Chapter: 11 th 12th std standard Bio Botany plant tree Biology Higher secondary school College Notes
Cells : Absorption and Movement - Imbibition, Diffusion
Cells : Absorption and Movement - Imbibition, Diffusion
Absorption of water
Absorption of water occurs in plants through roots. The zone of water absorption in root is about 20 - 200 mm from the root tip and this is the root hair zone. The ultimate units of water absorption are the root hairs. The root hair is a unicellular tubular extension bound by an outer cell wall followed by plasma membrane, enclosing the protoplasm inside. The cytoplasm of the root hair contains a large central vacuole filled with cell sap.
Absorption of water by plants takes place due to the process, osmosis, which is a passive diffusion.
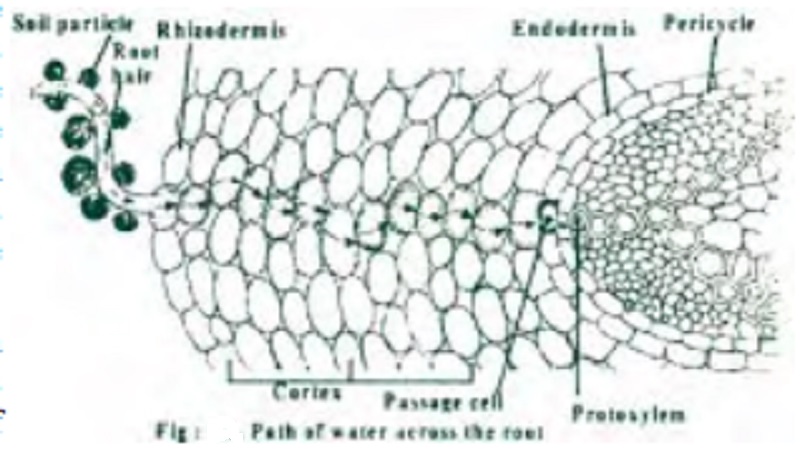
Path of water across the root
The root hairs are unicellular extensions of the roots found extending into the pore spaces of the soil particles. The pore spaces contain water and dissolved minerals in the form of soil solution. This water first gets adsorbed to the wall of the root hair, by imbibition thus wetting it. This forms a channel for further absorption of water by the living cells of the root in an active manner. From the root hair water reaches the cells of the rhizodermis, then through the cortical layers the water reaches the passage cells of the endodermis which are opposite to the protoxylem points. The water then passes through the parenchymatous pericycle and reaches the protoxylem.
The path of water is in a lateral direction and so is called lateral transport of water. Once the water reaches the xylem, it has to be transported in an upward direction to the shoot system and from there to the leaves. This is referred to as Ascent of Sap.
Imbibition
Imbibition is the uptake of water or other solvents by non-living substances such as gum, starch or wood causing swelling of these substances. Such substances are called imbibants. The phenomenon of imbibition creates a force called imbibitional force between the imbibant and the solvent. In plant cells, the cell wall is the imbibant which absorbs water and forms a channel for movement of water into the cell by diffusion and osmosis. Imbibition plays a very important role in most of the activities especially seed germination which involves absorption of water by seed coats, their swelling and rupture causing the emergence of the radicle andplumule.
Diffusion
Diffusion is the flow of matter, solid, liquids and gases from a region of higher concentration to a region of lower concentration until equilibrium is attained. Examples of diffusion are the smell of perfume, when we open a perfume bottle and the spread of colour when a crystal of potassium permanganate is put into a beaker of water.
When a substance undergoes diffusion, its particles start moving. When the moving particles counter a surface, the surface offers resistance to the impact of diffusing particles. This leads to development of pressure called diffusion pressure. Always diffusion occurs from a level of higher diffusion pressure to a level of lower diffusion pressure. A pure solvent has maximum diffusion pressure and addition of solutes lowers the diffusion pressure. The amount by which the diffusion pressure of a solution is lower than that of the pure solvent is called Diffusion Pressure Deficit (DPD). But the recent trend is to use the term water potential to explain diffusion of water.
Osmosis
Osmosis is a special type of diffusion of liquids. When two solutions of different concentrations are separated by a selectively permeable membrane, diffusion of water or solvent molecules takes place from the solution of lower concentration to the solution of higher concentration. This process is called Osmosis. In other words Osmosis is the diffusion of water or solvent from a region of its higher concentration to a region of its lower concentration through a selectively permeable membrane. This can also be expressed as the movement of water from a region of higher free energy of water or water potential to a region of lower free energy of water potential through a selectively permeable membrane.
Hypertonic, Hypotonic and Isotonic solutions
Imagine a system in which an aqueous solution A with high concentration of solute is separated by a selectively permeable membrane from an aqueous solution B with a low concentration of solute. Solution A is said to be hypertonic to solution B, and solution B hypotonic to solution A. In this situation, there will be a net movement of water or solvent molecules through the membrane from the hypotonic solution to the hypertonic solution by osmosis. This will continue until equilibrium is reached, at which point there is no further movement of water and the two solutions are described as isotonic.
Related Topics