Dual Nature of Radiation and Matter | Physics - Electron emission | 12th Physics : UNIT 8 : Dual Nature of Radiation and Matter
Chapter: 12th Physics : UNIT 8 : Dual Nature of Radiation and Matter
Electron emission
Electron emission
In metals, the electrons in the
outer most shells are loosely bound to the nucleus. Even at room temperature,
there are a large number of free electrons which are moving inside the metal in
a random manner. Though they move freely inside the metal, they cannot leave
the surface of the metal. The reason is that when free electrons reach the
surface of the metal, they are attracted by the positive nuclei of the metal.
It is this attractive pull which will not allow free electrons to leave the
metallic surface at room temperature.
In order to leave the metallic
surface, the free electrons must cross a potential barrier created by the
positive nuclei of the metal. The
potential barrier which prevents free electrons from leaving the metallic
surface is called surface barrier.
Free electrons possess some kinetic
energy and this energy is different for different electrons. The kinetic energy
of the free electrons is not sufficient to overcome the surface barrier.
Whenever an additional energy is given to the free electrons, they will have
sufficient energy to cross the surface barrier. And they escape from the
metallic surface. The liberation of
electrons from any surface of a substance is called electron emission.
The minimum energy needed for an electron to escape from the
metal surface is called work function of that metal. The work function of the metal is denoted by Ď•0 and
is measured in electron volt (eV).
The SI unit of energy is joule. But electron
volt is a commonly used unit of energy in atomic and nuclear physics.
One electron volt is defined as the
kinetic energy gained by an electron when accelerated by a potential difference
of 1 V.
1 eV = KE gained by the electron
= Work done by the electric field
= q V
= 1.602 × 10–19 C × 1 V
= 1.602 × 10–19 J
Suppose the maximum kinetic energy
of the free electron inside the metal is 0.5 eV and the energy needed to overcome the surface barrier of a metal
is 3 eV, then the minimum energy needed
for electron emission from the metallic surface is 3 – 0.5 = 2.5 eV. Here 2.5 eV is the work function of the metal.
The work function is different for
different metals and is a typical property of metals and the nature of their
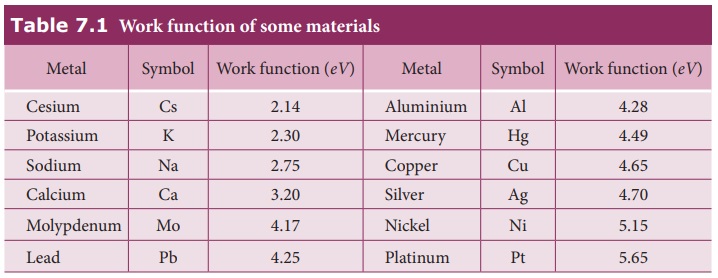
surface. Table 7.1 gives the approximate value of work function for various
metals. The material with smaller work function is more effective in electron
emission because extra energy required to release the free electrons from the
metal surface is smaller.
So the metal selected for electron
emission should have low work function. The electron emission is categorized
into different types depending upon the form of energy being utilized. There
are mainly four types of electron emission which are given below.
i) Thermionic emission
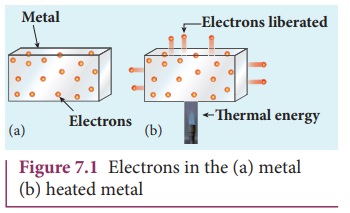
When a metal is heated to a high
temperature, the free electrons on the surface of the metal get sufficient
energy in the form of thermal energy so that they are emitted from the metallic
surface (Figure 7.1). This type of emission is known as thermionic emission.
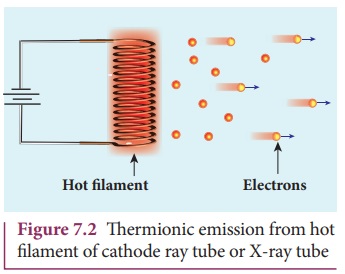
The intensity of the thermionic
emission (the number of electrons emitted) depends on the metal used and its
temperature. Examples: cathode ray
tubes, electron microscopes, X-ray tubes etc (Figure 7.2)
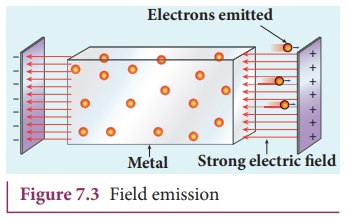
ii) Field emission
Electric field emission occurs when
a very strong electric field is applied across the metal. This strong field
pulls the free electrons and helps them to overcome the surface barrier of the
metal (Figure 7.3). Examples: Field
emission scanning electron microscopes, Field-emission display etc.
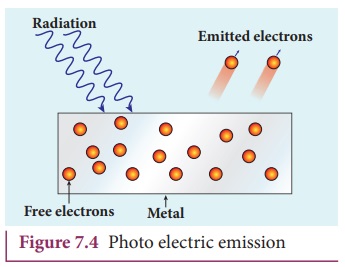
iii) Photo electric emission
When an electromagnetic radiation of
suitable frequency is incident on the surface of the metal, the energy is transferred
from the radiation to the free electrons. Hence, the free electrons get
sufficient energy to cross the surface barrier and the photo electric emission
takes place (Figure 7.4). The number of electrons emitted depends on the
intensity of the incident radiation. Examples:
Photo diodes, photo electric cells etc.
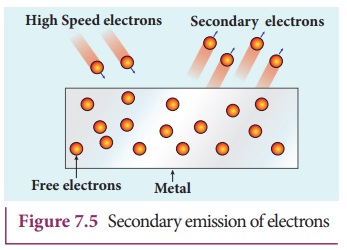
iv) Secondary emission
When a beam of fast moving electrons
strikes the surface of the metal, the kinetic energy of the striking electrons
is transferred to the free electrons on the metal surface. Thus the free
electrons get sufficient kinetic energy so that the secondary emission of
electron occurs (Figure 7.5). Examples: Image
intensifiers, photo multiplier tubes etc.
Related Topics