Chapter: Pharmaceutical Biotechnology: Fundamentals and Applications : Formulation of Biotech Products, Including Biopharmaceutical Considerations
Freeze Drying of Proteins - Excipients Used in Parenteral Formulations of biotech Products
Freeze Drying of Proteins
Proteins in solution often do not meet the preferred stability requirements for industrially produced phar-maceutical products (> 2 years), even when kept permanently under refrigerator conditions (cold chain). The abundant presence of water promotes chemical and physical degradation processes.
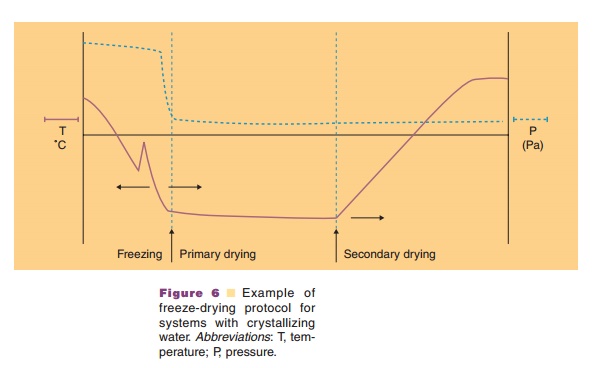
Freeze drying may provide the requested stability. During freeze-drying water is removed through sublimation and not by evaporation. Three stages can be discerned in the freeze-drying process: (i) a freezing step, (ii) the primary drying step, and (iii) the secondary drying step (Fig. 6). Table 2 explains what happens during these stages.
Freeze drying of a protein solution without the proper excipients causes, as a rule, irreversible damage to the protein. Table 3 lists excipients typically encountered in successfully freeze-dried protein products.


Related Topics