Chapter: 11 th 12th std standard Bio Botany plant tree Biology Higher secondary school College Notes
Plasmolysis and Significance of Plasmolysis
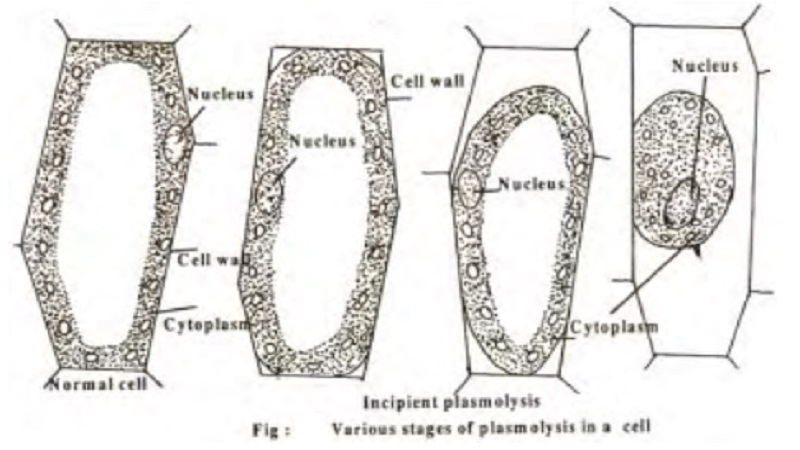
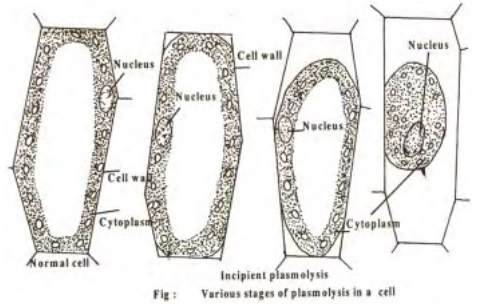
Plasmolysis
When a plant cell is placed in hypertonic solution, the process of exosmosis starts and water from the cell sap diffuses out into the solution of external medium. This causes a reduction in the tension of the cell wall and brings about the contraction of protoplasm due to the continuous loss of water. The protoplasm becomes rounded in shape due to contraction and such a cell is said to beplasmolysed and the phenomenon is referred to as plasmolysis. The initial stage of plasmolysis where the protoplasm just starts leaving the cell wall is called incipient plasmolysis.
When a completely plasmolysed cell is again placed in water or a hypotonic solution, endosmosis takes place and the protoplasm regains its original state and shape. i.e., the cell becomes fully turgid. This phenomenon is the reverse of plasmolysis and is calleddeplasmolysis.
Significance of Plasmolysis
1. Helps to understand the living nature of a cell.
2. Helps to preserve meat, jellies and used in pickling as their salting kills bacteria by plasmolysis.
3. Used to prove the permeability of cell wall and selectively permeable nature of plasma membrane.
Osmotic Pressure
Osmotic pressure of a solution is the pressure which must be applied to it in order to prevent the passage of solvent due to osmosis. In other words, it is that pressure which is needed to check the process of osmosis. The term osmotic potential is also used in place of osmotic pressure.
Turgor Pressure
When the plant cell is placed in water, it will swell but will not burst. Due to the negative osmotic potential of the cell sap, water moves into the cell and causes the plasma membrane to press against the cell wall. This pressure responsible for pressing the plasma membrane against cell wall is called turgor pressure.
Turgor pressure may also be defined as the hydrostatic pressure developed inside the cell on the cell wall due to endosmosis.
Wall Pressure
As a result of turgor pressure on the cell wall, the rigid cell wall exerts an equal pressure in the opposite direction called wall pressure. Under these conditions, the plant cell is said to be turgid.
When wall pressure becomes equal to turgor pressure, entry of water into the cell stops and the water potential (Psi denoted as �) becomes equal to that of the environment.
Diffusion Pressure Deficit (DPD) or Suction Pressure
The pressure exerted by diffusing particles is called diffusion pressure. When solute is added, the diffusion pressure of a solution is lowered. The amount by which diffusion pressure of a solution is lower than that of its pure solvent is known as diffusion pressure deficit which was described as suction pressure by Renner (1915). Recently a new term called water potential is used for DPD but with a negative value.
Related Topics