Chapter: 11th Chemistry : UNIT 8 : Physical and Chemical Equilibrium
Le-Chatelier's Principle
Le-Chatelier's
Principle
Many
chemical reactions that have industrial importance such as synthesis of ammonia
are reversible in nature. It is important to know the reaction conditions to
produce maximum yield. If a system at equilibrium is disturbed by modifying the
reaction conditions, then the system adjusts itself the new conditions to
re-establish the equilibrium.
The effect of change in reaction conditions such as temperature, pressure and concentration etc… on a system at equilibrium can be predicted by Le Chatelier-Braun principle.
It states that “If a system at equilibrium is disturbed,
then the system shifts itself in a direction that nullifies the effect of that
disturbance.”
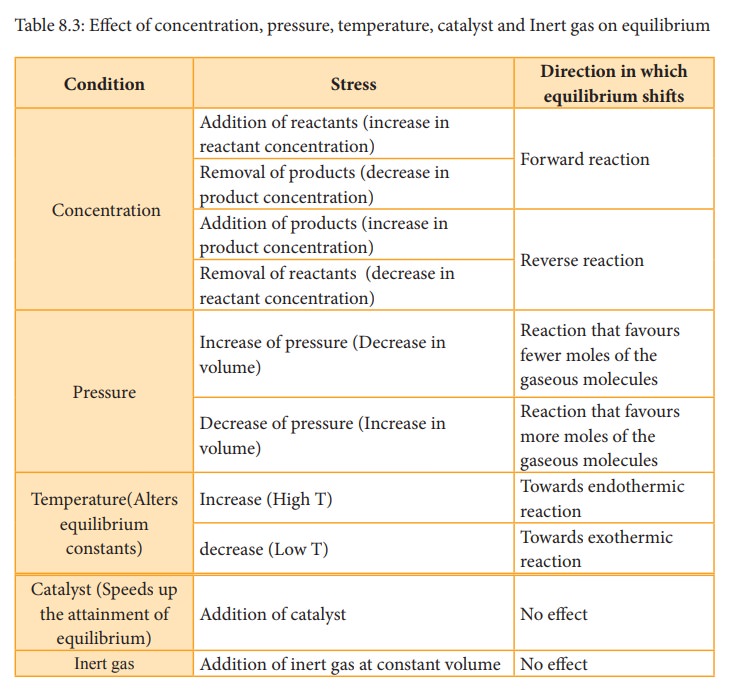
Effect of concentration
At
equilibrium, the concentration of the reactants and the products does not
change. The addition of more reactants or products to the reacting system at
equilibrium causes an increase in their respective concentrations.
According
to Le Chatelier’s principle, the effect of increase in concentration of a
substance is to shift the equilibrium in a direction that consumes the added
substance.
Let
us consider the reaction
H2(g) + I2(g) ⇌ 2HI(g)
The
addition of H2 or I2 to the equilibrium mixture, disturbs
the equilibrium. In order to minimize the stress, the system shifts the
reaction in a direction where H2and I2 are consumed.
i.e., the formation of additional HI would balance the effect of added
reactant. Hence, the equilibrium shifts to the right (forward direction) i.e.
the forward reaction takes place until the equilibrium is re-established.
Similarly, removal of HI (product) also favours the forward reaction.
If
HI is added to the equilibrium mixture, the concentration HI is increased, and
system proceeds in the reverse direction to nullify the effect of increase in
concentration of HI
Let
us explain the effect change in concentration by considering the formation of
HI from H2 and I 2. At equilibrium, the concentrations of
HI, H2 and I2 are 1 M, 0.2 M and 0.1 M respectively.
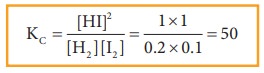
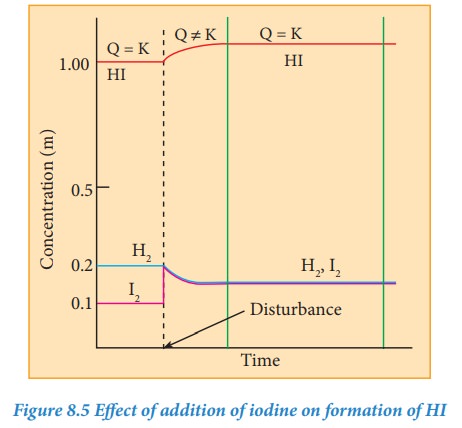
The
equilibrium was disturbed by adding 0.1 M iodine to the reaction mixture. After
sometime, the concentration of HI is found to be 1.092 M. Let us verify whether
the system proceeds to re-establish the equilibrium according to Le Chatelier’s
principle.
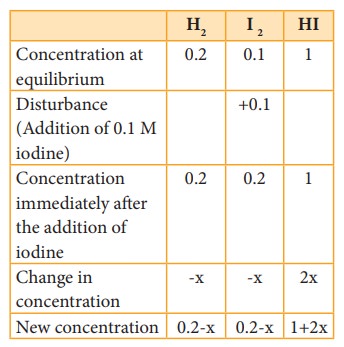
Now,
Concentration
HI = 1+2x = 1.092 M
2x
= 0.092
x
= 0.046 M
Therefore,
the concentration of hydrogen and iodine at this stage,
[H2] = 0.2 – X = 0.2 -0.046 = 0.154
[I2] = 0.2 – X = 0.2 -0.046 = 0.154
The
reaction quotient Q at this stage,
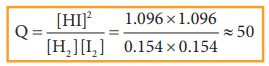
The
Q value is equal to Kc value.
So,
we can conclude that the system has reestablished the equilibrium at this stage
and the addition of iodine results in the increase in the HI concentration.
Effect of pressure
The
change in pressure has significant effect only on equilibrium systems with
gaseous components. When the pressure on the system is increased, the volume
decreases proportionately and the system responds by shifting the equilibrium
in a direction that has fewer moles of gaseous molecules.
Let
us consider the synthesis of ammonia from nitrogen and hydrogen.
N2(g) + 3H2(g) ⇌ 2NH3(g)
Let
the system be allowed to attain equilibrium in a cylinder with a piston. If we
press the piston down to increase the pressure, the volume decreases. The
system responds to this effect by reducing the number of gas molecules. i.e. it
favours the formation of ammonia. If we pull the piston upwards to reduce the
pressure, the volume increases. It favours the decomposition of ammonia

However,
when the total number of the moles of the gaseous reactants and the gaseous
products are equal, the change in pressure has no effect on system at
equilibrium.
Let
us consider the following reaction
H2(g) + I2 (g) ⇌ 2HI(g)
2 moles of reactants ⇒ 2 moles of product
Here,
the number of moles of reactants and products are equal. So, the pressure has
no effect on such equilibrium with Δng = 0.
Effect of temperature
If
the temperature of a system at equilibrium is changed, the system responds by
shifting the equilibrium in the direction that attempts to nullify the effect
of temperature.
Let
us consider the formation of ammonia.

In
this equilibrium, the forward reaction is exothermic i.e. the heat is liberated
while the reverse reaction is endothermic i.e. the heat is absorbed.
If
the temperature of the system is increased (by supplying heat energy), the
system responds by decomposing some of the ammonia molecules to nitrogen and
hydrogen by absorbing the supplied heat energy. Similarly, the system responds
to a drop in the temperature by forming more ammonia molecules from nitrogen
and hydrogen, which releases heat energy.
We
have already learnt that the change in pressure or concentration causes a change
in the equilibrium concentration such that the equilibrium constant remains the
same. However, in case of change in temperature, the equilibrium is
reestablished with a different equilibrium constant.
Effect of a catalyst
Addition of a catalyst does not affect the state of the equilibrium. The catalyst increases the rate of both the forward and the reverse reactions to the same extent. Hence, it does not change the equilibrium composition of the reaction mixture.
However, it speeds up the attainment of equilibrium by
providing a new pathway having a lower activation energy.
For
example, in the synthesis of NH3 by the Haber's process iron is used
as a catalyst.
Similarly,
in the contact process of manufacturing SO3, platinum or V2O5
is used as a catalyst
Effect of inert gas
When
an inert gas (i.e, a gas which does not react with any other species involved
in equilibrium) is added to an equilibrium system at constant volume, the total
number of moles of gases present in the container increases, that is, the total
pressure of gases increases. the partial pressure of the reactants and the
products are unchanged. Hence at constant volume, addition of inert gas has no
effect on equilibrium.
Table
8.3: Effect of concentration, pressure, temperature, catalyst and Inert gas on
equilibrium
Related Topics