Chapter: 11 th 12th std standard Bio Botany plant tree Biology Higher secondary school College Notes
Enzymes : Classification, Properties of Enzymes
Enzymes
Enzymes are biological catalysts, which activate various biochemical reactions of a living cell in a highly specific and precise manner. Enzymology is the study of enzymes. The name enzyme was coined by Kuhne in 1878. Pasteur recognised that some microorganisms like yeasts have got a capacity to cause fermentation in wine. In 1897, Buchner discovered that yeast extract could bring about fermentation of grape juice, like the living yeast cells. He also observed that the extract has lost its catalytic activity on boiling. He coined the word zymase for the active principle involved in the fermentation. The substance on which the enzyme acts is called substrate. Enzymes are essentially proteins but all proteins are not enzymes.
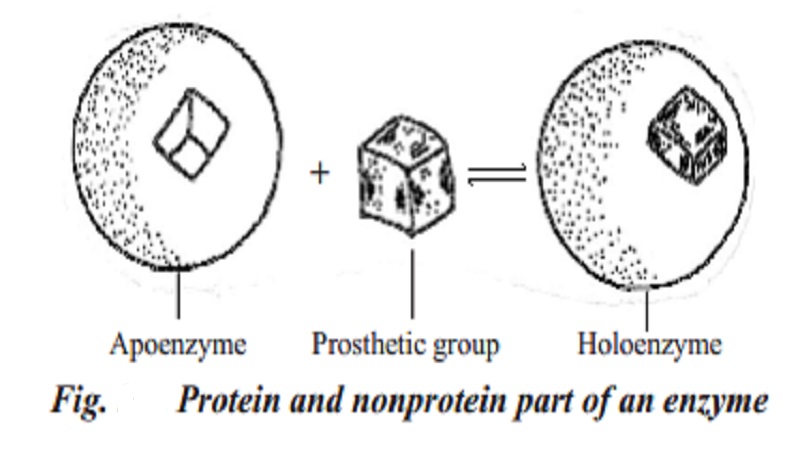
Many enzymes consists of a protein and non-protein component. They are called holoenzymes. The protein component of a holoenzyme is called apoenzyme and the non-protein component may be inorganic ions such as Mg2+ and Mn2+ . They are tightly attached to apoenzyme. They are called activators. The non-protein components may be organic substances such as NAD+, NADP+and FAD+. They are loosely attached to the apoenzyme. They are called coenzyme or cofactors. An enzyme will not function without its prosthetic group.
The specific region of the enzyme, which is involved in biochemical reaction, is termed active site. It is at the active site where the enzyme combines with its substrates to bring about biochemical reactions. An enzyme may have more than one active site. They are required only in very small quantities and yet capable of bringing about changes in large number of substrate molecules.
Classification of enzymes
Enzymes are classified based on the kinds of reactions they catalyze.
Oxireductase
These enzymes catalyze oxidation or reduction of their substrates by removing or adding electrons from or to substrates. eg. dehydrogenase, oxidase and reductase.
Transferase
These enzymes transfer a group from one substrate to other. eg. transaminase.
Hydrolases
These enzymes split larger molecules into smaller ones by the hydrolysis of water and breaking of specific covalent bonds. eg. carbohydrase.
AB + HOH -> AH+BOH (AB Substrate)
Lyases
These enzymes catalyze the cleavage of specific covalent bonds and removal of groups without hydrolysis. eg. Histidine decarboxylase.
Isomerases
The enzymes that catalyzes rearrangement of atoms within a molecule to form isomers are called isomerases. eg. phosphohexoisomerase.
Ligases
Ligases catalyze the formation of C - C, C - S, C - O and C - N bonds. The energy for the reaction is derived by the hydrolysis of ATP. eg. pyruvate carboxylase.
Properties of enzymes
1. Like inorganic catalysts, the enzymes are active in very small amounts and remain unchanged on completion of the reaction.
2. Enzymes are very specific in their action. i.e. a particular enzyme usually acts on a particular substrates to catalyze particular type of reaction. But some times, the same reaction may be catalyzed by more than one enzyme. Such enzymes are called isoenzymes.
3. Enzymes are very sensitive to heat. The temperature at which an enzyme shows its highest activity is called optimum temperature. The activity of enzyme declines both the above and below the optimum temperature. Protein part of the enzyme is the catalytic part. Temperature above 50'C normally denatures the protein and so the enzyme loses its activity.
4. The catalytic property of the enzyme is dependent on pH. The specific pH at which the enzyme activity is high is called optimum pH. Different types of enzymes have different optimum pH values. Most intracellular enzymes function will be around neutral pH. Tripsins are active in alkaline medium, diastase in neutral medium while pepsin in acidic medium.
5. Enzymes are sensitive to inhibitors. The chemical substance that combines with the enzymes and inhibits its catalytic activity is called an inhibitor. eg. Cyanides. A-><-B.
In most of the cases, the reactions catalyzed by the enzymes are reversible depending upon the requirements of the cell.
Theories explaining the mechanism of enzyme action
Two theories have been proposed to explain the mechanism and enzyme action. They are Fischer's Lock and key theory and Koshland's induced fit theory.
Fischer's Lock and key theory
Lock and key theory was proposed by Fisher. According to this theory, first a physical contact is made between the enzyme and the substrate. As only a specific key fits in a particular lock to open it, a specific substrate combines with the active site of specific enzyme. This combination leads to the production of enzyme - substrate complex. Then the enzyme acts on the substrate and changes it into products. After the reaction is over, enzyme is released from the enzyme - substrate complex and is ready to bind with another molecule of the substrate for further action. The cyclic reaction is summarized by the equation
E + S -><-[ES]-><-PS
( where, E - enzyme, S - substrate and P - product)
When a dissimilar substrate approaches the enzyme, it cannot combine with the active site of the enzyme, as a wrong key cannot open the lock. Thus, the enzyme action is inhibited.
Koshland's induced fit theory
Induced fit theory was pro-posed by Koshland. Proteins are not rigid. The substrate induces the enzyme to adjust its shape leading to the formation of enzyme sub-strate complex. Then, the enzyme acts on substrate and forms products. Many enzymes function in this way.
Mechanism of enzyme action
In a biochemical reaction, there is an energy barrier between the reactants and the products. Only those molecules which possess a certain amount of excess energy above the average energy of normal molecules are able to react to form products. This excess energy which a normal molecule must aquire in order to react is known as energy of activation (Ea). This energy of activation determines the rate of reaction. Higher the value of Ea, lower is the rate of reaction and greater stability. At higher
But in the case of enzyme catalyzed reaction, the rate of reaction is optimum at normal body temperature. Because all the molecules either energy-rich or energy-poor combine with the active site of the enzyme to form enzyme substrate complex. The latter breaks into enzyme and product. Thus, the enzyme acts by lowering the energy of activation of the reactions i.e. reducing the energy barrier and increases the rate of reaction.
Related Topics