Chapter: 11th Botany : Chapter 14 : Respiration
Electron Transport Chain (ETC) (Terminal oxidation) - Plant Respiration
Electron Transport Chain (ETC)
(Terminal oxidation)
During
glycolysis, link reaction and Krebs cycle the respiratory substrates are oxidised
at several steps and as a result many reduced coenzymes NADH + H+ and
FADH2 are produced. These reduced coenzymes are transported to inner
membrane of mitochondria and are converted back to their oxidised forms produce
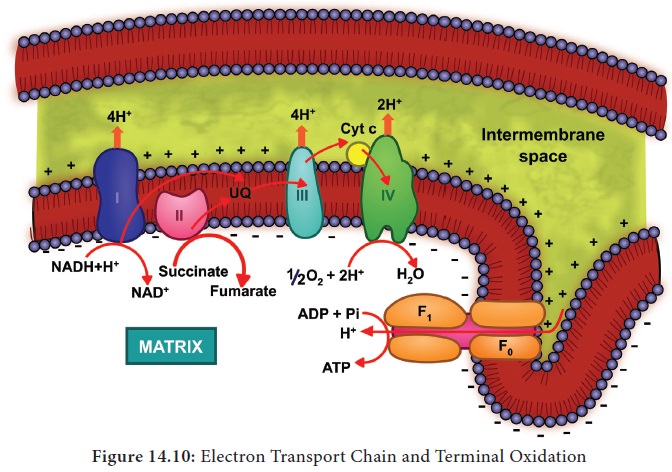
electrons and protons. In mitochondria, the inner membrane is folded in the
form of finger projections towards the matrix called cristae. In cristae many
oxysomes (F1 particles) are present which have electron transport
carriers are present. According to Peter
Mitchell’s Chemiosmotic theory this
electron transport is coupled to ATP
synthesis. Electron and hydrogen(proton) transport takes place across four
multiprotein complexes(I-IV). They are
1.
Complex-I
(NADH dehydrogenase).
It
contains a flavoprotein(FMN) and associated with non-heme iron Sulphur protein
(Fe-S). This complex is responsible for passing electrons and protons from
mitochondrial NADH (Internal) to
Ubiquinone(UQ).
In plants, an additional NADH dehydrogenase (External) complex is present on the outer surface of inner membrane
of mitochondria which can oxidise cytosolic NADH + H+.
Ubiquinone
(UQ) or Coenzyme Quinone(Co Q) is a small, lipid soluble electron, proton
carrier located within the inner membrane of mitochondria.
2.
Complex-II
(Succinic dehydrogenase) It contains FAD flavoprotein is associated with
non-heme iron Sulphur (Fe-S) protein. This complex receives electrons and
protons from succinate in Krebs cycle and is converted into fumarate and passes
to ubiquinone.
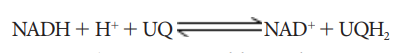
Succinate
+ UQ → Fumarate + UQH2
3.
Complex-III
(Cytochrome bc1 com-plex) This complex oxidises reduced
ubi-quinone (ubiquinol) and transfers the elec-trons through Cytochrome bc1
Complex (Iron Sulphur center bc1 complex) to cy-tochrome c.
Cytochrome c is a small pro-tein attached to the outer surface of inner
membrane and act as a mobile carrier to transfer electrons between complex III
to complex IV.
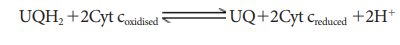
4. Complex IV (Cytochome c oxidase)
This
complex contains two copper centers (A and B) and cytochromes a and a3.
Complex IV is the terminal oxidase and brings about the reduction of 1/2 O2
to H2O.Two protons are needed to form a molecule of H2O
(terminal oxidation).
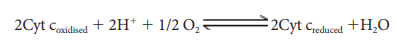
The
transfer of electrons from reduced coenzyme NADH to oxygen via complexes I to IV is coupled to the synthesis of ATP from ADP and inorganic phosphate (Pi) which is
called Oxidative phosphorylation. The F0F1-ATP
synthase (also called complex V)
consists of F0 and F1 . F1 converts ADP and Pi
to ATP and is attached to the matrix side of the inner membrane. F 0
is present in inner membrane and acts as a channel through which protons come
into matrix.
Oxidation of one molecule of NADH + H+ gives rise to 3 molecules of ATP and oxidation of one molecule FADH2 produces 2 molecules of ATP within a mitochondrion. But cytoplasmic NADH + H+ yields only two ATPs through external NADH dehydrogenase. Therefore, two reduced coenzyme (NADH + H+) molecules from glycolysis being extra mitochondrial will yield 2 x 2 = 4 ATP molecules instead of 6 ATPs (Figure 14.10) . The Mechanism of mitochondrial ATP synthesis is based on Chemiosmotic hypothesis. According to this theory electron carriers present in the inner mitochondrial membrane allow for the transfer of protons (H+). For the production of single ATP, 3 protons (H+) are needed. The terminal oxidation of external NADH bypasses the first phosphorylation site and hence only two ATP molecules are produced per external NADH oxidised through mitochondrial electron transport chain. However, in those animal tissues in which malate shuttle mechanism is present, the oxidation of external NADH will yield almost 3 ATP molecules.
Abnormal rise in respiratory rate of ripening in fruits is
called Climacteric. Examples are
apple, banana, mango, papaya, pear.
Complete
oxidation of a glucose molecule in aerobic respiration results in the net gain
of 36 ATP molecules in plants as
shown in table 14.2. Since huge amount of energy is generated in mitochondria
in the form of ATP molecules they are called ‘power house of the cell’.
In the case of aerobic prokaryotes due to lack of mitochondria each molecule of
glucose produces 38 ATP molecules.
Recent view
When the
cost of transport of ATPs from matrix into the cytosol is considered, the
number will be 2.5 ATPs for each NADH + H+ and 1.5 ATPs for each
FADH2 oxidised during electron transport system. Therefore, in
plant cells net yield of 30 ATP molecules for complete aerobic oxidation of one
molecule of glucose. But in those animal cells (showing malate shuttle
mechanism) net yield will be 32 ATP molecules.
Electron transport chain inhibitors
1. 2,4 DNP (Dinitrophenol) - It
prevents synthesis of ATP from ADP,
as it directs electrons from Co Q to O2
2. Cyanide - It prevents flow of electrons from Cytochrome a3 to O2
3. Rotenone - It prevents flow of electrons from NADH + H+/FADH2 to Co
Q
4. Oligomycin – It inhibits oxidative phosphorylation
Related Topics