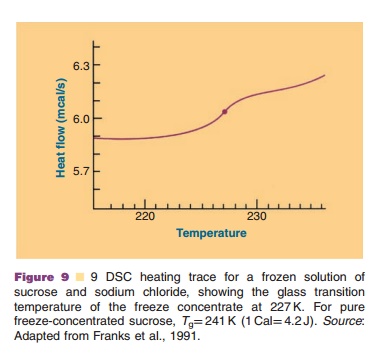
An example of a DSC scan providing informa-tion on the Tg is presented in Figure 9 (Franks et al., 1991). The Tg heavily depends on the composition of the system: excipients and water content. Lowering the water content of an amorphous system causes the Tg to shift to higher temperatures.
Related Topics